
Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Report 2026
Global Outlook – By Type of Therapy (Gene Augmentation, Immunotherapy, Other Type of Therapy), By Type of Gene Delivery Method Used (Ex Vivo, In Vivo), By Scale of Operation (Preclinical, Clinical, Commercial), By Target Therapeutic Area (Genetic Disorders, Hematological Disorders, Infectious Diseases, Metabolic Disorders, Ophthalmic Disorders, Muscle Disorders, Neurological Disorders, Other Target Therapeutic Area) – Market Size, Trends, Strategies, and Forecast to 2035
Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Overview
• Adeno-Associated Virus (AVV) Vectors In Gene Therapy market size has reached to $3.16 billion in 2025 • Expected to grow to $6.99 billion in 2030 at a compound annual growth rate (CAGR) of 17.2% • Growth Driver: Surge In Gene-based Therapies Fueling The Growth Of The Market Due To Rising Demand For Precise And Long-term Genetic Treatments • Market Trend: Strengthening Manufacturing Consistency In Advanced Therapies • North America was the largest region in 2025.What Is Covered Under Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market?
Adeno-associated virus (AAV) vectors in gene therapy are advanced delivery tools that transport genetic material into cells to treat various genetic disorders. Their primary goal is to enable targeted, long-lasting therapeutic effects by correcting or modifying faulty genes. AAV vectors are valued for their safety, low immunogenicity, and ability to deliver genes to both dividing and non-dividing cells, supporting the development of precise and durable gene therapies that advance personalized medicine and transform the treatment of rare and inherited diseases. The main types of adeno-associated virus (AVV) vectors are gene augmentation, immunotherapy, and other kind of therapy. Gene augmentation refers to the therapeutic introduction of a functional gene into a cell to replace or compensate for a defective or missing one. This can be performed using different gene delivery methods, such as ex vivo, and in vivo. This is conducted at various scales of operation, including preclinical, clinical, and commercial. The target therapeutic areas include genetic disorders, hematological disorders, infectious diseases, metabolic disorders, ophthalmic disorders, muscle disorders, and neurological conditions.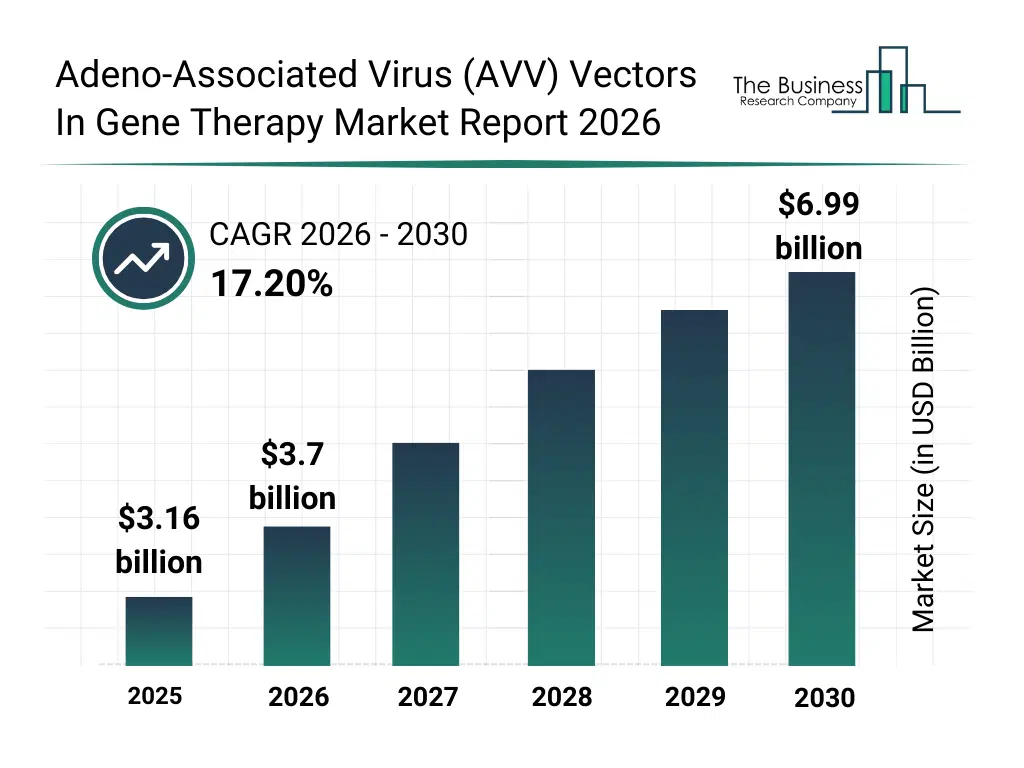
What Is The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Size and Share 2026?
The adeno-associated virus (avv) vectors in gene therapy market size has grown rapidly in recent years. It will grow from $3.16 billion in 2025 to $3.7 billion in 2026 at a compound annual growth rate (CAGR) of 17.3%. The growth in the historic period can be attributed to advancements in viral vector safety, rare genetic disorder research, clinical gene therapy success, regulatory approvals, academic research investments.What Is The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Growth Forecast?
The adeno-associated virus (avv) vectors in gene therapy market size is expected to see rapid growth in the next few years. It will grow to $6.99 billion in 2030 at a compound annual growth rate (CAGR) of 17.2%. The growth in the forecast period can be attributed to commercial gene therapy launches, scalable vector production demand, precision medicine expansion, orphan drug incentives, global clinical trial growth. Major trends in the forecast period include expansion of gene augmentation therapies, rising demand for rare disease gene treatments, growth of in vivo gene delivery, increased commercial-scale vector manufacturing, focus on long-term therapeutic durability.Global Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Segmentation
1) By Type of Therapy: Gene Augmentation, Immunotherapy, Other Type of Therapy 2) By Type of Gene Delivery Method Used: Ex Vivo, In Vivo 3) By Scale of Operation: Preclinical, Clinical, Commercial 4) By Target Therapeutic Area: Genetic Disorders, Hematological Disorders, Infectious Diseases, Metabolic Disorders, Ophthalmic Disorders, Muscle Disorders, Neurological Disorders, Other Target Therapeutic Area Subsegments: 1) By Gene Augmentation: Monogenic Disorders, Neurological Disorders, Muscular Disorders, Ophthalmological Disorders, Metabolic Disorders 2) By Immunotherapy: Oncology, Infectious Diseases, Autoimmune Disorders, Vaccine Development, T-Cell Engineering 3) By Other Type of Therapy: Gene Editing Support, RNA Interference, Neuroprotection and Neuroregeneration, Anti-Inflammatory Applications, Regenerative MedicineWhat Is The Driver Of The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market?
The increasing focus on gene-based therapies is expected to propel the growth of the adeno-associated virus (AAV) vectors in the gene therapy market going forward. Gene-based therapies are treatments that modify or manipulate genes to prevent, treat, or cure diseases at the molecular level. Advancements in genetic research drive the increasing focus on creating gene-based therapies, as they enhance the precision and effectiveness of gene editing techniques. Adeno-associated virus (AAV) vectors in gene therapy help create gene-based therapies by delivering therapeutic genes safely and efficiently into target cells, owing to their low immunogenicity and ability to provide long-term gene expression. For instance, in 2023, according to IQVIA, a US-based provider of advanced analytics and technology solutions for the life sciences industry, global spending on cell and gene therapies reached $5.9 billion, representing a 38% increase from 2022. Therefore, increasing focus on creating gene-based therapies is driving the growth of the adeno-associated virus (AAV) vectors market.Key Players In The Global Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market
Major companies operating in the adeno-associated virus (avv) vectors in gene therapy market are F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG, Astellas Gene Therapies Inc., Biogen Inc., Sarepta Therapeutics Inc., Oxford BioMedica plc, Rocket Pharmaceuticals Inc., Aldevron LLC, REGENXBIO Inc., uniQure N.V., Passage Bio Inc., Voyager Therapeutics Inc., Dyno Therapeutics Inc., MeiraGTx Holdings plc, Abeona Therapeutics Inc., 4D Molecular Therapeutics Inc., GenSight Biologics S.A., Taysha Gene Therapies Inc., LogicBio Therapeutics Inc.Global Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Trends and Insights
Major companies operating in the adeno-associated virus (AAV) vectors in the gene therapy market are focusing on advanced innovation, such as tailored vector diversity for assay versatility to enhance target tissue specificity, improve therapeutic efficacy, and accelerate the development of customized gene therapies across various indications. Tailored vector diversity for assay versatility refers to using multiple AAV serotypes or constructs to support various analytical tests and therapeutic applications. For instance, in May 2024, Charles River Laboratories, a US-based pharmaceutical company, introduced new reference materials for adeno-associated virus (AAV) and lentiviral vectors (LVV). These materials are intended to support the growing needs of cell and gene therapy (CGT) development. The portfolio facilitates a smoother transition from early-stage research to GMP-grade production. It helps standardize processes and improve consistency in viral vector manufacturing. This launch addresses a key challenge in scaling CGT programs toward clinical and commercial readiness.What Are Latest Mergers And Acquisitions In The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market?
In March 2023, Ginkgo Bioworks, a US-based biotech company, acquired StrideBio's AAV capsid discovery and engineering platform assets for an undisclosed amount. With this acquisition, Ginkgo Bioworks aims to enhance its end-to-end research and development capabilities in gene therapy, particularly developing novel adeno-associated virus (AVV) capsids for improved gene delivery. StrideBio is a US-based biotechnology company focused on developing engineered AAV capsids for gene therapy applications.Regional Insights
North America was the largest region in the adeno-associated virus (AVV) vectors in gene therapy market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market?
The adeno-associated virus (AAV) vectors in gene therapy market consists of revenues earned by entities by providing services such as gene delivery solutions, targeted therapeutic development, long-term gene expression, and treatment of genetic disorders through safe and efficient viral vector technologies. The market value includes the value of related goods sold by the service provider or included within the service offering. The adeno-associated virus (AAV) vectors in gene therapy market includes sales of viral vector delivery systems used for transporting genetic material into patient cells. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Report 2026?
The adeno-associated virus (avv) vectors in gene therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the adeno-associated virus (avv) vectors in gene therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.7 billion |
| Revenue Forecast In 2035 | $6.99 billion |
| Growth Rate | CAGR of 17.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type of Therapy, Type of Gene Delivery Method Used, Scale of Operation, Target Therapeutic Area |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG, Astellas Gene Therapies Inc., Biogen Inc., Sarepta Therapeutics Inc., Oxford BioMedica plc, Rocket Pharmaceuticals Inc., Aldevron LLC, REGENXBIO Inc., uniQure N.V., Passage Bio Inc., Voyager Therapeutics Inc., Dyno Therapeutics Inc., MeiraGTx Holdings plc, Abeona Therapeutics Inc., 4D Molecular Therapeutics Inc., GenSight Biologics S.A., Taysha Gene Therapies Inc., LogicBio Therapeutics Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
