
Viral Vector Manufacturing Market Report 2026
Global Outlook – By Type (Adenoviral Vectors, Adeno-Associated Viral Vectors, Lentiviral Vectors, Retroviral Vectors, Other Types), By Disease (Cancer, Genetic Disorders, Infectious Diseases, Other Diseases), By Workflow (Upstream Processing, Downstream Processing), By Application (Gene And Cell Therapy Development, Vaccine Development, Biopharmaceutical And Pharmaceutical Discovery, Biomedical Research), By End-User (Research Organizations, Biotech And Pharmaceutical Companies, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Viral Vector Manufacturing Market Overview
• Viral Vector Manufacturing market size has reached to $8.26 billion in 2025 • Expected to grow to $21.31 billion in 2030 at a compound annual growth rate (CAGR) of 21.1% • Growth Driver: Impact Of Rising Cancer And Infectious Diseases On The Viral Vector Manufacturing Market • Market Trend: Advanced Analytical Viral Vector Platform Propel Market Growth • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Vector Manufacturing Market?
Viral vector manufacturing refers to producing viral vectors, tools that deliver genetic material into cells. A viral vector is a tool for gene transfer that can manipulate a particular cell type or tissue to express therapeutic genes. The main types of viral vector manufacturing are adenoviral vectors, adeno-associated viral vectors, lentiviral vectors, retroviral vectors, and others. Adenoviral vectors refer to double-stranded DNA vectors that are not enclosed. The various diseases covered are cancer, genetic disorders, infectious diseases, and others with various workflows such as upstream processing and downstream processing. The various applications used are gene and cell therapy development, vaccine development, biopharmaceutical and pharmaceutical discovery, and biomedical research used in research organizations, biotech and pharmaceutical companies, and others.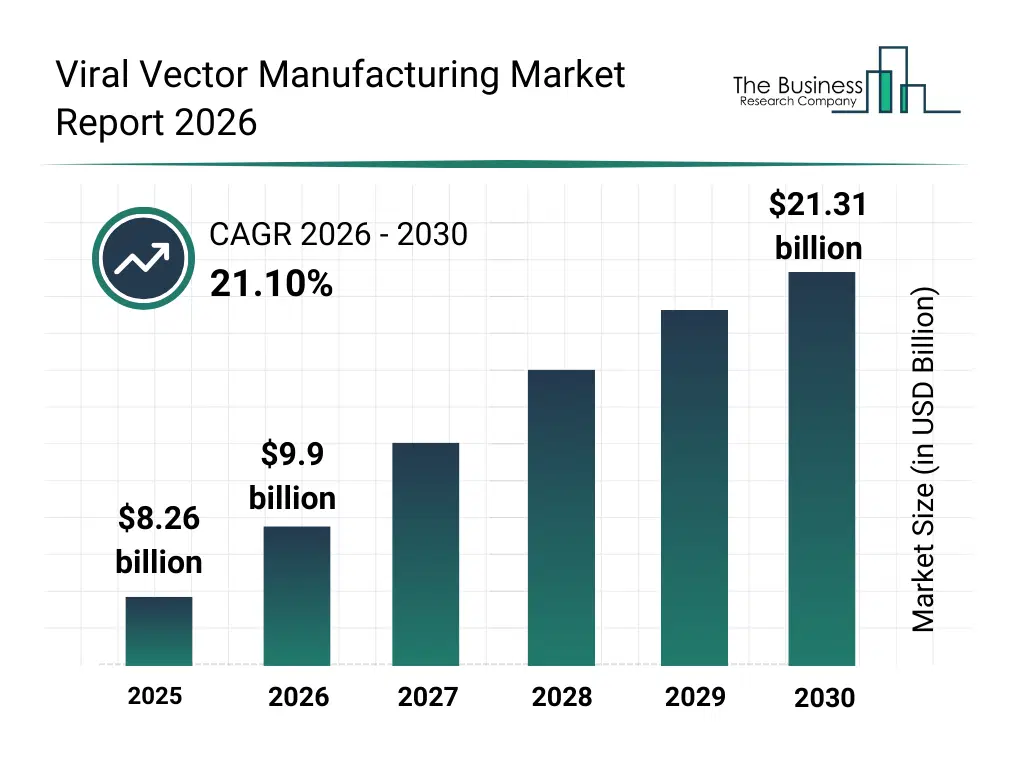
What Is The Viral Vector Manufacturing Market Size and Share 2026?
The viral vector manufacturing market size has grown rapidly in recent years. It will grow from $8.26 billion in 2025 to $9.9 billion in 2026 at a compound annual growth rate (CAGR) of 19.9%. The growth in the historic period can be attributed to limited manufacturing capacity, high production cost, reliance on traditional gene delivery methods, regulatory barriers, fragmented supply chains.What Is The Viral Vector Manufacturing Market Growth Forecast?
The viral vector manufacturing market size is expected to see exponential growth in the next few years. It will grow to $21.31 billion in 2030 at a compound annual growth rate (CAGR) of 21.1%. The growth in the forecast period can be attributed to growing demand for gene and cell therapies, advancements in viral vector technology, increased R&D investments, expansion of contract manufacturing organizations, rising adoption of personalized medicine. Major trends in the forecast period include gene therapy innovation, viral vector safety optimization, scalable manufacturing processes, regulatory compliance enhancement, advanced upstream and downstream processing.Global Viral Vector Manufacturing Market Segmentation
1) By Type: Adenoviral Vectors, Adeno-Associated Viral Vectors, Lentiviral Vectors, Retroviral Vectors, Other Types 2) By Disease: Cancer, Genetic Disorders, Infectious Diseases, Other Diseases 3) By Workflow: Upstream Processing, Downstream Processing 4) By Application: Gene And Cell Therapy Development, Vaccine Development, Biopharmaceutical And Pharmaceutical Discovery, Biomedical Research 5) By End-User: Research Organizations, Biotech And Pharmaceutical Companies, Other End Users Subsegments: 1) By Adenoviral Vectors: Serotype 5, Serotype 2, Serotype 26 2) By Adeno-Associated Viral Vectors: AAV2, AAV5, AAV8, AAV9, Others 3) By Lentiviral Vectors: HIV-1-Based Lentiviral Vectors, SIV-Based Lentiviral Vectors, Others 4) By Retroviral Vectors: Moloney Murine Leukemia Virus (MoMLV), Gammaretroviral Vectors, Others 5) By Other Types: Sendai Viral Vectors, Vesicular Stomatitis Virus (VSV)-Based Vectors, Measles Virus Vectors, OthersWhat Is The Driver Of The Viral Vector Manufacturing Market?
The increase in the prevalence of cancer and infectious diseases is expected to propel the growth of the viral vector manufacturing market going forward. An infectious disease refers to a condition brought on by a virus or its toxic byproduct and spread to a susceptible host via contact with an infected person, animal, or item. Cancer refers to a broad range of illnesses that can develop in any organ or tissue of the body when aberrant cells grow out of control, cross their usual boundaries to invade neighboring organs, and spread to other organs. Tumor antigens, or proteins on tumor cells, are produced via viral vectors to elicit the body's anticancer immune response. In pre-clinical and clinical trials, viral vectors have served as vaccines against various infectious illnesses. For instance, in July 2024, according to the Australian Institute of Health and Welfare, an Australia-based government agency, in Australia the number of cancer cases diagnosed increased from 160,570 in 2022 to 164,694 in 2023, reflecting a notable rise over the year. This upward trend highlights the growing prevalence of cancer within the country. Furthermore, in August 2024, according to the data published by the UK Health Security Agency, a UK-based executive agency, in England, the UK Health Security Agency (UKHSA) reported 368 measles cases in 2023, marking a nearly sevenfold increase from the 53 cases reported in 2022, with the West Midlands and London accounting for 44% and 33% of the cases, respectively. Therefore, the increase in the prevalence of cancer and infectious diseases is driving the growth of the viral vector manufacturing industry.Key Players In The Global Viral Vector Manufacturing Market
Major companies operating in the viral vector manufacturing market are Sanofi S.A., Merck Group, FUJIFILM Holdings Corporation, Lonza Group, Catalent Inc., AGC Biologics, Ultragenyx Pharmaceutical Inc., Novasep Holding SAS, Aldevron LLC, Oxford Biomedica plc, LakePharma Inc., Voyager Therapeutics Inc., Mustang Bio Inc., Regenxbio Inc., VGXI Inc., BioNTech IMFS GmbH, FinVector Oy, Vigene Biosciences Inc., Univercells Technologies, Sirion-Biotech GmbH, Cevec Pharmaceuticals GmbH, Batavia Biosciences BVGlobal Viral Vector Manufacturing Market Trends and Insights
Major companies operating in the viral vector manufacturing market are focusing on developing advanced products, such as viral vector platforms specifically designed for viral vector production and manufacturing. Viral vector platforms are comprehensive systems tailored to efficiently produce and process viral vectors for applications such as gene therapy and vaccine development. For instance, in May 2023, AGC Biologics, a US-based global biopharmaceutical contract development and manufacturing organization (CDMO), launched the BravoAAV and ProntoLVV platforms, designed to offer flexible and accelerated vector development and manufacturing for cell and gene therapy programs. These platforms, designed for flexible and accelerated vector development and manufacturing in cell and gene therapy programs, offer GMP product delivery in nine months, supported by a global regulatory and supply network and in-house plasmid DNA services. With custom processes, templated material approaches, and prequalified analytical methods, these platforms are significant contributors to the viral vector manufacturing market's projected growth, reaching $5.5 billion by 2035, by meeting the increasing demand for efficient development and delivery of life-changing gene therapies and vaccines.What Are Latest Mergers And Acquisitions In The Viral Vector Manufacturing Market?
In January 2024, Oxford Biomedica Plc, a UK-based biotechnology company, acquired ABL Europe from Institut Mérieux, for $16 million (15 million euros). Through this acquisition, Oxford Biomedica will the production facilities in Lyon and Strasbourg, France, and will be able to offer multi-viral vector CDMO capabilities, boasting six manufacturing sites in the EU, U.S., and U.K. ABL Europe is a France-based contract development and manufacturing organization (CDMO) that provides viral vector manufacturing services.Regional Insights
North America was the largest region in the viral vector manufacturing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector Manufacturing Market?
The viral vector manufacturing market includes revenues earned by entities through the production, packaging, and labeling of viral vector vaccines. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector Manufacturing Market Report 2026?
The viral vector manufacturing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector manufacturing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector Manufacturing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $9.9 billion |
| Revenue Forecast In 2035 | $21.31 billion |
| Growth Rate | CAGR of 19.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Disease, Workflow, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi S.A., Merck Group, FUJIFILM Holdings Corporation, Lonza Group, Catalent Inc., AGC Biologics, Ultragenyx Pharmaceutical Inc., Novasep Holding SAS, Aldevron LLC, Oxford Biomedica plc, LakePharma Inc., Voyager Therapeutics Inc., Mustang Bio Inc., Regenxbio Inc., VGXI Inc., BioNTech IMFS GmbH, FinVector Oy, Vigene Biosciences Inc., Univercells Technologies, Sirion-Biotech GmbH, Cevec Pharmaceuticals GmbH, Batavia Biosciences BV |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
