
Urinary Tract Obstruction Treatment Devices Market Report 2026
Global Outlook – By Type (Urinary Catheter, Ureteroscope, Shock Wave Lithotripters, Laser Therapy Systems, Other Types), By Application (Cancers, Stones, Benign Prostatic Hyperplasia (BPH), Hydronephrosis), By End-User (Hospitals, Ambulatory Surgical Centers, Surgical Centers, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Urinary Tract Obstruction Treatment Devices Market Overview
• Urinary Tract Obstruction Treatment Devices market size has reached to $1.4 billion in 2025 • Expected to grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 6% • Growth Driver: Rising Prevalence Of Kidney Disease To Drive Growth In The Market • Market Trend: Next-Generation Prostatic Stents Transforming Outpatient Treatment Approaches • North America was the largest region in 2025.What Is Covered Under Urinary Tract Obstruction Treatment Devices Market?
Urinary tract obstruction (UTO) treatment devices are medical tools used to manage blockages that disrupt normal urine flow from the kidneys to the bladder. They help clear obstructions, drain urine, or support healing, preventing further damage to the kidneys, bladder, and urinary tract by relieving pressure. The main types of urinary tract obstruction treatment devices are urinary catheters, ureteroscopes, shock wave lithotripters, laser therapy systems, and others. Urinary catheters are flexible tubes inserted into the bladder to drain urine, aiding in the management of urinary obstructions and preventing complications such as bladder overdistension. It is applicable for various applications, such as cancers, stones, benign prostatic hyperplasia (BPH), and hydronephrosis, and is used by several end users, including hospitals, ambulatory surgical centers, surgical centers, and others.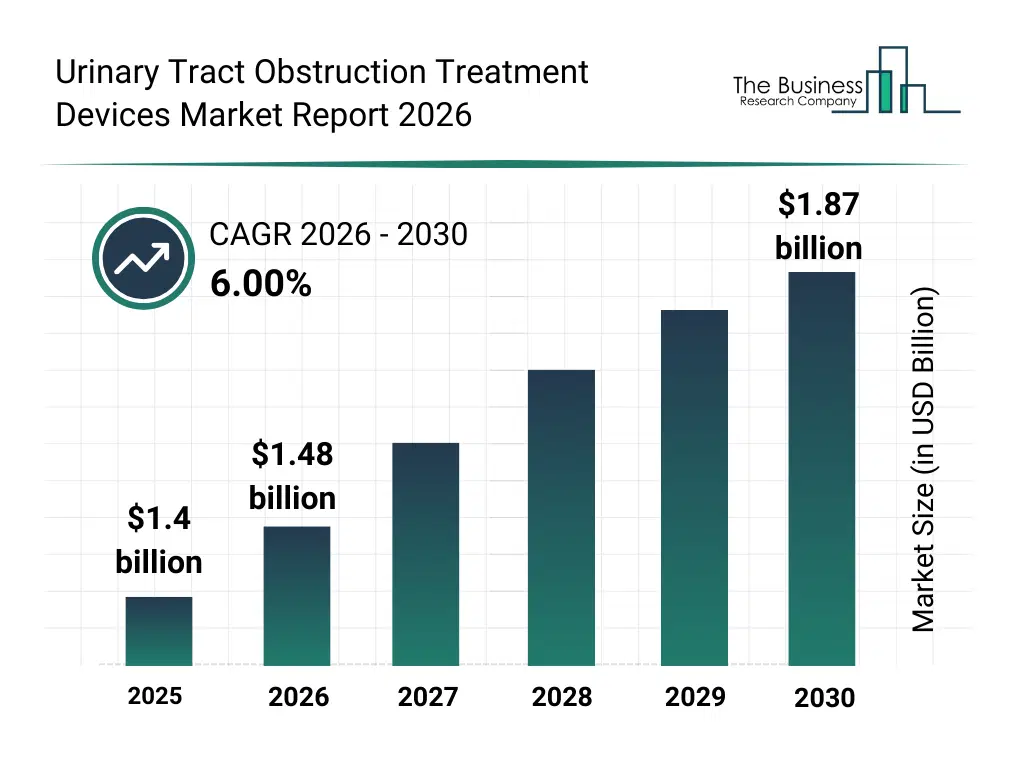
What Is The Urinary Tract Obstruction Treatment Devices Market Size and Share 2026?
The urinary tract obstruction treatment devices market size has grown strongly in recent years. It will grow from $1.4 billion in 2025 to $1.48 billion in 2026 at a compound annual growth rate (CAGR) of 6.1%. The growth in the historic period can be attributed to rise in kidney stone prevalence, aging population growth, BPH incidence increase, hospital urology expansion, catheterization procedure growth.What Is The Urinary Tract Obstruction Treatment Devices Market Growth Forecast?
The urinary tract obstruction treatment devices market size is expected to see strong growth in the next few years. It will grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 6.0%. The growth in the forecast period can be attributed to laser-based urology adoption, outpatient obstruction treatments, advanced stent biomaterials, robotic-assisted urology procedures, reduced hospital stay initiatives. Major trends in the forecast period include minimally invasive obstruction management, advanced laser lithotripsy systems, improved ureteral stent designs, endoscopic stone removal techniques, faster post-procedure recovery solutions.Global Urinary Tract Obstruction Treatment Devices Market Segmentation
1) By Type: Urinary Catheter, Ureteroscope, Shock Wave Lithotripters, Laser Therapy Systems, Other Types 2) By Application: Cancers, Stones, Benign Prostatic Hyperplasia (BPH), Hydronephrosis 3) By End-User: Hospitals, Ambulatory Surgical Centers, Surgical Centers, Other End-Users Subsegments: 1) By Urinary Catheter: Foley Urinary Catheters, Intermittent Urinary Catheters, External Urinary Catheters, Supra-Pubic Urinary Catheters, Hydrophilic-Coated Urinary Catheters 2) By Ureteroscope: Rigid Ureteroscopes, Flexible Ureteroscopes, Semi-Rigid Ureteroscopes, Digital Ureteroscopes 3) By Shock Wave Lithotripters: Extracorporeal Shock Wave Lithotripters (ESWL), Pneumatic Shock Wave Lithotripters, Electrohydraulic Shock Wave Lithotripters, Laser-Assisted Shock Wave Lithotripters 4) By Laser Therapy Systems: Holmium Laser Therapy Systems, Thulium Laser Therapy Systems, Diode Laser Therapy Systems, Nd:YAG Laser Therapy Systems 5) By Other Types: Stents For Urinary Tract Obstruction, Percutaneous Nephrostomy Devices, Ureteral Balloon Dilation Catheters, Ureteral Access Sheaths, Urological Drainage DevicesWhat Is The Driver Of The Urinary Tract Obstruction Treatment Devices Market?
The increasing prevalence of kidney disease is expected to propel the growth of the urinary tract obstruction treatment devices market. Kidney disease involves the dysfunction or damage to the kidneys, affecting their ability to filter waste and excess fluids from the bloodstream. The prevalence of kidney disease is increasing due to factors such as aging populations, rising rates of diabetes and hypertension, and unhealthy lifestyles. Kidney disease requires medical intervention to reduce pressure on the kidneys and preserve renal function. As the condition worsens, urinary tract obstructions become more common, increasing the need for devices to manage and prevent further kidney damage. For instance, in June 2023, according to the report published by Kidney Research UK, a UK-based non-profit organization, currently, approximately 7.2 million people in the UK are living with chronic kidney disease, representing over 10% of the population. Of these, 3.25 million are in the advanced stages of the disease, while an additional 3.9 million are in the early stages. By 2033, the number of individuals affected by chronic kidney disease is projected to rise to 7.61 million. Therefore, increasing prevalence of kidney disease is expected to drive the growth of the urinary tract obstruction treatment devices industry.Key Players In The Global Urinary Tract Obstruction Treatment Devices Market
Major companies operating in the urinary tract obstruction treatment devices market are Medtronic Plc, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Olympus Corporation, Coloplast Corporation, Smiths Group plc, Bio-Rad Laboratories, Teleflex Incorporated, Paul Hartmann AG, Karl Storz SE & Co. KG, Cook Medical, Hollister Incorporated, Amsino International Inc., Richard Wolf GmbH, Braun Melsungen AG, Angiplast Pvt. Ltd., Mais India Medical Devices Pvt. Ltd., Convatec Inc., VYGON (UK) LTD., United Endoscopy, Advin Health Care, Manish Medi Innovation, MicroPort Urocare (Jiaxing) Co. Ltd., Nikotech Pvt Ltd.Global Urinary Tract Obstruction Treatment Devices Market Trends and Insights
Major companies operating in the urinary tract obstruction treatment devices market are focusing on minimally invasive, retrievable prostatic implants to provide reversible, outpatient solutions that relieve obstruction while preserving tissue and function. A retrievable prostatic stent is a small, implantable nitinol device placed cystoscopically in the prostatic urethra to restore urine flow without surgery and can be removed or replaced if needed. For instance, in April 2025, Rivermark Medical, a US-based medical device company, launched the pivotal RAPID-III trial for the FloStent system, a minimally invasive nitinol prostatic implant designed to relieve lower urinary tract symptoms from benign prostatic hyperplasia (BPH) as a first-line, reversible outpatient treatment. The FloStent system aims to offer an alternative to medication and more invasive surgical procedures by gently expanding the prostatic urethra to improve urinary flow. The study is expected to generate critical clinical evidence on safety, durability, and patient quality-of-life improvements, supporting broader adoption of this implant-based approach for managing BPH.What Are Latest Mergers And Acquisitions In The Urinary Tract Obstruction Treatment Devices Market?
In October 2023, Laborie Medical Technologies Corp., a US-based diagnostic and therapeutic medical technology company, acquired Urotronic, Inc. for up to US$ 600 million. With this acquisition, Laborie Medical Technologies aims to strengthen its position in the urinary tract obstruction treatment devices market by expanding its interventional urology portfolio and accelerating the global adoption of Optilume drug-coated balloon technology as a minimally invasive solution for obstructive lower urinary tract conditions such as urethral strictures and benign prostatic hyperplasia (BPH). Urotronic, Inc. is a US-based medical device company that developed the Optilume drug-coated balloon platform for treating urethral strictures and benign prostatic hyperplasia.Regional Outlook
North America was the largest region in the urinary tract obstruction treatment devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Urinary Tract Obstruction Treatment Devices Market?
The urinary tract obstruction (UTO) treatment devices market consists of sales of ureteral stents, nephrostomy tubes, suprapubic catheters, and percutaneous nephrostomy kits. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Urinary Tract Obstruction Treatment Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.48 billion |
| Revenue Forecast In 2035 | $1.87 billion |
| Growth Rate | CAGR of 6.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic Plc, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Olympus Corporation, Coloplast Corporation, Smiths Group plc, Bio-Rad Laboratories, Teleflex Incorporated, Paul Hartmann AG, Karl Storz SE & Co. KG, Cook Medical, Hollister Incorporated, Amsino International Inc., Richard Wolf GmbH, Braun Melsungen AG, Angiplast Pvt. Ltd., Mais India Medical Devices Pvt. Ltd., Convatec Inc., VYGON (UK) LTD., United Endoscopy, Advin Health Care, Manish Medi Innovation, MicroPort Urocare (Jiaxing) Co. Ltd., Nikotech Pvt Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
