
Viral Vector Production (Research-Use) Market Report 2026
Global Outlook – By Type (Adeno-Associated Virus (AAV), Lentivirus, Adenovirus, Retrovirus, Other Types), By Production (Transient Transfection, Stable Cell Line, Viral Packaging System, Other Production Methods), By Workflow (Upstream Processing, Downstream Processing), By Application (Gene Therapy, Vaccines, Oncology, Infectious Diseases, Other Applications), By End-Use (Pharmaceutical And Biopharmaceutical Companies, Research Institutes) – Market Size, Trends, Strategies, and Forecast to 2035
Viral Vector Production (Research-Use) Market Overview
• Viral Vector Production (Research-Use) market size has reached to $1.77 billion in 2025 • Expected to grow to $3.56 billion in 2030 at a compound annual growth rate (CAGR) of 14.9% • Growth Driver: Rising Occurrence Of Genetic Disorders Fueling The Growth Of The Market Due To Advanced Parental Age And Gene Therapy Demand • Market Trend: Technological Innovation In Viral Vector Production Enhances Research Efficiency And Scalability • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Vector Production (Research-Use) Market?
Viral vector production (research-use) refers to the laboratory-based process of generating modified viruses that can efficiently deliver genetic material into target cells for experimental purposes. These vectors are engineered to be replication-deficient and safe for use in controlled research environments. The process involves vector design, production in host cells, and purification to ensure quality, safety, and effectiveness in scientific studies. The main types of viral vector production (research-use) are adeno-associated virus (AAV), lentivirus, adenovirus, retrovirus, and others. Adeno-associated viruses are tiny viruses that carry genetic information to cells; they are members of the Parvoviridae family and cannot reproduce independently, instead relying on other viruses. The various production methods include transient transfection, stable cell lines, viral packaging systems, and others by various workflows, including upstream processing and downstream processing. The different applications include gene therapy, vaccines, oncology, infectious diseases, and others and are used for several end-users, including pharmaceutical and biopharmaceutical companies and research institutes.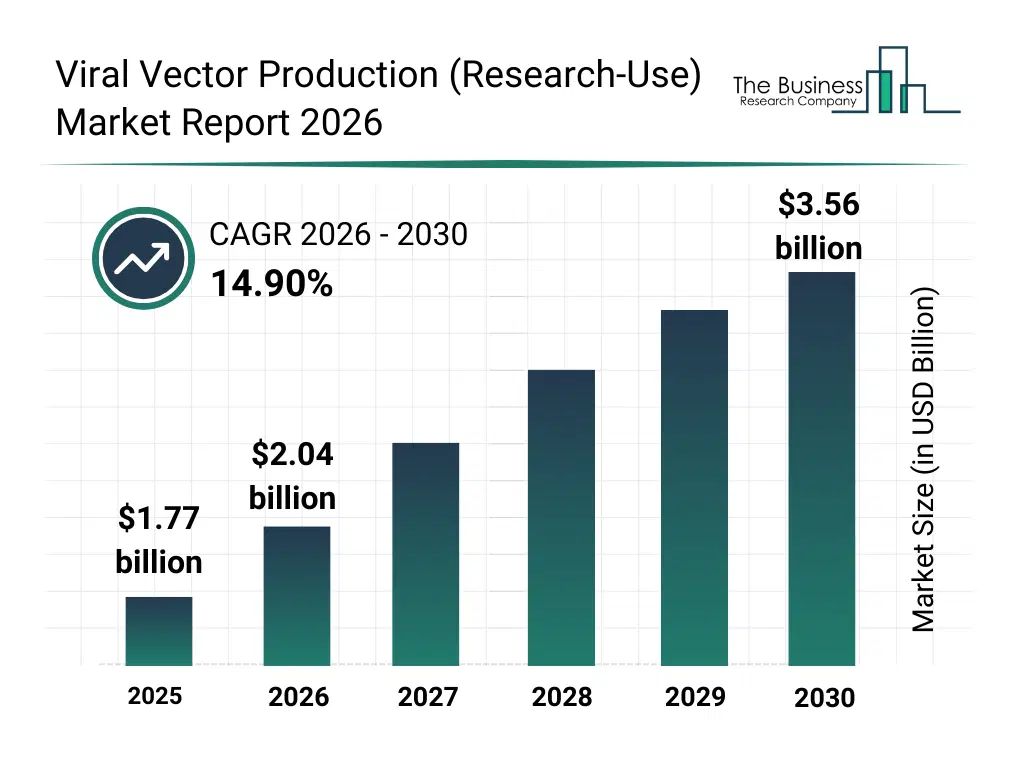
What Is The Viral Vector Production (Research-Use) Market Size and Share 2026?
The viral vector production (research-use) market size has grown rapidly in recent years. It will grow from $1.77 billion in 2025 to $2.04 billion in 2026 at a compound annual growth rate (CAGR) of 15.2%. The growth in the historic period can be attributed to increasing investment in gene therapy research, expansion of academic and translational research programs, availability of advanced cell culture technologies, rising use of viral vectors in oncology studies, growth of contract research activities.What Is The Viral Vector Production (Research-Use) Market Growth Forecast?
The viral vector production (research-use) market size is expected to see rapid growth in the next few years. It will grow to $3.56 billion in 2030 at a compound annual growth rate (CAGR) of 14.9%. The growth in the forecast period can be attributed to increasing demand for next-generation gene delivery tools, rising adoption of automation in research laboratories, expansion of CRISPR and gene editing research, growing collaboration between academia and biopharma, increasing focus on reproducibility and standardization. Major trends in the forecast period include increasing adoption of scalable viral vector manufacturing platforms, rising demand for high-purity research-grade vectors, growing use of suspension cell culture systems, expansion of automated upstream and downstream processing, enhanced focus on vector characterization and quality control.Global Viral Vector Production (Research-Use) Market Segmentation
1) By Type: Adeno-Associated Virus (AAV), Lentivirus, Adenovirus, Retrovirus, Other Types 2) By Production: Transient Transfection, Stable Cell Line, Viral Packaging System, Other Production Methods 3) By Workflow: Upstream Processing, Downstream Processing 4) By Application: Gene Therapy, Vaccines, Oncology, Infectious Diseases, Other Applications 5) By End-Use: Pharmaceutical And Biopharmaceutical Companies, Research Institutes Subsegments: 1) By Adeno-Associated Virus: Single-Stranded Adeno-Associated Virus, Self-Complementary Adeno-Associated Virus, Hybrid Adeno-Associated Virus 2) By Lentivirus: Human Immunodeficiency Virus-Based Lentivirus, Feline Immunodeficiency Virus-Based Lentivirus, Equine Infectious Anemia Virus-Based Lentivirus 3) By Adenovirus: Replication-Competent Adenovirus, Replication-Deficient Adenovirus, Helper-Dependent Adenovirus 4) By Retrovirus: Gamma Retrovirus, Spumavirus (Foamy Virus), Alpha Retrovirus 5) By Other Types: Herpes Simplex Virus Vectors, Baculovirus Vectors, Sendai Virus VectorsWhat Is The Driver Of The Viral Vector Production (Research-Use) Market?
The rising occurrence of genetic disorders is expected to propel the growth of the viral vector production (research-use) market going forward. Genetic disorders are diseases or conditions caused by abnormalities in an individual's deoxyribonucleic acid, which can be inherited from one or both parents or occur due to new genetic mutations. The rise in genetic disorders is due to increasing parental age, raising the likelihood of genetic mutations being passed to offspring. The growing prevalence of genetic disorders supports the growth of viral vector production (research-use) by driving demand for advanced gene delivery tools to study disease mechanisms and develop potential therapeutic approaches. For instance, in November 2024, according to the Centers for Disease Control and Prevention (CDC), a US-based government public health agency, Trisomy 21 (Down syndrome) occurs in about 1 in 643 births, resulting in approximately 5,713 new cases annually, and Trisomy 18 occurs in about 1 in 3,336 births, or roughly 1,101 cases yearly. Therefore, the rising occurrence of genetic disorders is expected to propel the growth of the viral vector production (research-use) industry.Key Players In The Global Viral Vector Production (Research-Use) Market
Major companies operating in the viral vector production (research-use) market are Sanofi S.A., Thermo Fisher Scientific Inc., Merck KGaA, Becton Dickinson and Company, Lonza Group AG, Sartorius AG, Charles River Laboratories International Inc., FUJIFILM Diosynth Biotechnologies U.S.A. Inc., Curia Inc., AGC Biologics GmbH, SK pharmteco Inc., Oxford Biomedica Plc, Waisman Biomanufacturing, Voyager Therapeutics Inc., FinVector Oy, Batavia Biosciences BV., GENEZEN LABORATORIES, BioNTech IMFS GmbH, VGXI Inc., Tonix Pharmaceuticals, Cell and Gene Therapy Catapult, Mustang Bio Inc.Global Viral Vector Production (Research-Use) Market Trends and Insights
Major companies operating in the viral vector production market are focusing on developing advanced solutions, such as specialized cell culture media, to improve transfection efficiency, increase viral vector yields, and ensure consistent quality for research applications. Cell culture media are nutrient-rich solutions designed to support the growth and maintenance of cells outside their natural environment. They contain essential components like amino acids, vitamins, and growth factors optimized for specific cell types and applications. For instance, in January 2023, FUJIFILM Irvine Scientific, a US-based life sciences company, introduced BalanCD HEK293 Viral Feed, the world’s first chemically defined nutrient feed specifically optimized for HEK293 suspension cultures used in AAV viral vector manufacturing. Through this launch, the company aims to significantly boost adeno-associated virus (AAV) yields, with up to 67% improvement in viral packaging efficiency compared to basal media. The feed is designed for scalable gene therapy and biotech workflows, offering consistent performance across formats and compatibility with multiple media systems.What Are Latest Mergers And Acquisitions In The Viral Vector Production (Research-Use) Market?
In January 2024, Oxford Biomedica Plc, a UK-based gene and cell therapy company, acquired ABL Europe for an undisclosed amount. With this acquisition, Oxford Biomedica aims to expand its presence in the European market and enhance its contract development and manufacturing organization (CDMO) offerings, particularly in the field of viral vector manufacturing. ABL Europe is a France-based company specializing in the development and production of viral vectors.Regional Insights
North America was the largest region in the virtual vector production (research-use) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector Production (Research-Use) Market?
The viral vector production (research-use) market consists of sales of baculovirus, herpes simplex virus (HSV), vaccinia virus, vesicular stomatitis virus (VSV), hybrid viral vectors, and measles virus vectors. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector Production (Research-Use) Market Report 2026?
The viral vector production (research-use) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector production (research-use) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector Production (Research-Use) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.04 billion |
| Revenue Forecast In 2035 | $3.56 billion |
| Growth Rate | CAGR of 15.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Production, Workflow, Application, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi S.A., Thermo Fisher Scientific Inc., Merck KGaA, Becton Dickinson and Company, Lonza Group AG, Sartorius AG, Charles River Laboratories International Inc., FUJIFILM Diosynth Biotechnologies U.S.A. Inc., Curia Inc., AGC Biologics GmbH, SK pharmteco Inc., Oxford Biomedica Plc, Waisman Biomanufacturing, Voyager Therapeutics Inc., FinVector Oy, Batavia Biosciences BV., GENEZEN LABORATORIES, BioNTech IMFS GmbH, VGXI Inc., Tonix Pharmaceuticals, Cell and Gene Therapy Catapult, Mustang Bio Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
