Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report 2026

Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report 2026
Global Outlook – By Service Type (Process Development, Manufacturing, Analytical Testing, Fill-Finish, Other Service Types), By Vector Type (Adenoviral Vectors, Lentiviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Vector Types), By Workflow (Upstream Manufacturing, Downstream Manufacturing), By Application (Gene Therapy, Vaccines, Cell Therapy, Other Applications), By End-User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Overview
• Viral Vector Contract Development And Manufacturing Organization (CDMO) market size has reached to $1.47 billion in 2025 • Expected to grow to $3.01 billion in 2030 at a compound annual growth rate (CAGR) of 15.3% • Growth Driver: Rising Investment In Gene Therapy Research Driving The Growth Of The Market Due To Its Potential For Long-Term And Curative Treatments • Market Trend: Advancements In Viral Vector Manufacturing To Enhance Scalable And Efficient Gene Therapy Production • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
A viral vector contract development and manufacturing organization (CDMO) is a specialized company that focuses on the development and large-scale production of viral vectors used in advanced biological therapies. It provides the technical capabilities, infrastructure, and regulatory compliance necessary to support the entire process of viral vector manufacturing. The main services of viral vector contract development and manufacturing organizations (CDMO) include process development, manufacturing, analytical testing, fill-finish, and others. Process development in pharmaceutical manufacturing involves designing and optimizing the production process to ensure efficient, scalable, and reproducible manufacturing. Different vector types include adenoviral vectors, lentiviral vectors, adeno-associated viral vectors, retroviral vectors, and others, and it offers various workflows, such as upstream manufacturing and downstream manufacturing. It is applied for gene therapy, vaccines, cell therapy, and others and is used by several end-users, including pharmaceutical and biotechnology companies, academic and research institutes, and others.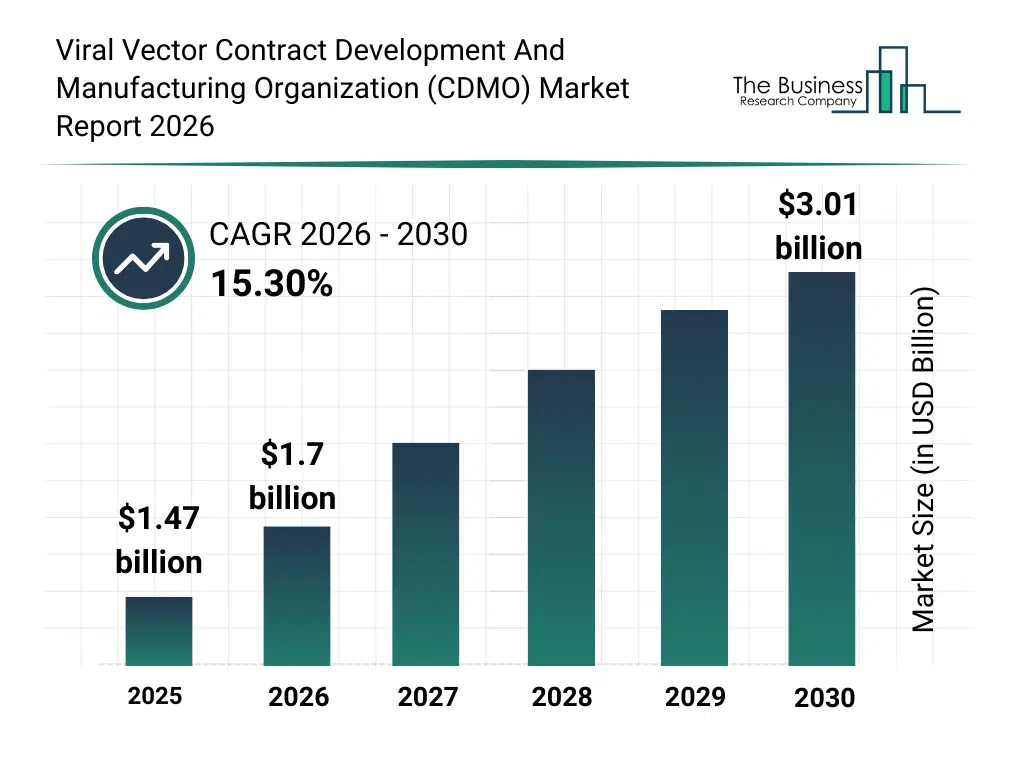
What Is The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Size and Share 2026?
The viral vector contract development and manufacturing organization (cdmo) market size has grown rapidly in recent years. It will grow from $1.47 billion in 2025 to $1.7 billion in 2026 at a compound annual growth rate (CAGR) of 15.6%. The growth in the historic period can be attributed to growing clinical pipeline of gene therapies, early outsourcing of viral vector production, expansion of academic research collaborations, limited in-house manufacturing capabilities of biotech firms, increasing regulatory approvals for viral vectors.What Is The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Growth Forecast?
The viral vector contract development and manufacturing organization (cdmo) market size is expected to see rapid growth in the next few years. It will grow to $3.01 billion in 2030 at a compound annual growth rate (CAGR) of 15.3%. The growth in the forecast period can be attributed to rising commercialization of gene and cell therapies, increasing investments in dedicated viral vector facilities, growing demand for scalable manufacturing platforms, expansion of global cdmo partnerships, advances in high-yield vector production technologies. Major trends in the forecast period include expanding capacity for large-scale viral vector production, increasing adoption of single-use manufacturing technologies, growing demand for end-to-end cdmo services, rising focus on regulatory-compliant cGMP facilities, enhanced integration of advanced analytics in vector manufacturing.Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Segmentation
1) By Service Type: Process Development, Manufacturing, Analytical Testing, Fill-Finish, Other Service Types 2) By Vector Type: Adenoviral Vectors, Lentiviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Vector Types 3) By Workflow: Upstream Manufacturing, Downstream Manufacturing 4) By Application: Gene Therapy, Vaccines, Cell Therapy, Other Applications 5) By End-User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Other End-Users Subsegments: 1) By Process Development: Upstream Process Development, Downstream Process Development, Analytical Development, Process Characterization 2) By Manufacturing: Clinical Manufacturing, Commercial Manufacturing, cGMP Manufacturing, Cell Bank Production, Fill-Finish Services 3) By Analytical Testing: Release Testing, Stability Testing, Bioassays, Characterization Studies, Regulatory Compliance Testing 4) By Fill-Finish: Aseptic Filling, Lyophilization, Final Packaging, Cold Chain Management, Labeling And Serialization 5) By Other Service Types: Regulatory Support, Supply Chain Management, Technology Transfer, Clinical Trial Support, Cold Chain LogisticsWhat Is The Driver Of The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
The growing investment in gene therapy research is expected to propel the growth of the viral vector contract development and manufacturing organization (CDMO) market going forward. Gene therapy research focuses on exploring methods to alter or regulate genes in human cells to correct genetic disorders or combat various diseases. Investment in gene therapy research is rising due to its potential to provide long-term or permanent cures for previously untreatable genetic diseases, driving strong interest from both healthcare innovators and investors. Viral vector CDMOs support gene therapy research by offering expert knowledge and advanced production facilities to manufacture high-quality viral vectors, which are crucial for the safe and effective delivery of therapeutic genes. For instance, in July 2025, according to the Office for Life Sciences, a UK-based government department, the government has committed up to $800 million (£600 million) to develop one of the world’s most advanced, secure, and AI-compatible health data platforms, integrating genomic, diagnostic, and clinical information at a population level to transform NHS and broader healthcare data into a hub for global research trials and AI investment. Therefore, the growing investment in gene therapy research is driving the growth of the viral vector contract development and manufacturing organization (CDMO) industry.Key Players In The Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market
Major companies operating in the viral vector contract development and manufacturing organization (cdmo) market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Genesail Biotech Co. Ltd., CEVEC Pharmaceuticals, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc.Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Trends and Insights
Major companies operating in the viral vector contract development and manufacturing organization (CDMO) market are focusing on developing advanced solutions, such as AAV manufacturing solutions, to support scalable and consistent production. AAV manufacturing solutions involve techniques and systems developed to efficiently produce safe and effective adeno-associated virus vectors for gene therapy. For instance, in August 2025, ProBio, a US-based contract development and manufacturing organization (CDMO), launched cGMP Adeno-Associated Virus (AAV) manufacturing services at its 128,000 sq. ft. cutting-edge facility in Hopewell, New Jersey. This expansion is intended to meet the increasing demand for high-quality viral vector production while demonstrating ProBio’s dedication to supporting the development of innovative gene therapies. The Hopewell facility is purpose-built to provide complete AAV manufacturing solutions in compliance with global regulatory and quality standards. ProBio’s capabilities now include fully integrated services such as GMP plasmid DNA production, AAV vector manufacturing, and final drug product formulation with aseptic fill/finish, all in a single U.S. location. This consolidated approach enhances coordination, reduces process handoffs, and speeds up timelines across the drug development lifecycle.What Are Latest Mergers And Acquisitions In The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
In May 2023, Siegfried, a Switzerland-based life sciences company, acquired 95% stake in DINAMIQS for an undisclosed amount. As a result of the acquisition, Siegfried will scale DINAMIQS’ capabilities to commercial level, positioning it as a leading biotech CDMO for cell and gene therapies. This move also strengthens Siegfried’s presence in the biologics sector and is expected to generate substantial mid- to long-term growth opportunities in a rapidly evolving market segment. DINAMIQS is a Switzerland-based company providing comprehensive contract development and manufacturing services for viral vectors.Regional Insights
North America was the largest region in the viral vector contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
The viral vector contract development and manufacturing organization (CDMO) market consists of revenues earned by entities by providing services such as cell line development, preclinical material production, quality control testing, and supply chain management. The market value includes the value of related goods sold by the service provider or included within the service offering. The viral vector contract development and manufacturing organization (CDMO) market also includes sales of helper plasmids, packaging cell lines, producer cell banks, and purified viral vector components. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report 2026?
The viral vector contract development and manufacturing organization (cdmo) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector contract development and manufacturing organization (cdmo) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.7 billion |
| Revenue Forecast In 2035 | $3.01 billion |
| Growth Rate | CAGR of 15.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Vector Type, Workflow, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Genesail Biotech Co. Ltd., CEVEC Pharmaceuticals, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Viral Vector Contract Development And Manufacturing Organization (CDMO) market was valued at $1.47 billion in 2025, increased to $1.7 billion in 2026, and is projected to reach $3.01 billion by 2030.
request a sample hereThe global Viral Vector Contract Development And Manufacturing Organization (CDMO) market is expected to grow at a CAGR of 15.3% from 2026 to 2035 to reach $3.01 billion by 2035.
request a sample hereSome Key Players in the Viral Vector Contract Development And Manufacturing Organization (CDMO) market Include, Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Genesail Biotech Co. Ltd., CEVEC Pharmaceuticals, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc. .
request a sample hereMajor trend in this market includes: Advancements In Viral Vector Manufacturing To Enhance Scalable And Efficient Gene Therapy Production. For further insights on this market.
request a sample hereNorth America was the largest region in the viral vector contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the viral vector contract development and manufacturing organization (cdmo) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here