
Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Report 2026
Global Outlook – By Type (Transthyretin Stabilizers, Nonsteroidal Anti-inflammatory Drugs (NSAID), RNAi Therapy, Other Types), By Indication (Wild type ATTR Amyloidosis, Hereditary ATTR amyloidosis), By End-Users (Hospitals, Homecare, Specialty Clinics, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Overview
• Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment market size has reached to $13.29 billion in 2025 • Expected to grow to $45.09 billion in 2030 at a compound annual growth rate (CAGR) of 27.6% • Growth Driver: Impact Of Chronic Disease Prevalence On The Growth Of The Market • Market Trend: Innovative Oral Treatments Drive Growth In The Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market?
Transthyretin amyloid cardiomyopathy (ATTR-CM) treatment refers to medical therapies and interventions aimed at managing ATTR-CM, a progressive and life-threatening disease caused by the buildup of misfolded transthyretin (TTR) proteins in the heart, leading to restrictive cardiomyopathy and heart failure. The main types of transthyretin amyloid cardiomyopathy (ATTR-CM) treatment are transthyretin stabilizers, nonsteroidal anti-inflammatory drugs (NSAIDs), RNAi therapy, and other types. Transthyretin stabilizers are drugs that prevent transthyretin protein misfolding, reducing amyloid deposits and slowing disease progression in ATTR-CM. The various clinical indications include wild-type ATTR amyloidosis and hereditary ATTR amyloidosis. The treatment is used by various end-users, including hospitals, home care, specialty clinics, and others.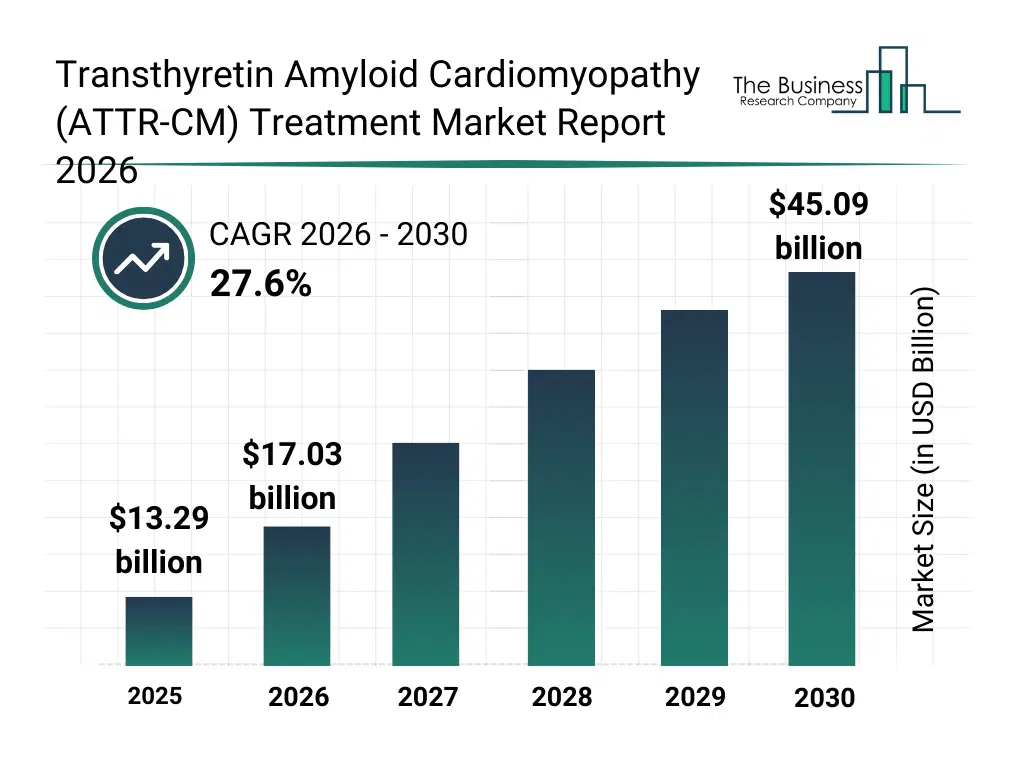
What Is The Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Size and Share 2026?
The transthyretin amyloid cardiomyopathy (attr-cm) treatment market size has grown exponentially in recent years. It will grow from $13.29 billion in 2025 to $17.03 billion in 2026 at a compound annual growth rate (CAGR) of 28.1%. The growth in the historic period can be attributed to improved disease awareness among cardiologists, increased diagnosis of rare cardiomyopathies, availability of targeted pharmacological therapies, expansion of specialty cardiac care centers, progress in genetic screening technologies.What Is The Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Growth Forecast?
The transthyretin amyloid cardiomyopathy (attr-cm) treatment market size is expected to see exponential growth in the next few years. It will grow to $45.09 billion in 2030 at a compound annual growth rate (CAGR) of 27.6%. The growth in the forecast period can be attributed to expansion of precision medicine approaches, growing development of next-generation gene-silencing therapies, rising investments in rare disease drug pipelines, increasing patient access to advanced cardiac diagnostics, stronger focus on disease-modifying treatments. Major trends in the forecast period include increasing adoption of transthyretin stabilizer therapies, rising use of rnAI-based treatment approaches, growing focus on early diagnosis and disease monitoring, expansion of personalized treatment regimens, increased investment in rare cardiac disease research.Global Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Segmentation
1) By Type: Transthyretin Stabilizers, Nonsteroidal Anti-inflammatory Drugs (NSAID), RNAi Therapy, Other Types 2) By Indication: Wild type ATTR Amyloidosis, Hereditary ATTR amyloidosis 3) By End-Users: Hospitals, Homecare, Specialty Clinics, Other End Users Subsegments: 1) By Transthyretin Stabilizers: Tafamidis, Diflunisal 2) By Nonsteroidal Anti-inflammatory Drugs (NSAIDs): Aspirin, Ibuprofen 3) By RNAi Therapy: Patisiran, Vutrisiran 4) By Other Types: Gene Silencing Therapies, Immunotherapy Approaches, Emerging Small Molecule TherapiesWhat Is The Driver Of The Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market?
The rising prevalence of chronic diseases is expected to propel the growth of the transthyretin amyloid cardiomyopathy (ATTR-CM) treatment market going forward. Chronic diseases are long-lasting conditions that progress slowly and persist for a year or more, often requiring continuous medical care. The rising prevalence of chronic diseases is attributed to factors such as aging populations, sedentary lifestyles, unhealthy diets, increasing obesity rates, and exposure to environmental pollutants. Transthyretin amyloid cardiomyopathy treatment helps by stabilizing transthyretin, reducing amyloid buildup, improving heart function, and alleviating symptoms to better manage related chronic conditions. For instance, in April 2025, according to the Centers for Disease Control and Prevention (CDC), a US-based federal agency. in 2023, approximately 194 million U.S. adults, or 76.4%, reported having at least one chronic condition. The prevalence was 59.5% among young adults, 78.4% among midlife adults, and 93.0% among older adults. Therefore, the rising prevalence of chronic diseases is driving the transthyretin amyloid cardiomyopathy (ATTR-CM) treatment industry.Key Players In The Global Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market
Major companies operating in the transthyretin amyloid cardiomyopathy (attr-cm) treatment market are Alnylam Pharmaceuticals Inc, BridgeBio Pharma Inc, Intellia Therapeutics Inc, SOM Biotech S L, Ionis Pharmaceuticals Inc, Eidos Therapeutics Inc, Prothena Corporation plc, Neurimmune Holding AG, Corino Therapeutics Inc, CRISPR Therapeutics AG, YolTech Therapeutics Co Ltd, Silence Therapeutics plc, Akcea Therapeutics IncGlobal Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Trends and Insights
Major companies operating in the transthyretin amyloid cardiomyopathy (ATTR-CM) treatment market are focusing on developing innovative treatments for ATTR-CM, such as oral prescription medication to target and stabilize transthyretin (TTR) proteins or reduce amyloid fibril buildup in the heart. Oral prescription medication refers to drugs prescribed by a healthcare provider that are taken by mouth, typically in the form of tablets, capsules, or liquid solutions. For instance, in November 2024, BridgeBio Pharma Inc., a US-based biotechnology company, received approval from the U.S. Food and Drug Administration for Attruby (acoramidis) for the treatment of adults with transthyretin amyloid cardiomyopathy (ATTR-CM). This stabilizes the TTR protein, reducing heart-related deaths and hospitalizations. Clinical trials have shown that it is well-tolerated and provides rapid benefits by targeting the root cause of the disease.What Are Latest Mergers And Acquisitions In The Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market?
In March 2024, Bayer AG, a Germany-based pharmaceutical company, acquired exclusive European marketing rights for acoramidis from BridgeBio Pharma for up to $310 million. With this acquisition, Bayer aims to leverage its established cardiovascular commercial infrastructure to bring a new oral therapy for Transthyretin Amyloid Cardiomyopathy (ATTR-CM) to the European market, thereby expanding its presence in rare cardiovascular disease therapeutics. BridgeBio is a US-based biopharmaceutical company focused on genetic and rare diseases, developing acoramidis as a highly potent, orally administered small-molecule transthyretin (TTR) stabilizer intended to treat ATTR-CM.Regional Outlook
North America was the largest region in the transthyretin amyloid cardiomyopathy (ATTR-CM) treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market?
The transthyretin amyloid cardiomyopathy (ATTR-CM) market consists of revenues earned by entities by providing services such as diagnosis and screening services, pharmaceutical and drug therapies, patient monitoring and disease management, specialized treatment centers and hospital services. The market value includes the value of related goods sold by the service provider or included within the service offering. The transthyretin amyloid cardiomyopathy (ATTR-CM) market also includes sales of tafamidis, patisiran, vutrisiran, and acoramidis. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Report 2026?
The transthyretin amyloid cardiomyopathy (attr-cm) treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the transthyretin amyloid cardiomyopathy (attr-cm) treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Transthyretin Amyloid Cardiomyopathy (ATTR-CM) Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $17.03 billion |
| Revenue Forecast In 2035 | $45.09 billion |
| Growth Rate | CAGR of 28.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Indication, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Alnylam Pharmaceuticals Inc, BridgeBio Pharma Inc, Intellia Therapeutics Inc, SOM Biotech S L, Ionis Pharmaceuticals Inc, Eidos Therapeutics Inc, Prothena Corporation plc, Neurimmune Holding AG, Corino Therapeutics Inc, CRISPR Therapeutics AG, YolTech Therapeutics Co Ltd, Silence Therapeutics plc, Akcea Therapeutics Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
