
Surgical Mesh Implants Market Report 2026
Global Outlook – By Type (Non-Absorbable Surgical Mesh, Absorbable Surgical Mesh, Other Types), By Surgical Procedure Type (Laparoscopic Surgery, Open Surgery, Robotic-assisted Surgery, Endoscopic Surgery), By Material (Polypropylene, Polyester, Polytetrafluoroethylene, Other Materials), By Application (Hernia Repair, Pelvic Organ Prolapse Repair, Gastroesophageal Reflux Disease (GERD) Repair, Other Surgical Applications), By End-User (Hospitals, Ambulatory Surgical Centers (ASCs), Specialized Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Surgical Mesh Implants Market Overview
• Surgical Mesh Implants market size has reached to $3.97 billion in 2025 • Expected to grow to $5.46 billion in 2030 at a compound annual growth rate (CAGR) of 6.5% • Growth Driver: Minimally Invasive Surgeries Driving The Market Growth Due To Increasing Adoption And Improved Patient Outcomes • Market Trend: Surgical Innovations Drive Demand For Fully Absorbable Repair Solutions • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Surgical Mesh Implants Market?
Surgical mesh implants refer to medical devices made from synthetic or biological materials that are used to provide additional support to weakened or damaged tissue during surgical procedures. They are commonly employed in soft tissue reinforcement surgeries and other reinforcement surgeries. These implants help promote tissue growth and healing while restoring structural integrity. The main types of surgical mesh implants include non-absorbable surgical mesh, absorbable surgical mesh, and others. Non-absorbable surgical mesh refers to a type of mesh made from synthetic materials that remain permanently in the body to provide long-term support to weakened or damaged tissue. The multiple surgical procedure types include techniques such as laparoscopic surgery, open surgery, robotic-assisted surgery, and endoscopic surgery, and materials used in surgical mesh implants are polypropylene, polyester, polytetrafluoroethylene, and others. The various applications involved are hernia repair, pelvic organ prolapse repair, gastroesophageal reflux disease (GERD) repair, and others, and they are used by several end-users such as hospitals, ambulatory surgical centers (ASCs), specialized clinics, and research institutions.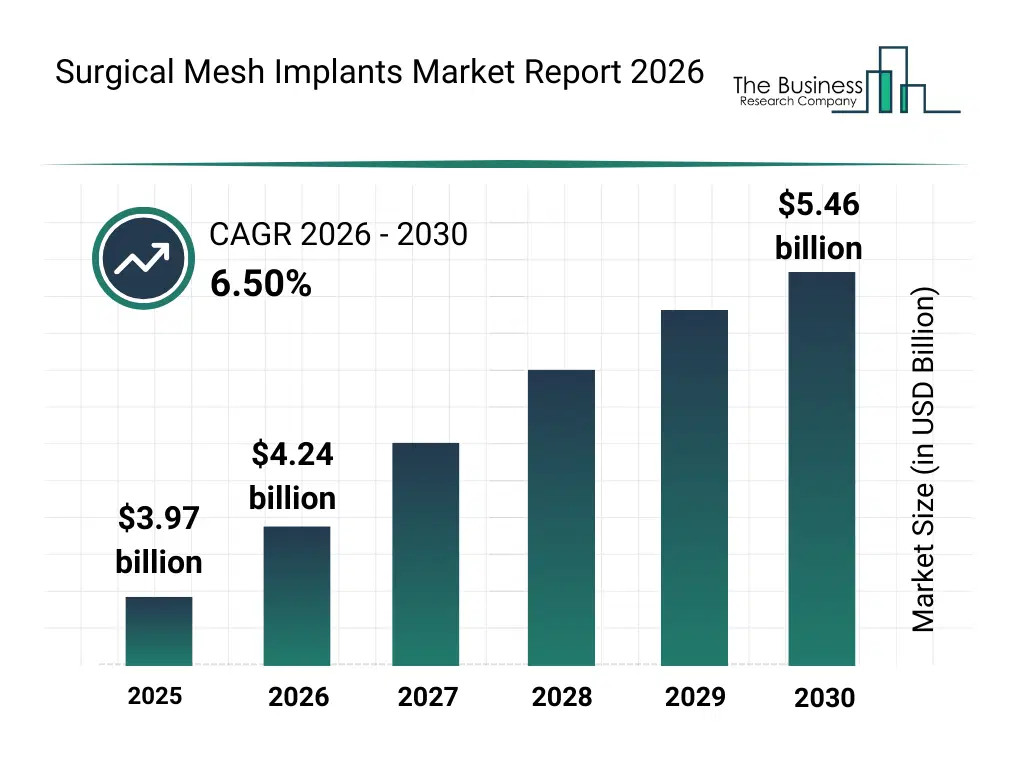
What Is The Surgical Mesh Implants Market Size and Share 2026?
The surgical mesh implants market size has grown strongly in recent years. It will grow from $3.97 billion in 2025 to $4.24 billion in 2026 at a compound annual growth rate (CAGR) of 6.8%. The growth in the historic period can be attributed to increasing prevalence of hernia and pelvic organ prolapse, advancements in surgical mesh materials, rising hospital infrastructure investment, growth in laparoscopic and open surgeries, demand for cost-effective surgical solutions.What Is The Surgical Mesh Implants Market Growth Forecast?
The surgical mesh implants market size is expected to see strong growth in the next few years. It will grow to $5.46 billion in 2030 at a compound annual growth rate (CAGR) of 6.5%. The growth in the forecast period can be attributed to expansion of robotic-assisted surgery, development of bioabsorbable and hybrid mesh, rising awareness of minimally invasive procedures, growth in geriatrics and chronic disease patients, adoption of personalized surgical solutions. Major trends in the forecast period include minimally invasive surgical techniques adoption, advanced biocompatible materials development, personalized surgical mesh solutions, enhanced post-surgical recovery focus, rising demand for pelvic health solutions.Global Surgical Mesh Implants Market Segmentation
1) By Type: Non-Absorbable Surgical Mesh, Absorbable Surgical Mesh, Other Types 2) By Surgical Procedure Type: Laparoscopic Surgery, Open Surgery, Robotic-assisted Surgery, Endoscopic Surgery 3) By Material: Polypropylene, Polyester, Polytetrafluoroethylene, Other Materials 4) By Application: Hernia Repair, Pelvic Organ Prolapse Repair, Gastroesophageal Reflux Disease (GERD) Repair, Other Surgical Applications 5) By End-User: Hospitals, Ambulatory Surgical Centers (ASCs), Specialized Clinics Subsegments: 1) By Non-Absorbable Surgical Mesh: Polypropylene Mesh, Expanded Polytetrafluoroethylene (ePTFE) Mesh, Polyester Mesh, Composite Mesh 2) By Absorbable Surgical Mesh: Polyglycolic Acid (PGA) Mesh, Polylactic Acid (PLA) Mesh, Polyglycolide-co-lactide (PGLA) Mesh, Poly-4-hydroxybutyrate (P4HB) Mesh 3) By Other Types: Biologic Mesh, Hybrid Mesh, Coated/Barrier Mesh, 3D-printed MeshWhat Is The Driver Of The Surgical Mesh Implants Market?
The growth in minimally invasive surgeries is expected to propel the growth of the surgical mesh implants market going forward. Minimally invasive surgery involves surgical procedures performed through small incisions, often using laparoscopic or robotic-assisted techniques. Minimally invasive surgeries are rising due to their ability to provide quicker recovery, enabling patients to return to normal activities sooner while also reducing postoperative discomfort and hospital stay durations. As more minimally invasive procedures are performed, demand for mesh implants increases in applications such as hernia repair and pelvic surgeries because these implants are often used in such procedures. For instance, in January 2025, according to the Intuitive Surgical Inc., a US-based medical device company, in 2024, customers used our products in almost 2.7 million procedures, a 17% increase over 2023. Altogether, the total number of procedures performed on da Vinci platforms has now reached nearly 17 million. Therefore, growth in minimally invasive surgeries is driving the growth of the surgical mesh implants industry.Key Players In The Global Surgical Mesh Implants Market
Major companies operating in the surgical mesh implants market are Johnson And Johnson, Medtronic plc, W L Gore And Associates Inc, Cook Medical, Integra LifeSciences Corporation, Ethicon Inc, B Braun Melsungen AG, Feg Textiltechnik mbH, Cousin Biotech, Proxy Biomedical Ltd, Betatech Medical, Dipromed Srl, Novus Scientific Pte Ltd, Atrium Medical Corporation, LifeCell Corporation, Hernia Mesh S R L, THT Bio Science Inc, Gore Medical, Bard Medical, Covidien Ltd, Surgical Specialties Corporation, Aspen Surgical Products IncGlobal Surgical Mesh Implants Market Trends and Insights
Major companies operating in the surgical mesh implants market are focusing on developing advanced innovations such as fully bioabsorbable designs to enhance patient outcomes, reduce long-term complications, and meet the growing demand for non-permanent implant solutions. Fully bioabsorbable designs refer to implantable meshes made from materials that naturally degrade and are absorbed by the body over time, eliminating the need for permanent implants. For instance, in April 2025, Becton, Dickinson and Company, a US-based medication company, announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Phasix ST Umbilical Hernia Patch, which has now been launched commercially in the U.S. market. This patch is notable for being the first and only fully absorbable hernia mesh specifically developed for repairing umbilical hernias. Constructed with a bioabsorbable polymer, the patch gradually dissolves in the body, eliminating the need for permanent implants. This launch represents a significant innovation in soft tissue repair, addressing both clinical requirements and patient preferences for non-permanent mesh options.What Are Latest Mergers And Acquisitions In The Surgical Mesh Implants Market?
In February 2024, RTI Surgical Inc., a US-based contract development and manufacturing organization (CDMO), acquired Cook Biotech Inc. for an undisclosed amount. Through this acquisition, RTI Surgical Inc. seeks to establish itself as the only regenerative medicine CDMO capable of providing both allograft and xenograft biomaterials at scale, enhancing its leadership in soft tissue and broadening its market presence. Cook Biotech Inc. is a US-based regenerative medicine company that manufactures a biologic hernia repair mesh.Regional Outlook
North America was the largest region in the surgical mesh implants market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Surgical Mesh Implants Market?
The surgical mesh implants market consists of sales of plug mesh, coated mesh, lightweight mesh, and heavyweight mesh. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Surgical Mesh Implants Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.24 billion |
| Revenue Forecast In 2035 | $5.46 billion |
| Growth Rate | CAGR of 6.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Surgical Procedure Type, Material, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson And Johnson, Medtronic plc, W L Gore And Associates Inc, Cook Medical, Integra LifeSciences Corporation, Ethicon Inc, B Braun Melsungen AG, Feg Textiltechnik mbH, Cousin Biotech, Proxy Biomedical Ltd, Betatech Medical, Dipromed Srl, Novus Scientific Pte Ltd, Atrium Medical Corporation, LifeCell Corporation, Hernia Mesh S R L, THT Bio Science Inc, Gore Medical, Bard Medical, Covidien Ltd, Surgical Specialties Corporation, Aspen Surgical Products Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
