
Adeno-Associated Viral Vectors Market Report 2026
Global Outlook – By Type Of Therapy (Gene Augmentation, Immunotherapy, Other Type Of Therapies), By Type Of Gene Delivery Method Used (Ex Vivo, In Vivo), By Target Therapeutic Area (Genetic Disorders, Hematological Disorders, Infectious Diseases, Metabolic Disorders, Ophthalmic Disorders, Muscle Disorders, Neurological Disorders, Other Target Therapeutic Areas ), By Scale Of Operation (Preclinical, Clinical, Commercial), By Application Area (Gene Therapy, Cell Therapy, Vaccines) - Market Size, Trends, And Global Forecast 2026-2035
Adeno-Associated Viral Vectors Market Overview
• Adeno-Associated Viral Vectors market size has reached to $3.17 billion in 2025 • Expected to grow to $6.65 billion in 2030 at a compound annual growth rate (CAGR) of 15.6% • Growth Driver: Rising Prevalence Of Genetic Disorders Fuels Growth In The Adeno-Associated Viral Vectors Market • Market Trend: Advancements In Off-the-Shelf Rep/Cap Plasmids • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Adeno-Associated Viral Vectors Market?
Adeno-associated viral vectors (AAV) are small, non-pathogenic viruses commonly used in gene therapy. They are engineered to deliver therapeutic genes into target cells, offering potential treatments for genetic disorders, cancers, and other diseases. AAV's safety profile and ability to efficiently infect both dividing and non-dividing cells make them valuable tools in biomedical research and clinical applications. The main type of adeno-associated viral vectors therapy are gene augmentation, immunotherapy, and others. Gene augmentation is a technique where a functional gene is introduced to replace or supplement a defective or missing gene in a patient's cells. The type of gene delivery method are ex vivo and in vivo, which works on target therapeutic areas such as genetic disorders, hematological disorders, infectious diseases, metabolic disorders, ophthalmic disorders, muscle disorders, neurological disorders, and others. The scale of operation includes preclinical, clinical, and commercial, applied for gene therapy, cell therapy, and vaccines.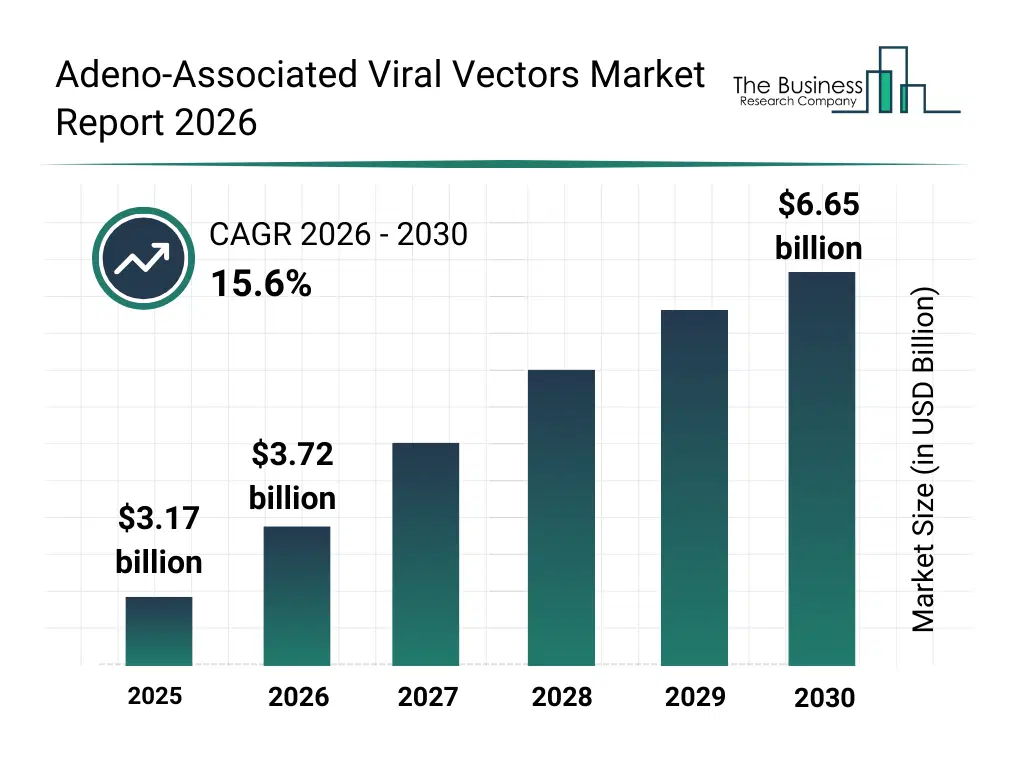
What Is The Adeno-Associated Viral Vectors Market Size and Share 2026?
The adeno-associated viral vectors market size has grown rapidly in recent years. It will grow from $3.17 billion in 2025 to $3.72 billion in 2026 at a compound annual growth rate (CAGR) of 17.6%. The growth in the historic period can be attributed to early development of aav vector platforms, limited clinical adoption in rare diseases, initial regulatory guidelines for gene therapy, rising research in genetic disorder treatment, increasing funding for preclinical studies.What Is The Adeno-Associated Viral Vectors Market Growth Forecast?
The adeno-associated viral vectors market size is expected to see rapid growth in the next few years. It will grow to $6.65 billion in 2030 at a compound annual growth rate (CAGR) of 15.6%. The growth in the forecast period can be attributed to advancements in capsid engineering and vector design, growth in commercial gene therapy launches, increasing demand for personalized medicine, expansion of global clinical trials, rising collaborations between biotech and pharma companies. Major trends in the forecast period include expansion of aav-based gene therapies for rare genetic disorders, increasing investment in in vivo and ex vivo gene delivery methods, growing adoption of aav vectors in immunotherapy applications, rising focus on clinical and commercial-scale gene therapy programs, enhanced regulatory oversight and standardization of gene therapy products.Global Adeno-Associated Viral Vectors Market Segmentation
1) By Type Of Therapy: Gene Augmentation, Immunotherapy, Other Type Of Therapies 2) By Type Of Gene Delivery Method Used: Ex Vivo, In Vivo 3) By Target Therapeutic Area: Genetic Disorders, Hematological Disorders, Infectious Diseases, Metabolic Disorders, Ophthalmic Disorders, Muscle Disorders, Neurological Disorders, Other Target Therapeutic Areas 4) By Scale Of Operation: Preclinical, Clinical, Commercial 5) By Application Area: Gene Therapy, Cell Therapy, Vaccines Subsegments: 1) By Gene Augmentation: Inherited Genetic Disorders, Muscular Dystrophy, Cystic Fibrosis, Hemophilia 2) By Immunotherapy: Cancer Immunotherapy, Viral Infections Immunotherapy 3) By Other Types Of Therapies: Gene Editing, RNA TherapyWhat Is The Driver Of The Adeno-Associated Viral Vectors Market?
The increasing prevalence of genetic disorders is expected to propel the growth of the adeno-associated viral vectors market going forward. Genetic disorders are conditions caused by abnormalities in an individual's DNA, leading to physical or developmental abnormalities. The improved diagnostic techniques, greater awareness, and higher reproductive age, along with environmental factors and genetic drift are leading to the rise in the prevalence of genetic disorders. Adeno-associated viral vectors are utilized in gene therapy for genetic disorders, delivering corrective genes into target cells to potentially treat conditions like muscular dystrophy or cystic fibrosis. For instance, in February 2023, according to the World Health Organization (WHO), a Switzerland-based specialized agency of the United Nations responsible for international public health, congenital diseases caused an estimated 240,000 baby deaths globally within 28 days of birth each year. An additional 170,000 children between the ages of 1 month and 5 years die as a result of congenital diseases. Therefore, the increasing prevalence of genetic disorders is driving the adeno-associated viral vectors industry.Key Players In The Global Adeno-Associated Viral Vectors Market
Major companies operating in the adeno-associated viral vectors market are Pfizer Inc., Astellas Pharma, Biogen Inc., Charles River Laboratories International Inc., BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., PTC Therapeutics, Ultragenyx Pharmaceutical, Amicus Therapeutics Inc., Oxford Biomedica, Asklepios BioPharmaceutical Inc., uniQure biopharma B.V., Spark Therapeutics Inc., Akouos Inc., Adverum Biotechnologies Inc., Passage Bio Inc., AVROBIO Inc., MeiraGTx Holdings plc, GenSight Biologics S.A., Freeline Therapeutics, Aspa Therapeutics Inc., Adrenas Therapeutics Inc., 4D Molecular Therapeutics, Abeona Therapeutics Inc., Neurophth TherapeuticsGlobal Adeno-Associated Viral Vectors Market Trends and Insights
Major companies operating in the adeno-associated viral vectors market are focusing on offering off-the-shelf availability of replication-capsid plasmid to gain a competitive edge in the market. Rep/Cap plasmids, commonly utilized in gene therapy for adeno-associated virus (AAV) vector production, are readily available from numerous commercial suppliers catering to molecular biology research needs. For instance, in January 2024, Charles River Laboratories International Inc., a US-based pharmaceutical company, introduced off-the-shelf replication-capsid plasmid range streamlines AAV-based gene therapy initiatives. This expansion of their product portfolio complements existing lentiviral packaging and AAV Helper plasmid offerings, cutting manufacturing efforts by as much as 66%. These ready-to-use plasmids undergo batch production with meticulous documentation, adhering to CMC guidelines and accompanied by Certification of Analysis (COA) to facilitate IND and Clinical Trial Application (CTA) submissions.What Are Latest Mergers And Acquisitions In The Adeno-Associated Viral Vectors Market?
In May 2024, Merck KGaA, a Germany-based company that offers pharmaceuticals, biotechnology, and chemical materials acquired Mirus Bio LLC for $600 million. With this acquisition, Merck KGaA aims to enhance its capabilities in the life sciences sector by integrating Mirus Bio LLC’s advanced technologies for RNA-based research and therapeutic applications, thereby strengthening its position in the growing market for genetic engineering and biotechnology solutions, and furthering its commitment to developing innovative biopharmaceutical products. Mirus Bio LLC is a US-based company that offers adeno-associated viral (AAV) vectors and gene delivery technologies.Regional Outlook
North America was the largest region in the adeno-associated viral vectors market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Adeno-Associated Viral Vectors Market?
The adeno-associated viral vectors market consists of sales of recombinant adeno-associated viral vectors (AAV), serotype-specific adeno-associated viral vectors (AAV), and custom adeno-associated viral vectors (AAV). Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Adeno-Associated Viral Vectors Market Report 2026?
The adeno-associated viral vectors market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the adeno-associated viral vectors industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Adeno-Associated Viral Vectors Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.72 billion |
| Revenue Forecast In 2035 | $6.65 billion |
| Growth Rate | CAGR of 17.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Therapy, Type Of Gene Delivery Method Used, Target Therapeutic Area, Scale Of Operation, Application Area |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Astellas Pharma, Biogen Inc., Charles River Laboratories International Inc., BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., PTC Therapeutics, Ultragenyx Pharmaceutical, Amicus Therapeutics Inc., Oxford Biomedica, Asklepios BioPharmaceutical Inc., uniQure biopharma B.V., Spark Therapeutics Inc., Akouos Inc., Adverum Biotechnologies Inc., Passage Bio Inc., AVROBIO Inc., MeiraGTx Holdings plc, GenSight Biologics S.A., Freeline Therapeutics, Aspa Therapeutics Inc., Adrenas Therapeutics Inc., 4D Molecular Therapeutics, Abeona Therapeutics Inc., Neurophth Therapeutics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
