
Spinal Trauma Devices Market Report 2026
Global Outlook – By Product (Spinal Fusion Devices, Spinal Biologics, Vertebral Compression Fracture Treatment Devices, Spinal Bone Growth Stimulators, Spinal Bone Grafts), By Technology (Fusion And Fixation Technology, Vertebral Compression Fracture Treatment, Motion Preservation Technologies), By Type Of Surgery (Open Surgery, Minimally Invasive Surgery), By End User (Hospitals And Clinics, Ambulatory Surgery Centers) – Market Size, Trends, Strategies, and Forecast to 2035
Spinal Trauma Devices Market Overview
• Spinal Trauma Devices market size has reached to $8.91 billion in 2025 • Expected to grow to $13.08 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: Surge In Road Accidents Fueling The Growth Of The Market Due To Increasing Incidence Of Spinal Injuries Requiring Specialized Devices • Market Trend: Advancements In Navigation-Enabled Surgical Instruments • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Spinal Trauma Devices Market?
Spinal trauma devices are specialized medical instruments used to manage spinal injuries or fractures resulting from trauma. They help in stabilizing and aligning the spine to facilitate recovery and include components such as screws, rods, plates, and cages. The main products of spinal trauma devices are spinal fusion devices, spinal biologics, vertebral compression fracture treatment devices, spinal bone growth stimulators, and spinal bone grafts. Spinal fusion devices are used to permanently join two or more vertebrae, thereby eliminating motion between them. These devices leverage various technologies, such as fusion and fixation, vertebral compression fracture treatment, and motion preservation technologies. Spinal trauma devices are utilized in different types of surgical procedures, including open surgery and minimally invasive surgery. They are used across a range of healthcare settings, including hospitals, clinics, and ambulatory surgery centers.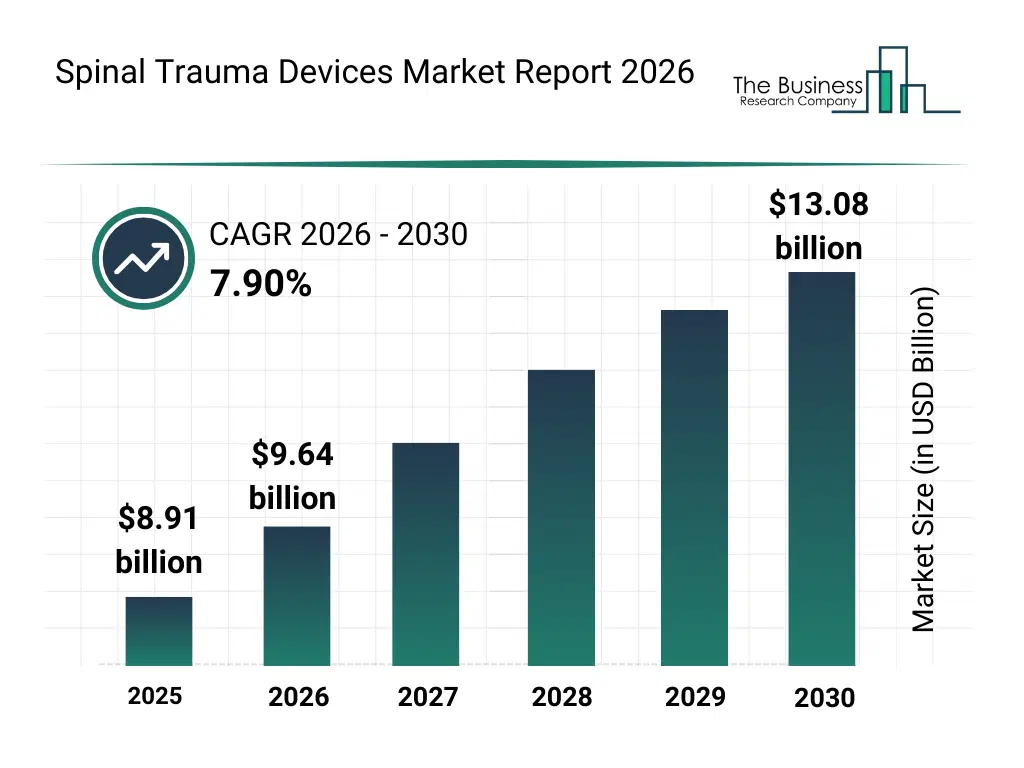
What Is The Spinal Trauma Devices Market Size and Share 2026?
The spinal trauma devices market size has grown strongly in recent years. It will grow from $8.91 billion in 2025 to $9.64 billion in 2026 at a compound annual growth rate (CAGR) of 8.1%. The growth in the historic period can be attributed to increasing incidence of road accidents and spinal injuries, growing use of spinal fusion devices in trauma care, advancements in vertebral fracture treatment technologies, rising availability of specialized trauma centers, increased adoption of surgical spinal stabilization.What Is The Spinal Trauma Devices Market Growth Forecast?
The spinal trauma devices market size is expected to see strong growth in the next few years. It will grow to $13.08 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to increasing demand for minimally invasive trauma surgeries, rising investments in advanced trauma care infrastructure, growing use of biologics in spinal healing, expansion of ambulatory spine surgery facilities, increasing focus on faster patient recovery outcomes. Major trends in the forecast period include increasing adoption of minimally invasive trauma fixation devices, rising use of advanced spinal biologics, growing preference for motion preservation technologies, expansion of rapid stabilization systems for acute trauma, enhanced focus on post-trauma spinal alignment.Global Spinal Trauma Devices Market Segmentation
1) By Product: Spinal Fusion Devices, Spinal Biologics, Vertebral Compression Fracture Treatment Devices, Spinal Bone Growth Stimulators, Spinal Bone Grafts 2) By Technology: Fusion And Fixation Technology, Vertebral Compression Fracture Treatment, Motion Preservation Technologies 3) By Type Of Surgery: Open Surgery, Minimally Invasive Surgery 4) By End User: Hospitals And Clinics, Ambulatory Surgery Centers Subsegments: 1) By Spinal Fusion Devices: Pedicle Screw Systems, Plates, Rods, Cages 2) By Spinal Biologics: Bone Graft Substitutes, Platelet-Rich Plasma (PRP), Bone Morphogenetic Proteins (BMPs) 3) By Vertebral Compression Fracture Treatment Devices: Balloon Kyphoplasty Devices, Vertebroplasty Devices 4) By Spinal Bone Growth Stimulators: Implanted Bone Growth Stimulators, External Bone Growth Stimulators, Ultrasonic Bone Growth Stimulators 5) By Spinal Bone Grafts: Autografts, Allografts, Synthetic Bone GraftsWhat Is The Driver Of The Spinal Trauma Devices Market?
The rising number of road accidents is expected to propel the growth of the spinal trauma devices market going forward. Road accidents refer to traffic collisions involving vehicles, pedestrians, or cyclists, leading to severe injuries, including spinal trauma. The rising number of road accidents is due to reckless driving behaviors, such as speeding, distracted driving, driving under the influence, and ignoring traffic signals, which significantly increase the risk of collisions and injuries. Road accidents can cause spinal injuries through sudden impact or forceful movement, necessitating the use of spinal trauma devices to stabilize and protect the spine. For instance, in October 2025, according to Road Safety Data Hub, an Australia-based government initiative to provide a central source of road safety data, the national road fatality rate for the 12 months ending October 2025 was 4.9 deaths per 100,000 population, representing a 5.2% year-on-year increase versus the prior comparable period. Therefore, the rising number of road accidents is driving the growth of the spinal trauma devices industry.Key Players In The Global Spinal Trauma Devices Market
Major companies operating in the spinal trauma devices market are Johnson & Johnson, Medtronic plc, Stryker Corporation, Zimmer Biomet Holdings Inc., Smith & Nephew plc, Globus Medical Inc., MicroPort Scientific Corporation, ZimVie Inc., Orthofix Medical Inc., Alphatec Holdings Inc., RTI Surgical Inc., B. Braun SE, Xtant Medical Holdings Inc., Spineart SA, GS Medical, Genesys Orthopedic Systems LLC, ChoiceSpine LLC, DePuy Synthes, NuVasive Inc., SeaSpine Holdings Corporation, K2M Group Holdings Inc., Life Spine Inc.Global Spinal Trauma Devices Market Trends and Insights
Major companies operating in the spinal trauma devices market are focusing on developing advanced products, such as integrated navigation-enabled instruments, to enhance surgical precision, streamline workflows, and improve patient outcomes. An integrated navigation-enabled instrument system combines advanced implants with instruments designed for seamless integration with enabling technologies, aiming to address complex spine conditions more effectively. For instance, in April 2024, DePuy Synthes, a US-based medical device manufacturing company, launched the TriALTIS Spine System. This next-generation posterior thoracolumbar pedicle screw system offers a comprehensive implant portfolio and advanced instrumentation designed for integration with power and navigation technologies. This combines a new portfolio of implants with a digital ecosystem, the TriALTIS Spine System aims to address unmet clinical needs and help surgeons achieve more consistent outcomes in treating complex spine conditions, including degenerative, tumor, trauma, and deformity pathologies.What Are Latest Mergers And Acquisitions In The Spinal Trauma Devices Market?
In August 2024, Stryker Corporation, a US-based medical technologies company, acquired Vertos Medical for an undisclosed amount. With this acquisition, Stryker Corporation aims to strengthen its portfolio in minimally invasive spinal treatments and expand its presence in the growing interventional spine market by leveraging Vertos Medical's innovative solutions for lumbar spinal stenosis. Vertos Medical is a US-based medical device company focuses on treating chronic lower back pain from lumbar spinal stenosis.Regional Outlook
North America was the largest region in the spinal trauma devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Spinal Trauma Devices Market?
The spinal trauma devices market consists of sales of pinal rods, screws, plates, cages, and artificial discs. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Spinal Trauma Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $9.64 billion |
| Revenue Forecast In 2035 | $13.08 billion |
| Growth Rate | CAGR of 8.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Technology, Type Of Surgery, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Medtronic plc, Stryker Corporation, Zimmer Biomet Holdings Inc., Smith & Nephew plc, Globus Medical Inc., MicroPort Scientific Corporation, ZimVie Inc., Orthofix Medical Inc., Alphatec Holdings Inc., RTI Surgical Inc., B. Braun SE, Xtant Medical Holdings Inc., Spineart SA, GS Medical, Genesys Orthopedic Systems LLC, ChoiceSpine LLC, DePuy Synthes, NuVasive Inc., SeaSpine Holdings Corporation, K2M Group Holdings Inc., Life Spine Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
