Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Report 2026

Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Report 2026
Epidemiology, Pipeline Analysis, Market Insights & Forecasts
Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Overview
• The Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test market growth in the historic period has been driven by increasing prevalence of allergic contact dermatitis, expansion of dermatology clinics • Market expansion is supported by increasing demand for personalized allergy management, expansion of outpatient diagnostic testing • Growth Driver: Impact Of Increasing Skin Allergy Prevalence On The Growth Of The Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market?
The thin-layer rapid use epicutaneous (T.R.U.E.) test is a diagnostic tool used to identify allergens that cause allergic contact dermatitis. It involves applying a set of pre-dosed allergens on the skin using a patch, which is then worn for a specified period to observe allergic reactions. The purpose of the T.R.U.E. Test is to accurately diagnose contact allergies and help in the management of allergic skin conditions. The main types of indications for the thin-layer rapid use epicutaneous (T.R.U.E.) test are allergic contact dermatitis and atopic dermatitis. Allergic contact dermatitis is a type of skin inflammation caused by an allergic reaction to substances that come into contact with the skin, leading to symptoms like redness, itching, and swelling. The formulation includes patch test kits and liquid test solutions. and it is distributed through various distribution channels, such as hospitals, clinics, home healthcare, and pharmacies.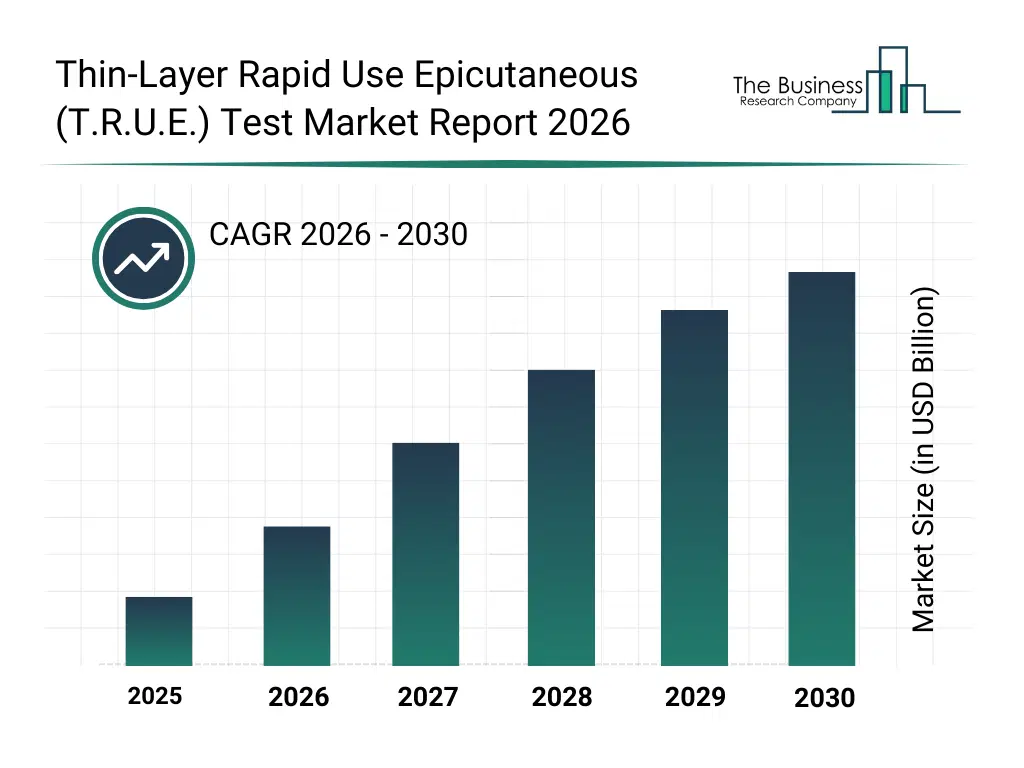
What Is The Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Size and Share 2026?
The growth in the historic period can be attributed to increasing prevalence of allergic contact dermatitis, expansion of dermatology clinics, rising occupational skin allergy cases, availability of standardized allergen panels, improved awareness of contact allergy diagnosis.What Is The Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Growth Forecast?
The growth in the forecast period can be attributed to increasing demand for personalized allergy management, expansion of outpatient diagnostic testing, rising adoption of home-based diagnostic solutions, growing investments in dermatology diagnostics, increasing regulatory emphasis on early allergy detection. Major trends in the forecast period include increasing adoption of standardized patch testing kits, rising demand for accurate allergy diagnostics, expansion of dermatology diagnostic services, growing use of pre-dosed allergen panels, improved patient compliance with rapid testing.Global Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Segmentation
1) By Indication: Allergic Contact Dermatitis, Atopic Dermatitis 2) By Formulation: Patch Test Kits, Liquid Test Solutions 3) By Distribution Channel: Hospitals, Clinics, Home Healthcare, PharmaciesWhat Are The Drivers Of The Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market?
The increasing prevalence of skin allergies is significantly driving the growth of the thin-layer rapid use epicutaneous (T.R.U.E.) test market in the future. Skin allergies are immune system reactions triggered by exposure to allergens, leading to symptoms such as redness, itching, swelling, or rashes on the skin. The prevalence of skin allergies can be attributed to factors such as environmental pollution, lifestyle changes, increased exposure to allergens, and genetic predispositions. The T.R.U.E. Test is used to accurately diagnose allergic contact dermatitis by identifying specific allergens, helping to manage the rising prevalence of skin allergies. For instance, in December 2023, according to PubMed, a US-based online biomedical database maintained by a government agency, the global prevalence of atopic dermatitis (AD) was estimated at 2.6% [95% uncertainty interval (UI) 1.9–3.5], affecting approximately 204.05 million people worldwide, including around 101.27 million adults and 102.78 million children. Therefore, the growing prevalence of skin allergies and AD is expected to drive the growth of the T.R.U.E. test market. The increase in investments for healthcare infrastructure is set to propel the growth of the thin-layer rapid use epicutaneous (T.R.U.E.) test market moving forward. Healthcare infrastructure refers to the physical and organizational structures, facilities, equipment, and resources necessary for the delivery of healthcare services, including hospitals, clinics, medical equipment, and trained personnel. Investments in healthcare infrastructure are increasing to address rising healthcare demands, improve service quality, and enhance accessibility amid growing populations and advancing medical technologies. Increasing investments in healthcare infrastructure are crucial for the TRUE test, as they enhance the availability of advanced diagnostic facilities and support the adoption of innovative technologies, ensuring accurate and accessible allergy testing. For instance, in March 2023, according to the Department of Health and Social Care, a UK-based government department, the government is dedicated to constructing 40 new hospitals by 2030. For the first time, it has been confirmed that the New Hospital Programme will receive more than $25 billion (£20 billion) in funding. This investment will support the development of hospital infrastructure across the country. Therefore, the increasing investments for healthcare infrastructure will drive the growth of the thin-layer rapid use epicutaneous (T.R.U.E.) test industry. The rise in the geriatric population is expected to drive the thin-layer rapid use epicutaneous (T.R.U.E.) test market going forward. The geriatric population refers to individuals aged 65 and older, typically considered to be in the later stages of adulthood, often requiring specialized care due to age-related health issues. The geriatric population is rising due to increased life expectancy, advancements in healthcare, and improved living standards, leading to a larger number of older adults. The TRUE Test supports the rising geriatric population by identifying allergens causing skin reactions like contact dermatitis, helping manage common allergic conditions in older adults, and improving their quality of life. For instance, in October 2025, according to the National Council on Aging (NCOA), a US-based non-profit organization, reported that global life expectancy has reached 73.5 years in 2025, an increase of 8.6 years. Therefore, the rising geriatric population will drive the thin-layer rapid use epicutaneous (T.R.U.E.) test industry growth.Key Players In The Global Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market
Major companies operating in the thin-layer rapid use epicutaneous (t.r.u.e.) test market are Mekos Laboratories ASRegional Insights
North America was the largest region in the thin-layer rapid use epicutaneous (T.R.U.E.) test market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, India, China, South Korea, Japan, UK, France, Germany, USA, Canada.What Defines the Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market?
The thin-layer rapid use epicutaneous (T.R.U.E.) test market consists of sales of diagnostic equipment, test accessories, training, and support materials. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Report 2026?
The thin-layer rapid use epicutaneous (t.r.u.e.) test market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the thin-layer rapid use epicutaneous (t.r.u.e.) test industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $ billion |
| Revenue Forecast In 2035 | $ billion |
| Growth Rate | No CAGR found from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Indication, Formulation, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Mekos Laboratories AS |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test market was valued at $ billion in 2025, increased to $ billion in 2026, and is projected to reach $ billion by 2030.
The global Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test market is expected to grow at a CAGR of nan% from 2026 to 2035 to reach $ billion by 2035.
Some Key Players in the Thin-Layer Rapid Use Epicutaneous (T.R.U.E.) Test market Include, Mekos Laboratories AS .
Major trend in this market includes: nan. For further insights on this market.
Request for SampleNorth America was the largest region in the thin-layer rapid use epicutaneous (T.R.U.E.) test market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the thin-layer rapid use epicutaneous (t.r.u.e.) test market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
