
Uterine Fibroid Embolization Device Market Report 2026
Global Outlook – By Technology (Surgical Techniques, Laparoscopic Techniques, Ablation Techniques, Embolization Techniques), By Mode Of Treatment (Invasive Treatment, Minimally Invasive Treatment), By End User (Hospitals, Clinics, Ambulatory Surgical Center) – Market Size, Trends, Strategies, and Forecast to 2035
Uterine Fibroid Embolization Device Market Overview
• Uterine Fibroid Embolization Device market size has reached to $5.93 billion in 2025 • Expected to grow to $7.33 billion in 2030 at a compound annual growth rate (CAGR) of 4.3% • Growth Driver: Rising Demand for Minimally Invasive Surgeries Fuels Growth of Uterine Fibroid Embolization Device • Market Trend: Innovative Products Enhancing Minimally Invasive Treatment Of Uterine Fibroids • North America was the largest region in 2025.What Is Covered Under Uterine Fibroid Embolization Device Market?
A uterine fibroid embolization device is a medical device that is used to treat noncancerous tumors using a minimally invasive process. Uterine fibroids are noncancerous growths of the uterus. This device restricts blood supply to uterine fibroids, causing them to shrink and eventually disappear. The main types of technologies in uterine fibroid embolization devices are surgical techniques, laparoscopic techniques, ablation techniques and embolization techniques. Surgical techniques refer to the specific methods and procedures used by medical professionals. It is used in various modes of treatment, such as invasive treatment, minimally invasive treatment and non-invasive treatment and it includes a variety of end users, such as hospitals, clinics, and ambulatory surgical centers.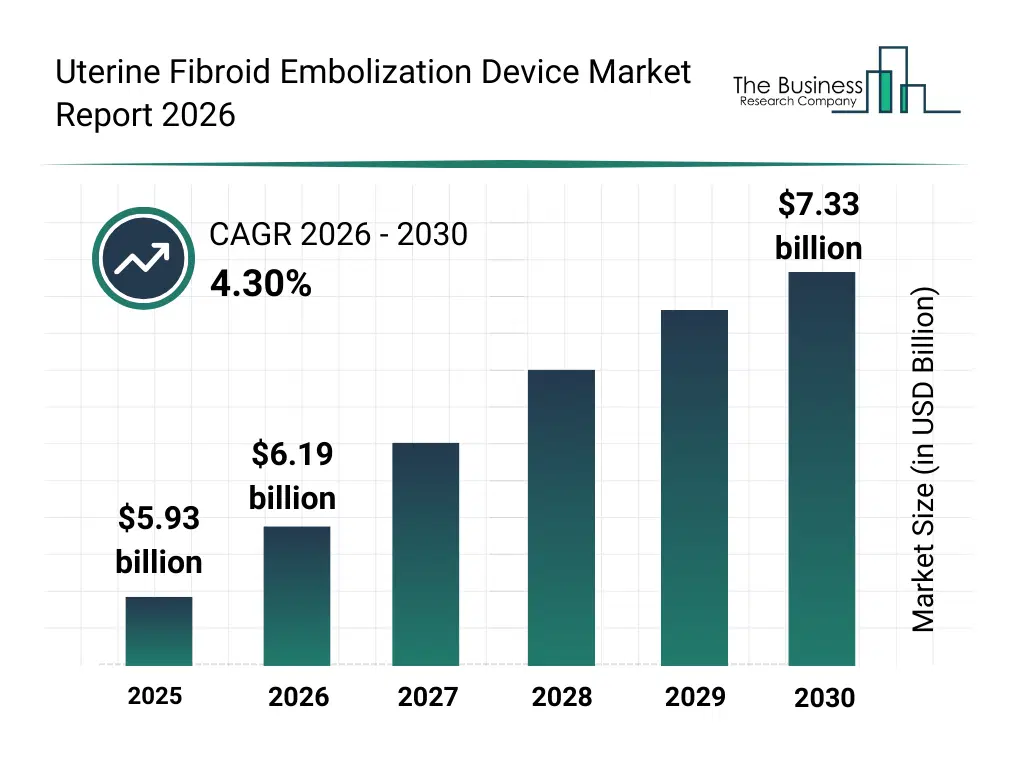
What Is The Uterine Fibroid Embolization Device Market Size and Share 2026?
The uterine fibroid embolization device market size has grown steadily in recent years. It will grow from $5.93 billion in 2025 to $6.19 billion in 2026 at a compound annual growth rate (CAGR) of 4.4%. The growth in the historic period can be attributed to rising prevalence of uterine fibroids among women of reproductive age, growing number of hospital and clinic-based gynecological procedures, increasing adoption of uterine artery embolization in interventional radiology, greater awareness of alternatives to open surgical myomectomy, expansion of minimally invasive treatment workflows in tertiary care centers.What Is The Uterine Fibroid Embolization Device Market Growth Forecast?
The uterine fibroid embolization device market size is expected to see steady growth in the next few years. It will grow to $7.33 billion in 2030 at a compound annual growth rate (CAGR) of 4.3%. The growth in the forecast period can be attributed to rising demand for uterus-sparing fibroid treatments in younger patient populations, growing clinical shift toward minimally invasive and non-invasive treatment modes, increasing procedural penetration in ambulatory surgical centers, expanding use of ablation and embolization devices for fibroid size reduction, rising investments in advanced delivery catheters and embolic systems to improve treatment precision. Major trends in the forecast period include rising adoption of minimally invasive embolization techniques for uterine fibroid treatment, increasing clinical preference for organ-preserving alternatives to hysterectomy, growing utilization of image-guided interventional devices for targeted fibroid therapy, expanding use of embolization procedures in outpatient and ambulatory surgical settings, increasing demand for precision delivery systems to improve procedural safety and outcomes.Global Uterine Fibroid Embolization Device Market Segmentation
1) By Technology: Surgical Techniques, Laparoscopic Techniques, Ablation Techniques, Embolization Techniques 2) By Mode Of Treatment: Invasive Treatment, Minimally Invasive Treatment 3) By End User: Hospitals, Clinics, Ambulatory Surgical Center Subsegments: 1) By Surgical Techniques: Open Surgery, Myomectomy 2) By Laparoscopic Techniques: Laparoscopic Myomectomy, Laparoscopic Hysterectomy 3) By Ablation Techniques: Radiofrequency Ablation, Microwave Ablation, Cryoablation 4) By Embolization Techniques: Transcatheter Arterial Embolization (TAE), Uterine Artery Embolization (UAE)What Is The Driver Of The Uterine Fibroid Embolization Device Market?
The growing demand for minimally invasive surgeries is expected to propel the growth of the uterine fibroid embolization device market going forward. Minimally invasive surgeries are surgical approaches that use small incisions or natural body openings to access and treat internal structures, organs, or tissues while minimizing trauma to the patient. The demand for minimally invasive procedures is increasing as more patients and healthcare providers prefer treatments that reduce recovery time, lower complication risks, and improve overall comfort. Uterine fibroid embolization devices support these procedures by enabling precise and effective treatment of fibroids, improving patient outcomes and procedural efficiency. For instance, in January 2025, according to AOS Open, a US-based medical journal, the rate of minimally invasive surgeries in hospitals with robotic-assisted systems has risen to 65.8% in 2025, up from 60.5% in 2022. Therefore, the growing demand for minimally invasive surgeries drives the growth of the uterine fibroid embolization device industry.Key Players In The Global Uterine Fibroid Embolization Device Market
Major companies operating in the uterine fibroid embolization device market are Johnson and Johnson Pvt Ltd., Medtronic plc., Boston Scientific Corporation, Olympus Corporation, Terumo Corporation, Ethicon Inc., Hologic Inc., CooperSurgical Inc., Cook Medical Inc., Karl Storz GmbH, Merit Medical Systems, CONMED Corporation, Lumenis Ltd., Richard Wolf GmbH, BTG International Ltd., Healthium MedTech Pvt. Ltd., Hironic Co.Ltd, Minerva Surgical Inc., Gynesonics Inc., Advanced Surgical Concepts PLLCGlobal Uterine Fibroid Embolization Device Market Trends and Insights
Major companies operating in the uterine fibroid embolization device market are developing innovative products such as Minimally invasive fibroid management software to enhance precision and efficiency in uterine fibroid treatment. Minimally invasive fibroid management software refers to an advanced digital platform designed to assist clinicians in the planning, guidance, and execution of minimally invasive procedures for the treatment of uterine fibroids, enhancing procedural precision, safety, and overall patient outcomes. For instance, in September 2024, Gynesonics, a US–based medical device company specializing in minimally invasive gynecologic treatments, launched SMART OS 2.4 software for the Sonata System to enhance the management of uterine fibroids. SMART OS 2.4 improves the proprietary SMART Guide by reducing the minimum ablation zone size from 2.0 × 1.3 cm to 1.6 × 1.2 cm, decreasing ablation volume by 38%, and optimizing the Thermal Safety Border for smaller fibroids. These enhancements allow for more precise, controlled treatments while preserving healthy uterine tissue, improving both procedural efficiency and patient outcomes.What Are Latest Mergers And Acquisitions In The Uterine Fibroid Embolization Device Market?
In October 2024, Hologic Inc., a US-based medical technology company, acquired Gynesonics for $350 million. With this acquisition, Hologic aims to broaden its portfolio of minimally invasive treatment options for women's health, specifically targeting the management of uterine fibroids and heavy menstrual bleeding. Gynesonics Inc. is a US-based medical technology company involved in the development of minimally invasive solutions for the treatment of symptomatic uterine fibroids.Regional Outlook
North America was the largest region in the uterine fibroid embolization device market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Uterine Fibroid Embolization Device Market?
The uterine fibroid embolization device market consists of revenues earned by entities by provide services such as minimally invasive procedures, non-invasive procedures, minimally invasive procedures, radiofrequency ablation, medication and hormonal therapy, and embolization procedures. The market value includes the value of related goods sold by the service provider or included within the service offering. The uterine fibroid embolization device market also includes sales of medications, hysteroscopic devices, laparoscopic devices, radiofrequency ablation devices, endometrial ablation devices, hysterectomy devices, and robotic surgical systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Uterine Fibroid Embolization Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.19 billion |
| Revenue Forecast In 2035 | $7.33 billion |
| Growth Rate | CAGR of 4.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Mode Of Treatment, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson and Johnson Pvt Ltd., Medtronic plc., Boston Scientific Corporation, Olympus Corporation, Terumo Corporation, Ethicon Inc., Hologic Inc., CooperSurgical Inc., Cook Medical Inc., Karl Storz GmbH, Merit Medical Systems, CONMED Corporation, Lumenis Ltd., Richard Wolf GmbH, BTG International Ltd., Healthium MedTech Pvt. Ltd., Hironic Co.Ltd, Minerva Surgical Inc., Gynesonics Inc., Advanced Surgical Concepts PLLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
