
Viral Diagnostic Test Kits Market Report 2026
Global Outlook – By Testing Approach (Traditional Tests, Rapid Test), By Test Type (Direct Fluorescent Antibody (DFA) Test, Immunochromatographic Assay, Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) Based Test, Agglutination Assay, Solid Phase Assay, Flow Through Assay, Other Test Types), By End-User (Research Laboratories, Clinics, Hospitals, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Viral Diagnostic Test Kits Market Overview
• Viral Diagnostic Test Kits market size has reached to $17.48 billion in 2025 • Expected to grow to $24.87 billion in 2030 at a compound annual growth rate (CAGR) of 7.1% • Growth Driver: Rising Prevalence of Viral Infections Driving Growth of the Viral Diagnostic Test Kits Market Due to Increased Demand for Rapid and Accurate Detection • Market Trend: Innovative Product Development in the Viral Diagnostic Test Kits Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Diagnostic Test Kits Market?
Viral diagnostic test kits are medical devices or tools used to detect the presence of viral infections in clinical samples taken from patients. These kits typically contain reagents, antibodies, or other substances interacting with specific viral antigens or genetic material to produce a measurable signal. The tests can identify the presence of viral particles, antigens, or genetic material (like RNA or DNA) in samples such as blood, saliva, nasal swabs, or other bodily fluids. The main tests of the viral diagnostic test kits are traditional tests and rapid tests. Traditional viral diagnostic tests refer to established laboratory-based methods that typically involve culturing the virus, detecting viral antigens, or measuring antibody responses. The various test types include direct fluorescent antibody (DFA) test, immunochromatographic assay, reverse transcriptase polymerase chain reaction (RT-PCR) based test, agglutination assay, solid phase assay, flow through assay, and others and are used by various end-users such as research Laboratories, clinics, hospitals, and others.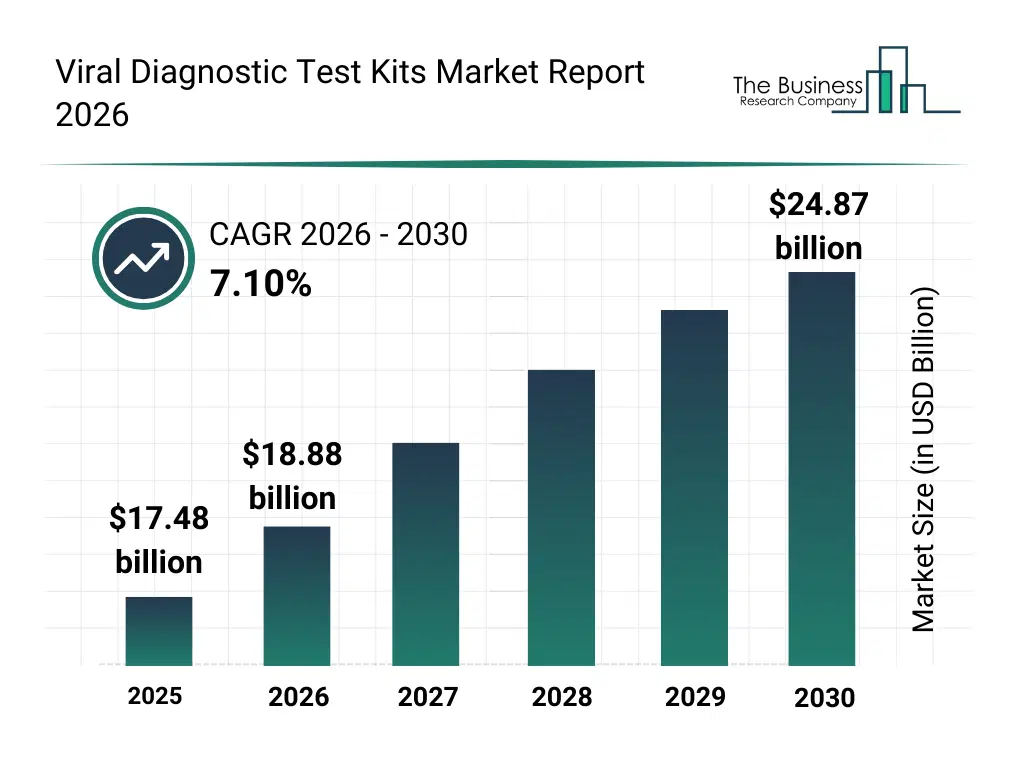
What Is The Viral Diagnostic Test Kits Market Size and Share 2026?
The viral diagnostic test kits market size has grown strongly in recent years. It will grow from $17.48 billion in 2025 to $18.88 billion in 2026 at a compound annual growth rate (CAGR) of 8.0%. The growth in the historic period can be attributed to recurring outbreaks of viral infections, expansion of clinical diagnostic laboratories, increased adoption of PCR-based testing, rising demand for early disease detection, improvements in laboratory infrastructure.What Is The Viral Diagnostic Test Kits Market Growth Forecast?
The viral diagnostic test kits market size is expected to see strong growth in the next few years. It will grow to $24.87 billion in 2030 at a compound annual growth rate (CAGR) of 7.1%. The growth in the forecast period can be attributed to increasing preparedness for future pandemics, growing demand for decentralized testing, rising investments in molecular diagnostics, expansion of home-based testing solutions, technological advancements in assay sensitivity and speed. Major trends in the forecast period include increasing adoption of rapid diagnostic test kits, rising use of molecular-based viral detection, growing demand for point-of-care testing solutions, expansion of multiplex viral assay development, enhanced focus on high-sensitivity diagnostic accuracy.Global Viral Diagnostic Test Kits Market Segmentation
1) By Testing Approach: Traditional Tests, Rapid Test 2) By Test Type: Direct Fluorescent Antibody (DFA) Test, Immunochromatographic Assay, Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) Based Test, Agglutination Assay, Solid Phase Assay, Flow Through Assay, Other Test Types 3) By End-User: Research Laboratories, Clinics, Hospitals, Other End Users Subsegments: 1) By Traditional Tests: Enzyme-Linked Immunosorbent Assay (ELISA), Polymerase Chain Reaction (PCR) Tests, Western Blot, Viral Culture Testing 2) By Rapid Test: Lateral Flow Assays (LFA), Immunochromatographic Strip Tests, Reverse Transcription Loop-Mediated Isothermal Amplification (RT-LAMP), Rapid Antigen TestsWhat Is The Driver Of The Viral Diagnostic Test Kits Market?
The growing prevalence of viral infections is expected to propel the growth of the viral diagnostic test kits market going forward. Viral infections are caused by microscopic viruses that invade host cells, replicate within them, and can lead to symptoms ranging from mild illness to life-threatening conditions. The rise in viral infections is driven by factors such as global travel, urbanization, unsafe injection practices, and limited access to timely diagnosis and treatment. Viral diagnostic test kits, which detect viral nucleic acids (RNA or DNA), antigens, or antibodies in biological samples such as blood or swabs, are critical for identifying infections, guiding treatment decisions, and controlling outbreaks. For instance, in April 2025, according to the Centers for Disease Control and Prevention (CDC), a US-based federal agency, there were 2,214 new acute hepatitis?B cases in 2023, corresponding to an estimated 14,400 infections after accounting for underreporting, and 17,650 newly reported chronic hepatitis?B cases. The same report also shows 4,966 new acute hepatitis?C cases and 101,525 new chronic hepatitis?C cases in 2023. Therefore, the rising prevalence of viral infections is driving the growth of the viral diagnostic test kits industry.Key Players In The Global Viral Diagnostic Test Kits Market
Major companies operating in the viral diagnostic test kits market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson and Company, Quest Diagnostics Incorporated, Grifols S.A., Hologic Inc., PerkinElmer Inc., bioMérieux SA, Quidel Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Ortho Clinical Diagnostics, Seegene Inc., OraSure Technologies Inc., Meridian Bioscience Inc., DiaSorin S.p.A., Trinity Biotech plcGlobal Viral Diagnostic Test Kits Market Trends and Insights
Major companies operating in the viral diagnostic test kits market are developing innovative products, such as virus detection kit to sustain their position in the market. A virus detection kit refers to a diagnostic tool designed to identify the presence of specific viruses in biological samples, providing rapid and accurate results for effective disease monitoring and management. For instance, in August 2024, BlueStain Global Ltd, a UK-based company, launched MPox virus detection kit. The Bluestain MPox virus detection kit is a rapid diagnostic test designed to detect the mpox virus with high speed and accuracy, delivering results in just 15 to 30 minutes. This innovative kit is intended to enhance public health responses by facilitating early detection and containment of mpox outbreaks, particularly in resource-limited settings.What Are Latest Mergers And Acquisitions In The Viral Diagnostic Test Kits Market?
In June 2023, Avacta Life Sciences, a UK-based life sciences company, acquired Coris Bioconcept for £7.4 million ($9.35 million). With this acquisition, Avacta aims to expand its rapid diagnostic test portfolio, enhancing healthcare diagnostics capabilities and broadening market reach, particularly in infectious disease management and point-of-care testing contexts. Coris Bioconcept is a Belgium-based company that develops and manufactures rapid diagnostic test kits, primarily lateral flow tests, to detect viral and bacterial pathogens.Regional Outlook
North America was the largest region in the viral diagnostic test kits market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Viral Diagnostic Test Kits Market?
The viral diagnostic test kits market consists of sales of viral culture kits, molecular diagnostic kits, antibody detection kits, antigen detection kits, and multiplex PCR kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Viral Diagnostic Test Kits Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $18.88 billion |
| Revenue Forecast In 2035 | $24.87 billion |
| Growth Rate | CAGR of 8.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Testing Approach, Test Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson and Company, Quest Diagnostics Incorporated, Grifols S.A., Hologic Inc., PerkinElmer Inc., bioMérieux SA, Quidel Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Ortho Clinical Diagnostics, Seegene Inc., OraSure Technologies Inc., Meridian Bioscience Inc., DiaSorin S.p.A., Trinity Biotech plc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
