
Urethral Stricture Treatment Devices Market Report 2026
Global Outlook – By Product Type (Urethral Dilators, Urethral Stents, Urethral Balloon Dilation Catheters, Urinary Catheters), By Stricture Location (Anterior, Posterior), By End-User (Hospitals, Specialty Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Urethral Stricture Treatment Devices Market Overview
• Urethral Stricture Treatment Devices market size has reached to $1.47 billion in 2025 • Expected to grow to $2.14 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Rising BPH Cases Propel Market Growth • Market Trend: Collaboration With Public Health Systems Revolutionizes Market • North America was the largest region in 2025.What Is Covered Under Urethral Stricture Treatment Devices Market?
Urethral stricture treatment devices are medical instruments designed to treat urethral stricture, a condition where the urethra becomes narrowed, often leading to urinary retention and difficulty in urination. These devices help in widening or bypassing the narrowed area of the urethra to restore normal urine flow, with applications in both surgical and non-surgical treatment options. The main types of products in urethral stricture treatment devices include urethral dilators, urethral stents, urethral balloon dilation catheters, and urinary catheters. Urethral dilators are tools used to widen or stretch a narrowed urethra, making it easier for urine to pass, and commonly used during procedures to treat urethral strictures by gradually increasing the diameter of the urethra. The stricture locations are anterior, and posterior for end users such as hospitals, specialty clinics, and others.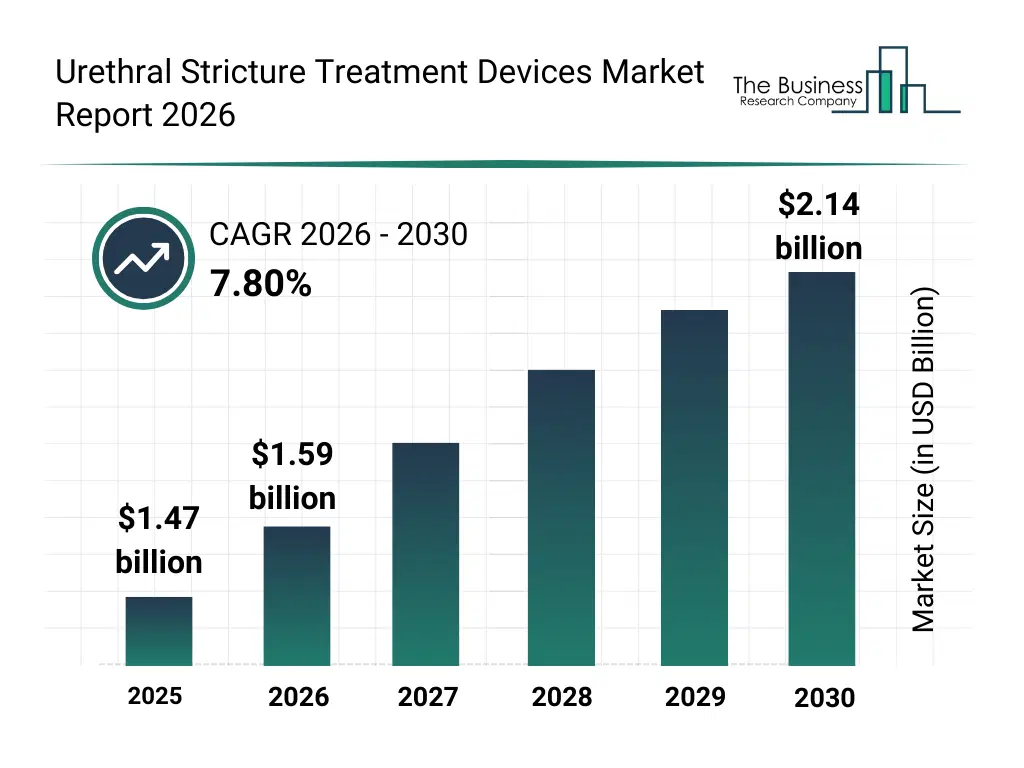
What Is The Urethral Stricture Treatment Devices Market Size and Share 2026?
The urethral stricture treatment devices market size has grown strongly in recent years. It will grow from $1.47 billion in 2025 to $1.59 billion in 2026 at a compound annual growth rate (CAGR) of 8.0%. The growth in the historic period can be attributed to rise in urological disorders, aging male population, catheterization procedures growth, hospital urology department expansion, surgical stricture treatments.What Is The Urethral Stricture Treatment Devices Market Growth Forecast?
The urethral stricture treatment devices market size is expected to see strong growth in the next few years. It will grow to $2.14 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to growth in minimally invasive urology, demand for long-term stent solutions, advanced biomaterial adoption, outpatient procedure expansion, personalized urology devices. Major trends in the forecast period include minimally invasive stricture management, balloon dilation technologies, advanced urethral stents, improved catheter materials, outpatient urology procedures.Global Urethral Stricture Treatment Devices Market Segmentation
1) By Product Type: Urethral Dilators, Urethral Stents, Urethral Balloon Dilation Catheters, Urinary Catheters 2) By Stricture Location: Anterior, Posterior 3) By End-User: Hospitals, Specialty Clinics, Other End-Users Subsegments: 1) By Urethral Dilators: Semi-Rigid Urethral Dilators, Flexible Urethral Dilators, Balloon Urethral Dilators, Graded Urethral Dilators 2) By Urethral Stents: Self-Expanding Urethral Stents, Balloon-Expandable Urethral Stents, Metallic Urethral Stents, Silicone Urethral Stents 3) By Urethral Balloon Dilation Catheters: Single-Balloon Dilation Catheters, Double-Balloon Dilation Catheters, Coaxial Balloon Dilation Catheters, Hydrophilic-Coated Balloon Dilation Catheters 4) By Urinary Catheters: Foley Urinary Catheters, Intermittent Urinary Catheters, External Urinary Catheters, Supra-Pubic Urinary CathetersWhat Is The Driver Of The Urethral Stricture Treatment Devices Market?
The rising prevalence of benign prostatic hyperplasia (BPH) and neurogenic bladder is projected to propel the urethral stricture treatment devices market going forward. BPH refers to a common condition in older men characterized by an enlarged prostate gland that often leads to urinary obstruction and increases the risk of urethral strictures. The rising prevalence of benign prostatic hyperplasia (BPH) is primarily driven by aging, with a higher incidence in men over 50, and factors such as lifestyle, hormonal changes, and chronic conditions contribute to its progression. Urethral stricture treatment devices are commonly used in urological conditions such as benign prostatic hyperplasia (BPH), where post-surgical complications or prolonged catheterization may lead to urethral narrowing, requiring interventions such as dilators, stents, or urethrotomy tools to restore normal urinary flow. For instance, in September 2023, according to a report published by the Royal Pharmaceutical Society, a UK-based professional body, approximately 3 million people in the UK are affected by BPH, with its incidence being around 50% in males aged 60–70 years and rising to 90% in males aged over 80 years. Therefore, the rising prevalence of BPH and neurogenic bladder will drive the urethral stricture treatment device industry.Key Players In The Global Urethral Stricture Treatment Devices Market
Major companies operating in the urethral stricture treatment devices market are Cardinal Health, Medtronic plc, Siemens Healthineers AG, Medline Industries LP., Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, B. Braun SE, Olympus Corporation, Coloplast Ltd, Teleflex Incorporated, Karl Storz SE & Co. KG, Cook Medical, ConvaTec Inc., Ambu A/S, Richard Wolf GmbH, Laborie Medical Technologies, Bactiguard AB, UROMED Kurt Drews KG, Pnn Medical A/S, Hydrumedical SA, Allium Ltd., Med pro Medical B.V., Urotronic Inc., Rocamed SAM, Zhejiang ChuangXiang Medical Technology Co. LtdGlobal Urethral Stricture Treatment Devices Market Trends and Insights
Major companies operating in the urethral stricture treatment devices market are adopting a strategic partnership approach to enhance technology integration and expand market reach. A strategic partnership refers to a collaborative relationship between two or more organizations where they combine their resources, expertise, and efforts to achieve common goals or objectives. For instance, in April 2024, Laborie Medical Technologies Corp., a US-based diagnostic and therapeutic medical technology company, partnered with the NHS Specialized Services Device Programme (SSDP) to include its Optilume Urethral Drug-Coated Balloon (DCB) in the program. This collaboration ensures reimbursement for NHS hospitals in England, facilitating wider patient access to the innovative device. Optilume integrates balloon dilation with anti-proliferative drug delivery to reduce the recurrence of urethral strictures. This minimally invasive technology represents a transformative alternative to traditional surgery or repeated treatments, addressing a critical need within the urology community.What Are Latest Mergers And Acquisitions In The Urethral Stricture Treatment Devices Market?
In October 2023, Laborie Medical Technologies Corp., a US-based diagnostic and therapeutic medical technology company, acquired Urotronic Inc. for an undisclosed amount. This acquisition enables Laborie to integrate Urotronic’s Optilume drug-coated balloon technology into its portfolio, enhancing its interventional urology solutions. The technology addresses critical conditions such as urethral strictures and benign prostatic hyperplasia (BPH). Urotronic Inc. is a US-based medical device company specializing in urethral stricture treatment devices and technologies for urological treatments.Regional Outlook
North America was the largest region in the urethral stricture treatment devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Urethral Stricture Treatment Devices Market?
The urethral stricture treatment devices market consists of sales of laser urethrotomy devices, biodegradable stents, lubricants, urethral calibration sets, and bougies. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Urethral Stricture Treatment Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.59 billion |
| Revenue Forecast In 2035 | $2.14 billion |
| Growth Rate | CAGR of 8.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Stricture Location, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health, Medtronic plc, Siemens Healthineers AG, Medline Industries LP., Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, B. Braun SE, Olympus Corporation, Coloplast Ltd, Teleflex Incorporated, Karl Storz SE & Co. KG, Cook Medical, ConvaTec Inc., Ambu A/S, Richard Wolf GmbH, Laborie Medical Technologies, Bactiguard AB, UROMED Kurt Drews KG, Pnn Medical A/S, Hydrumedical SA, Allium Ltd., Med pro Medical B.V., Urotronic Inc., Rocamed SAM, Zhejiang ChuangXiang Medical Technology Co. Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
