
Veterinary Pharmacovigilance Market Report 2026
Global Outlook – By Type (In House, Outsourced), By Solution (Software, Services), By Drug Class (Biologics, Anti Infectives, Other Drug Classes), By Animal Type (Companion Animals, Livestock Animals, Other Animal Types), By End User (Veterinary Hospitals, Veterinary Companies, Academic And Research Institutes) – Market Size, Trends, Strategies, and Forecast to 2035
Veterinary Pharmacovigilance Market Overview
• Veterinary Pharmacovigilance market size has reached to $0.91 billion in 2025 • Expected to grow to $1.67 billion in 2030 at a compound annual growth rate (CAGR) of 12.7% • Growth Driver: Rising Pet Medicine Demand Fuels Veterinary Pharmacovigilance Market Growth • Market Trend: Strengthening Veterinary Drug Safety With Pharmacovigilance And Regulatory Frameworks • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Veterinary Pharmacovigilance Market?
Veterinary pharmacovigilance is the science and activities related to detecting, assessing, understanding, and preventing adverse effects or any other drug-related problems in animals. This involves monitoring and evaluating the safety and effectiveness of veterinary medicines after they have been approved for use. The goal is to ensure that the benefits of veterinary medicines outweigh the risks, promoting animal health and welfare while safeguarding human health and the environment. The main types of veterinary pharmacovigilance include in-house and outsourced. In-house veterinary pharmacovigilance refers to the monitoring and assessment of adverse drug reactions and other safety-related data performed directly by the pharmaceutical or veterinary company. Solutions such as software and services are used for product types such as biologics, anti-infectives, and others. The animal types include companion animals, livestock animals, and others, and end users included are veterinary hospitals, veterinary companies, and academic and research institutes.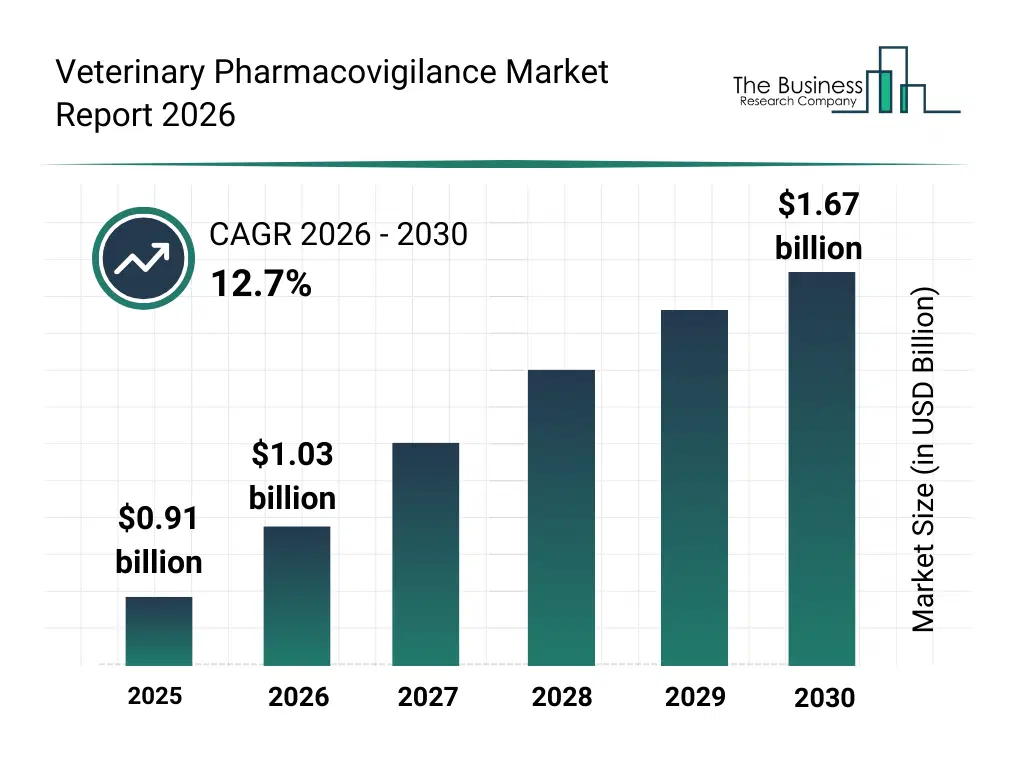
What Is The Veterinary Pharmacovigilance Market Size and Share 2026?
The veterinary pharmacovigilance market size has grown rapidly in recent years. It will grow from $0.91 billion in 2025 to $1.03 billion in 2026 at a compound annual growth rate (CAGR) of 13.1%. The growth in the historic period can be attributed to limited formal pharmacovigilance frameworks in veterinary medicine, increasing use of veterinary pharmaceuticals, growth of companion animal healthcare spending, rising cases of adverse drug reactions in animals, regulatory mandates for drug safety monitoring.What Is The Veterinary Pharmacovigilance Market Growth Forecast?
The veterinary pharmacovigilance market size is expected to see rapid growth in the next few years. It will grow to $1.67 billion in 2030 at a compound annual growth rate (CAGR) of 12.7%. The growth in the forecast period can be attributed to strengthening global veterinary drug regulations, increasing development of biologics and anti infectives, rising outsourcing of pharmacovigilance activities, growing demand for real-time safety monitoring solutions, expanding livestock and companion animal populations. Major trends in the forecast period include increasing regulatory scrutiny on veterinary drug safety, growing adoption of outsourced pharmacovigilance services, rising focus on post-marketing surveillance of veterinary drugs, expansion of adverse event reporting systems, increasing awareness of drug safety among veterinarians.Global Veterinary Pharmacovigilance Market Segmentation
1) By Type: In House, Outsourced 2) By Solution: Software, Services 3) By Drug Class: Biologics, Anti Infectives, Other Drug Classes 4) By Animal Type: Companion Animals, Livestock Animals, Other Animal Types 5) By End User: Veterinary Hospitals, Veterinary Companies, Academic And Research Institutes Subsegments: 1) By In House: Internal Pharmacovigilance Departments, In-House Monitoring And Reporting Systems, Veterinary Adverse Event Management Software 2) By Outsourced: Third-Party Pharmacovigilance Services, Contract Research Organizations (CROs) For Pharmacovigilance, Pharmacovigilance Consulting And Support ServicesWhat Is The Driver Of The Veterinary Pharmacovigilance Market?
The growing demand for animal health and medicinal products is expected to propel the growth of the veterinary pharmacovigilance market going forward. Animal health and medicine products are products designed to maintain the health and well-being of pets, including food, medications, and grooming items. The growing demand for animal health and medicinal products is driven by rising pet ownership, advancements in veterinary medicine, and the need for improved livestock productivity. Additionally, increased awareness of animal welfare and a higher incidence of animal diseases contribute to the surge in demand. Veterinary pharmacovigilance in pet care ensures the safety and efficacy of pet medications by monitoring and assessing adverse effects and ensuring regulatory compliance. For instance, in March 2024, according to the Republic of Estonia's Agency of Medicines (Ravimiamet), an Estonia-based national regulatory authority, in 2023, the veterinary medicinal products sector expanded by 6.9%, reaching 18.6 million euros, up from 17.4 million euros in 2022. Therefore, the growing demand for animal health and medicinal products will drive the growth of the veterinary pharmacovigilance industry.Key Players In The Global Veterinary Pharmacovigilance Market
Major companies operating in the veterinary pharmacovigilance market are Accenture PLC, Zoetis Inc, Boehringer Ingelheim Animal Health, Elanco Animal Health Incorporated, Covetrus Inc, IDEXX Laboratories, Inc., Ceva Santé Animale, Virbac SA, Merck Animal Health, Huvepharma EOOD, Norbrook Laboratories Ltd, ProPharma Group, ArisGlobal LLC, PQE Group, Chanelle Pharma Group Ltd, Clinvet International, Ennov SAS, Sarjen Systems Pvt. Ltd, PharSafer Associates Ltd, Medfiles Oy, Azierta Contract Science Support Consulting, Knoell Germany GmbH, Triveritas Ltd, Leon Research, Biologit, Indivirtus GroupGlobal Veterinary Pharmacovigilance Market Trends and Insights
Major companies operating in the veterinary pharmacovigilance market are focusing on focus on strengthening regulatory frameworks with data analytics developments, such as adverse event reporting (REAS) and safety report preparation, to ensure the safety and effectiveness of veterinary drugs. Adverse event reporting (REAS) and safety report preparation involve documenting and analyzing harmful reactions to veterinary drugs to assess safety and ensure compliance with regulatory standards. For instance, in August 2024, the Ministry of Agriculture and Rural Development, a Mexico-based governmental agency, launched a veterinary pharmacovigilance system. The Veterinary Pharmacovigilance System, a monitoring tool developed by SENASICA to ensure the safety, quality, and efficacy of veterinary drugs in Mexico. It collects data on adverse reactions from veterinary product administrators, including pharmaceutical companies, distributors, veterinary hospitals, and veterinarians, to identify, assess, and mitigate risks associated with these medications. The system features the veterinary pharmacovigilance guide (REAS) for documenting adverse reactions and monitoring efficacy, residue limits, and environmental issues.What Are Latest Mergers And Acquisitions In The Veterinary Pharmacovigilance Market?
In January 2024, Veterinary Pharmaceutical Solutions, a US-based veterinary services company, acquired Diamond Animal Health, Inc. for an undisclosed amount. The acquisition aims to expand Veterinary Pharmaceutical Solutions' product offerings, enhance research and development capabilities, and strengthen operational efficiencies within the animal health industry. Diamond Animal Health Inc. is a US-based contract manufacturing and research company providing regulatory affairs consulting, a part of veterinary pharmacovigilance services.Regional Outlook
North America was the largest region in the veterinary pharmacovigilance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Veterinary Pharmacovigilance Market?
The veterinary pharmacovigilance market consists of revenues earned by entities by providing services such as monitoring adverse drug reactions, assessing medication safety, collecting and analyzing safety data, conducting risk management, and ensuring regulatory compliance for veterinary medicines. The market value includes the value of related goods sold by the service provider or included within the service offering. The veterinary pharmacovigilance market also includes sales of databases, reporting systems, safety assessment tools, and monitoring devices. Values in this market are ‘factory gate’ values, which is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Veterinary Pharmacovigilance Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.03 billion |
| Revenue Forecast In 2035 | $1.67 billion |
| Growth Rate | CAGR of 13.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Solution, Drug Class, Animal Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Accenture PLC, Zoetis Inc, Boehringer Ingelheim Animal Health, Elanco Animal Health Incorporated, Covetrus Inc, IDEXX Laboratories, Inc., Ceva Santé Animale, Virbac SA, Merck Animal Health, Huvepharma EOOD, Norbrook Laboratories Ltd, ProPharma Group, ArisGlobal LLC, PQE Group, Chanelle Pharma Group Ltd, Clinvet International, Ennov SAS, Sarjen Systems Pvt. Ltd, PharSafer Associates Ltd, Medfiles Oy, Azierta Contract Science Support Consulting, Knoell Germany GmbH, Triveritas Ltd, Leon Research, Biologit, Indivirtus Group |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
