
Ventricular Assist Device Market Report 2026
Global Outlook – By Product (Left Ventricular Assist Devices (LVADs), Right Ventricular Assist Devices (RVADs), Biventricular Assist Devices (BIVADs)), By Flow Mechanism (Pulsatile Flow, Continuous Flow), By Design (Transcutaneous Ventricular Assist Devices, Implantable Ventricular Assist Devices), By Application (Bridge-To-Transplant (BTT) Therapy, Destination Therapy, Bridge-To-Recovery (BTR) Therapy, Bridge-To-Candidacy (BTC) Therapy), By End Users (Hospital, Ambulatory Surgical Centres, Cardiology Centres) – Market Size, Trends, Strategies, and Forecast to 2035
Ventricular Assist Device Market Overview
• Ventricular Assist Device market size has reached to $2.37 billion in 2025 • Expected to grow to $4.34 billion in 2030 at a compound annual growth rate (CAGR) of 12.8% • Growth Driver: Increasing Prevalence Of Cardiovascular Disorders Driving Market Growth Due To Higher Demand For Mechanical Circulatory Support • Market Trend: Embedding Laser?Welding Technology to Miniaturize and Enhance Artificial Hearts • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Ventricular Assist Device Market?
A ventricular assist device refers to an implantable mechanical circulatory device used to treat heart failure by pumping blood from the heart to the rest of the body, improving circulation to vital organs. A wide range of ventricular assist devices are approved by the US FDA to provide long-term or permanent support to patients with end-stage heart failure. The main types of ventricular assist devices include left ventricular assist devices (LVADs), right ventricular assist devices (RVADs), and biventricular assist devices (BIVADs). The left ventricular assist devices refer to a battery-operated mechanical pump that helps pump blood from the left ventricle to the vital organs of the body. They are differentiated based on design into transcutaneous ventricular assist devices and implantable ventricular assist devices, and they operate on different flows which include pulsatile flow and continuous flow. These devices are designed into transcutaneous ventricular assist devices, and implantable ventricular assist devices and used in various applications such as bridge-to-transplant (BTT) therapy, destination therapy, bridge-to-recovery (BTR) therapy, and bridge-to-candidacy (BTC) therapy. The various end-users are hospitals, ambulatory surgical centers, and cardiology centers.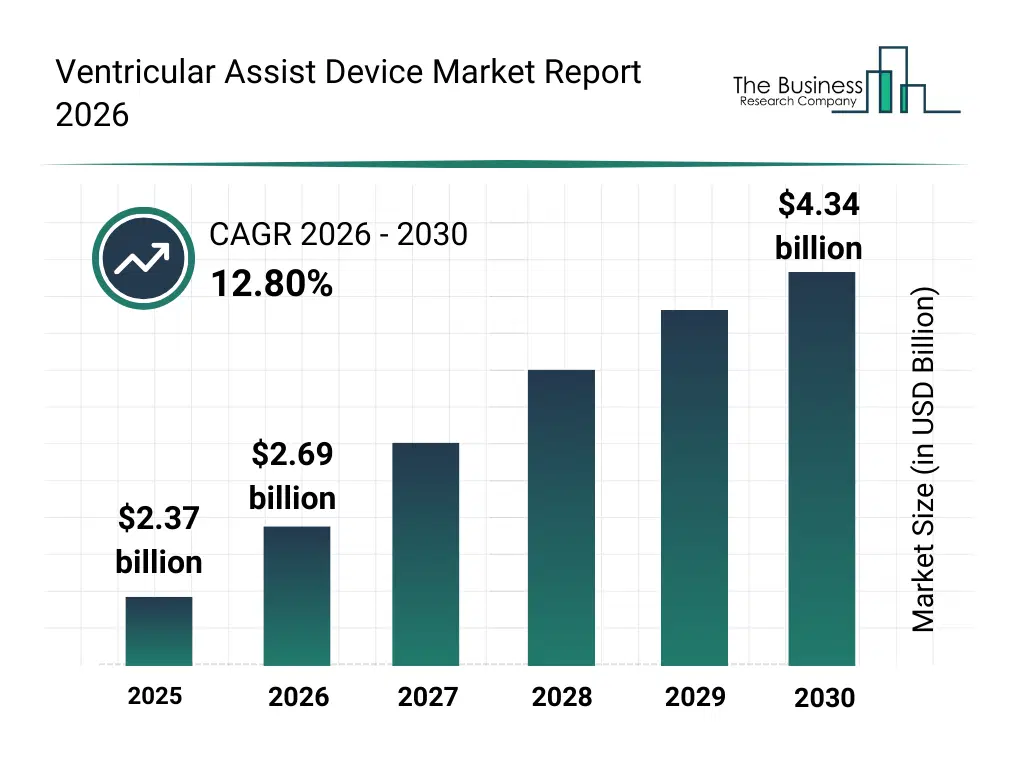
What Is The Ventricular Assist Device Market Size and Share 2026?
The ventricular assist device market size has grown rapidly in recent years. It will grow from $2.37 billion in 2025 to $2.69 billion in 2026 at a compound annual growth rate (CAGR) of 13.3%. The growth in the historic period can be attributed to increasing prevalence of end-stage heart failure, limited availability of donor hearts, advancements in mechanical circulatory support technologies, rising clinical acceptance of LVAD therapy, improved surgical outcomes.What Is The Ventricular Assist Device Market Growth Forecast?
The ventricular assist device market size is expected to see rapid growth in the next few years. It will grow to $4.34 billion in 2030 at a compound annual growth rate (CAGR) of 12.8%. The growth in the forecast period can be attributed to increasing aging population with cardiac disorders, rising adoption of long-term mechanical support, technological advancements in device durability, expansion of home-based patient monitoring, growing investments in advanced cardiac care infrastructure. Major trends in the forecast period include advancements in miniaturized implantable pump designs, growing adoption of continuous-flow devices, integration of remote patient monitoring capabilities, improved biocompatible materials usage, expansion of destination therapy applications.Global Ventricular Assist Device Market Segmentation
1) By Product: Left Ventricular Assist Devices (LVADs), Right Ventricular Assist Devices (RVADs), Biventricular Assist Devices (BIVADs) 2) By Flow Mechanism: Pulsatile Flow, Continuous Flow 3) By Design: Transcutaneous Ventricular Assist Devices, Implantable Ventricular Assist Devices 4) By Application: Bridge-To-Transplant (BTT) Therapy, Destination Therapy, Bridge-To-Recovery (BTR) Therapy, Bridge-To-Candidacy (BTC) Therapy 5) By End Users: Hospital, Ambulatory Surgical Centres, Cardiology Centres Subsegments: 1) By Left Ventricular Assist Devices (LVADs): Implantable LVADs, Temporary LVADs 2) By Right Ventricular Assist Devices (RVADs): Implantable RVADs, Temporary RVADs 3) By Biventricular Assist Devices (BIVADs): Full Support BIVADs, Hybrid BIVADsWhat Are The Drivers Of The Ventricular Assist Device Market?
The increase in the prevalence of cardiovascular disorders is driving the growth of the ventricular assist device market going forward. Cardiovascular disorders are heart diseases caused by high blood pressure, smoking, high cholesterol, inactivity, and other factors. The increase in the prevalence of cardiovascular disorders is also due to noncontagious diseases such as diabetes, chronic respiratory illness, and heart attacks due to hypertension. The significant increase in cardiovascular disorders is expected to boost demand for ventricular assist devices as they act as a vital support system for severely ill cardiac patients. For instance, in October 2024, according to the Centers for Disease Control and Prevention, a US-based public health institute, in 2023, about 1 out of every 6 deaths from cardiovascular diseases was among adults younger than 65 years old, and a total of 919,032 people died from cardiovascular disease, representing 1 in every 3 deaths. Therefore, the increase in the prevalence of cardiovascular disorders is expected to boost demand for ventricular assist devices industry. The rise in awareness regarding heart failure treatment is expected to drive the growth of the ventricular assist device market. Heart failure is a disorder that causes the heart to weaken or harden and pump blood less effectively. Heart failure is the heart's inability to provide enough blood to the body, and coronary artery disease, heart attack, high blood pressure, and damage to the heart muscle are some of the causes of heart failure. The rise in awareness regarding heart failure treatments is expected to boost the utilization of ventricular assist devices to treat heart failure. For example, in September 2025, according to the British Heart Foundation, a UK-based nonprofit organization, by 2030, the number of people living with cardiovascular disease in the UK could rise by around 1 million, and by 2040, the total could increase by about 2 million compared to current levels. Therefore, the rise in awareness regarding heart failure treatment is expected to propel the growth of the ventricular assist device industry.Key Players In The Global Ventricular Assist Device Market
Major companies operating in the ventricular assist device market are Abiomed, Abbott Laboratories, Medtronic, LivaNova PLC, Terumo Corporation, Berlin Heart GmbH, Cirtec, Jarvik Heart Inc., SynCardia Systems LLC, Calon Cardio-Technology Ltd, Huadong Medicine Co. Ltd, Shenzhen Hexin Zondan Medical Equipment Co. Ltd, Sun Medical Technology Research Corp., CorWave SA, CH Biomedical (USA) Inc, Evaheart Inc, BiVACOR Inc, ReliantHeart Inc, Cardiac Assist Inc, Heart Ware International, Braile Biomedica LtdGlobal Ventricular Assist Device Market Trends and Insights
Major companies operating in the ventricular assist device (VAD) market are focusing on technological advancements, such as laser‑welding technology, to reduce the size and weight of implantable devices, improve biocompatibility, and enhance structural integrity. By using precise laser welding, manufacturers can create lighter, more compact pumps without compromising durability, ultimately making the devices more suitable for a wider range of patients, and improving long-term outcomes. For instance, in March 2024, Evaheart Medical, a China-based artificial heart company, unveiled its EVA‑Pulsar ventricular assist device, which weighs only 262 grams, thanks to laser-welding technology. This lightweight design helps closely mimic natural pulse pressure (20–30 mmHg) and supports more physiological cardiac output, mitigating risks associated with continuous-flow devices. EVA‑Pulsar has also gained regulatory approval in Japan, and clinical trials are underway in the U.S., reflecting its global potential.What Are Latest Mergers And Acquisitions In The Ventricular Assist Device Market?
In October 2024, Johnson & Johnson, a US based healthcare company, completed the acquisition of V Wave Ltd., for an undisclosed amount. With this acquisition, Johnson & Johnson aims to deepen its cardiovascular portfolio by integrating V Wave’s minimally invasive Ventura Interatrial Shunt which helps reduce elevated left atrial pressure in patients with heart failure with reduced ejection fraction into its cardiovascular portfolio. V Wave Ltd. is an Israel-based medical device company focused on developing implantable devices to treat heart failure via structural interventional cardiology.Regional Outlook
North America was the largest region in the ventricular assist device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Ventricular Assist Device Market?
The ventricular assist device market consists of sales of pulsatile and nonpulsatile LVADs. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Ventricular Assist Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.69 billion |
| Revenue Forecast In 2035 | $4.34 billion |
| Growth Rate | CAGR of 13.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Flow Mechanism, Design, Application, End Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abiomed, Abbott Laboratories, Medtronic, LivaNova PLC, Terumo Corporation, Berlin Heart GmbH, Cirtec, Jarvik Heart Inc., SynCardia Systems LLC, Calon Cardio-Technology Ltd, Huadong Medicine Co. Ltd, Shenzhen Hexin Zondan Medical Equipment Co. Ltd, Sun Medical Technology Research Corp., CorWave SA, CH Biomedical (USA) Inc, Evaheart Inc, BiVACOR Inc, ReliantHeart Inc, Cardiac Assist Inc, Heart Ware International, Braile Biomedica Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
