
Virtual Clinical Trials Market Report 2026
Global Outlook – By Design (Observational Trials, Interventional Trials, Expanded Access Trials), By Phases (Phase II, Phase III, Phase IV), By Indication (CNS, Autoimmune Or Inflammation, Cardiovascular Disease, Metabolic Or Endocrinology, Infectious Disease, Oncology, Genitourinary, Ophthalmology, Other Indications) – Market Size, Trends, Strategies, and Forecast to 2035
Virtual Clinical Trials Market Overview
• Virtual Clinical Trials market size has reached to $12.95 billion in 2025 • Expected to grow to $21.58 billion in 2030 at a compound annual growth rate (CAGR) of 10.7% • Growth Driver: Rising Number Of Clinical Trials Propel The Growth Of The Virtual Clinical Trials Market • Market Trend: Launch Of A No-Code Digital Platform Enhancing Efficiency In Clinical Trials • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Virtual Clinical Trials Market?
The virtual clinical trials refers to remote or decentralized clinical trials or studies that incorporate digital health technologies and enable remote participation outside the traditional brick-and-mortar clinical trial site using tablets, smartphone apps, or wearable sensors. Virtual clinical trials allow significant digital changes in clinical research methodology, resulting in a more patient-centric ecosystem. The main design types of virtual clinical trials include observational trials, interventional trials, and expanded access trials. The observational trials refer to virtual clinical trials solutions and services used in the observational trials. Observational studies aim to learn what occurs to people in various contexts. The study team observes the participants but does not influence the therapies they get. The virtual clinical trials are used in Phase I, Phase II, Phase III, and Phase IV, and they are used in clinical trials of drugs are devices used in the treatment of CNS, autoimmune/inflammation, cardiovascular diseases, metabolic/endocrinology, infectious diseases, oncology, genitourinary, ophthalmology, and others.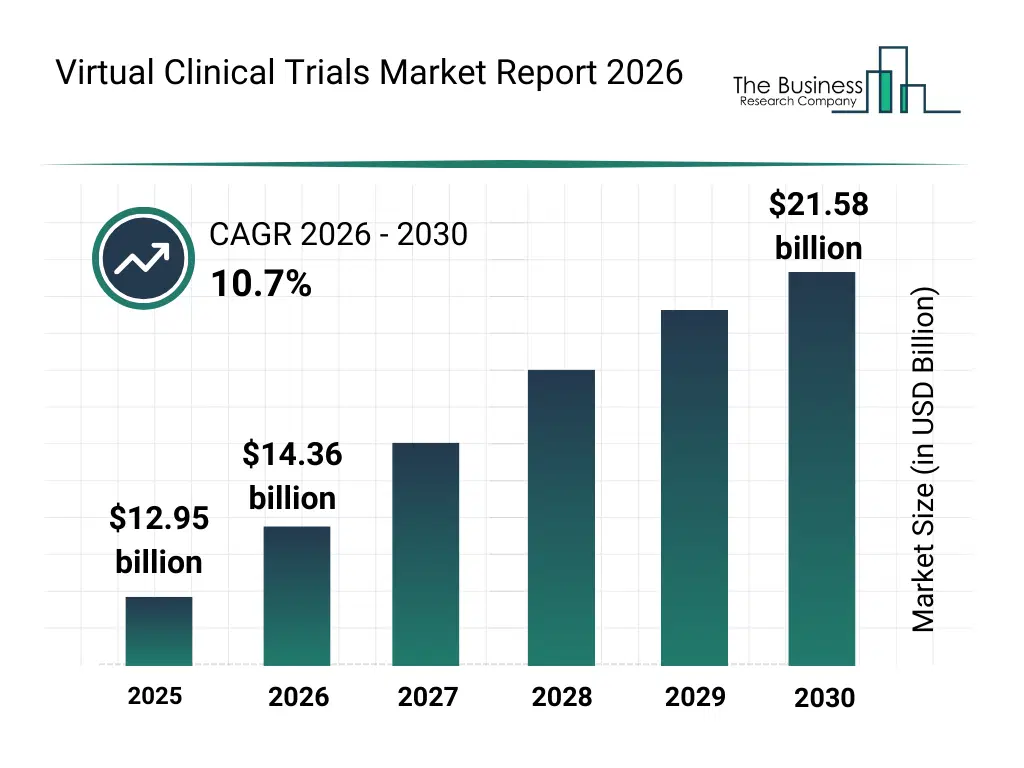
What Is The Virtual Clinical Trials Market Size and Share 2026?
The virtual clinical trials market size has grown rapidly in recent years. It will grow from $12.95 billion in 2025 to $14.36 billion in 2026 at a compound annual growth rate (CAGR) of 10.9%. The growth in the historic period can be attributed to rising need for faster drug development, challenges in traditional clinical trial participation, increasing technology adoption in healthcare research, regulatory support for decentralized models, rising patient engagement initiatives.What Is The Virtual Clinical Trials Market Growth Forecast?
The virtual clinical trials market size is expected to see rapid growth in the next few years. It will grow to $21.58 billion in 2030 at a compound annual growth rate (CAGR) of 10.7%. The growth in the forecast period can be attributed to growing demand for remote healthcare solutions, expansion of precision medicine trials, increasing use of digital patient monitoring tools, rising investment in digital clinical infrastructure, continuous innovation in decentralized trial technologies. Major trends in the forecast period include growing adoption of remote clinical trial participation, increasing use of wearables and connected health devices, rising shift toward patient centric clinical research models, growing utilization of digital platforms for clinical monitoring, increasing focus on reducing site based clinical dependencies.Global Virtual Clinical Trials Market Segmentation
1) By Design: Observational Trials, Interventional Trials, Expanded Access Trials 2) By Phases: Phase II, Phase III, Phase IV 3) By Indication: CNS, Autoimmune Or Inflammation, Cardiovascular Disease, Metabolic Or Endocrinology, Infectious Disease, Oncology, Genitourinary, Ophthalmology, Other Indications Subsegments: 1) By Observational Trials: Prospective Cohort Studies, Retrospective Studies, Case-Control Studies 2) By Interventional Trials: Randomized Controlled Trials (RCTs), Non-Randomized Trials, Adaptive Trials 3) By Expanded Access Trials: Single-Patient Expanded Access, Multi-Patient Expanded Access, Compassionate Use ProgramsWhat Is The Driver Of The Virtual Clinical Trials Market?
The increasing number of clinical trials is expected to propel the growth of the virtual clinical trials market going forward. Clinical trials are research studies conducted on human participants to evaluate the safety, efficacy, and potential benefits of new medical treatments, interventions, or drugs. The rise in clinical trials has paved the way for the growth and development of virtual clinical trials, offering a more efficient, cost-effective, and patient-friendly approach to conducting clinical research. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based organization, highlighted that the number of pharmaceutical industry trials launched in the UK increased for the second consecutive year, rising from 411 in 2022 to 426 in 2023. Therefore, an increasing number of clinical trials will drive the growth of the virtual clinical trials industry during forecast period.Key Players In The Global Virtual Clinical Trials Market
Major companies operating in the virtual clinical trials market report are ICON plc, Parexel International Pvt. Ltd., IQVIA Holdings Inc., Covance Research Products Inc., Pharmaceutical Research Associates Inc., Medidata Solution Inc., Oracle Corporation, Clario Tech Ltd., Medable Inc., Science 37 Holdings Inc., THREAD Research, Clinical Ink Inc., Veeva Systems Inc., Clinerion Ltd., CRF Health Group Limited, Crio Inc., Deep Lens Inc., Florence Healthcare Inc., goBalto Inc., Medrio Inc., Mint Medical Ltd., Mytrus Inc., OpenClinica LLC, PatientWing, RealTime-CTMS LLC, Reify Health, Sanguine Biosciences Inc., TriNetX Inc., VirTrial LLCGlobal Virtual Clinical Trials Market Trends and Insights
Major companies operating in the virtual clinical trials market are focusing on developing advanced solutions, such as no-code digital trial platforms, to streamline complex clinical workflows. A no-code digital solution refers to a simplified technology that enables users to design, configure, and deploy clinical trial components without deep technical expertise, thereby reducing development time, improving accuracy, and enhancing operational efficiency. For instance, in August 2024, Medable Inc., a US-based clinical trial technology company, launched Medable Studio into its digital trial platform. This innovation replaces traditional manual and service-heavy configuration processes by allowing sponsors to build, translate, validate, and launch eCOA, eConsent, Televisit, and sensor-based workflows directly through an intuitive, point-and-click web interface. It delivers real-time visibility into study content, scheduling, translations, and user flows while enabling teams to easily customize protocol-fit designs without relying on lengthy professional service cycles. Integrated with Medable’s broader digital trial ecosystem, including remote data capture, patient engagement tools, and consent workflows, it offers a streamlined digital experience. The no-code platform accelerates study start-up, improves operational control, and helps reduce trial timelines and costs.What Are Latest Mergers And Acquisitions In The Virtual Clinical Trials Market?
In February 2024, PCM Trials, a US-based home health care service, acquired EmVenio Research Inc. for an undisclosed amount. Through this acquisition, PCM Trials aims to enhance its and EmVenio's capabilities in recruiting and retaining diverse populations for clinical research, crucial for regulatory approval. This partnership provides PCM Trials access to EmVenio's site network focused on underserved communities, facilitating patient-centric clinical trials and improving retention and diversity. EmVenio Research Inc. is a US-based company focused on enabling virtual clinical trials through advanced technology and data analytics for more efficient research processes.Regional Outlook
North America was the largest region in the virtual clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Virtual Clinical Trials Market?
The virtual clinical trials market includes revenues earned by entities by providing services such as pilot studies and feasibility studies, prevention trials, screening trials, treatment trials, multi-arm multi-stage (mams) trials, cohort studies, case control studies, and cross-sectional studies. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Virtual Clinical Trials Market Report 2026?
The virtual clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the virtual clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Virtual Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $14.36 billion |
| Revenue Forecast In 2035 | $21.58 billion |
| Growth Rate | CAGR of 10.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Design, Phases, Indication |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | ICON plc, Parexel International Pvt. Ltd., IQVIA Holdings Inc., Covance Research Products Inc., Pharmaceutical Research Associates Inc., Medidata Solution Inc., Oracle Corporation, Clario Tech Ltd., Medable Inc., Science 37 Holdings Inc., THREAD Research, Clinical Ink Inc., Veeva Systems Inc., Clinerion Ltd., CRF Health Group Limited, Crio Inc., Deep Lens Inc., Florence Healthcare Inc., goBalto Inc., Medrio Inc., Mint Medical Ltd., Mytrus Inc., OpenClinica LLC, PatientWing, RealTime-CTMS LLC, Reify Health, Sanguine Biosciences Inc., TriNetX Inc., VirTrial LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
