
Balloon Ablation Catheters Market Report 2026
Global Outlook – By Product Type (Single-Use Balloon Ablation Catheters, Reusable Balloon Ablation Catheters), By Application (Cardiac Arrhythmia, Atrial Fibrillation, Ventricular Tachycardia, Other Applications), By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Balloon Ablation Catheters Market Overview
• Balloon Ablation Catheters market size has reached to $1.28 billion in 2025 • Expected to grow to $2.04 billion in 2030 at a compound annual growth rate (CAGR) of 9.7% • Growth Driver: Escalating Incidence Of Cardiac Arrhythmias Driving The Market Growth Due To Rising Cases And Improve Diagnosis • Market Trend: Innovative Catheter Technology Advances Precision And Safety In Cardiac Ablation • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Balloon Ablation Catheters Market?
Balloon ablation catheters are specialized medical devices featuring an inflatable balloon integrated with energy delivery mechanisms, such as radiofrequency, cryo, laser, or thermal elements, at the catheter tip. They are designed to achieve precise tissue ablation by inflating the balloon to occlude targeted areas while applying energy to create circumferential lesions that disrupt abnormal electrical pathways. The main product types of balloon ablation catheters include single-use balloon ablation catheters and reusable balloon ablation catheters. Single-use balloon ablation catheters refer to devices designed to deliver controlled energy to cardiac tissue to treat abnormal heart rhythms in a single procedure. They find application in cardiac arrhythmia, atrial fibrillation, ventricular tachycardia, and other applications, serving end users including hospitals, ambulatory surgical centers, specialty clinics, and other end-users.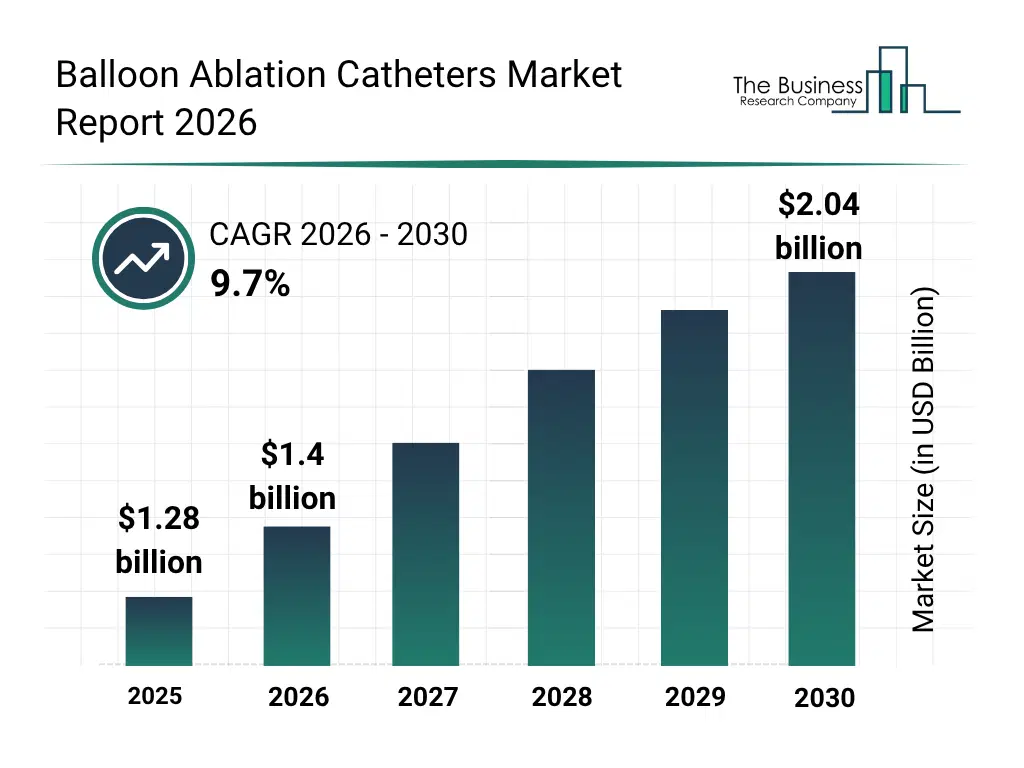
What Is The Balloon Ablation Catheters Market Size and Share 2026?
The balloon ablation catheters market size has grown strongly in recent years. It will grow from $1.28 billion in 2025 to $1.4 billion in 2026 at a compound annual growth rate (CAGR) of 9.5%. The growth in the historic period can be attributed to increasing prevalence of cardiac arrhythmias, adoption of minimally invasive ablation procedures, advancements in catheter technologies, rising cardiovascular disease awareness, growth of hospital cardiac care units.What Is The Balloon Ablation Catheters Market Growth Forecast?
The balloon ablation catheters market size is expected to see strong growth in the next few years. It will grow to $2.04 billion in 2030 at a compound annual growth rate (CAGR) of 9.7%. The growth in the forecast period can be attributed to growing demand for atrial fibrillation treatments, increasing adoption of advanced energy delivery mechanisms, rising use of catheter-based therapies in specialty clinics, expansion of ambulatory surgical centers, increasing integration with imaging and monitoring systems. Major trends in the forecast period include increasing adoption of single-use balloon ablation catheters, rising demand for radiofrequency and cryo balloon catheters, growing focus on minimally invasive cardiac procedures, expansion of reusable balloon ablation catheter applications, rising integration of catheter systems with real-time imaging.Global Balloon Ablation Catheters Market Segmentation
1) By Product Type: Single-Use Balloon Ablation Catheters, Reusable Balloon Ablation Catheters 2) By Application: Cardiac Arrhythmia, Atrial Fibrillation, Ventricular Tachycardia, Other Applications 3) By End User: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End-Users Subsegments: 1) By Single-Use Balloon Ablation Catheters: Cryo Balloon Catheter, Radiofrequency Balloon Catheter, Laser Balloon Catheter, Microwave Balloon Catheter 2) By Reusable Balloon Ablation Catheters: Cryo Balloon Catheter, Radiofrequency Balloon Catheter, Laser Balloon Catheter, Microwave Balloon CatheterWhat Is The Driver Of The Balloon Ablation Catheters Market?
The escalating incidence of cardiac arrhythmias is expected to propel the growth of the balloon ablation catheters market going forward. Cardiac arrhythmias are conditions in which the heart beats irregularly, too fast, or too slow due to electrical conduction disturbances in the heart. The incidence of cardiac arrhythmias is rising due to the aging population, as older adults are more prone to heart rhythm disorders. Balloon ablation catheters help manage cardiac arrhythmias by precisely delivering energy to targeted areas of the heart tissue to disrupt abnormal electrical pathways, restore normal heart rhythm, and reduce the risk of complications associated with irregular heartbeats. For instance, March 2024, according to the Centers for Disease Control and Prevention (CDC), a US-based government agency, an estimated 12.1 million people in the United States are projected to have atrial fibrillation (AFib) by 2050. Therefore, the escalating incidence of cardiac arrhythmias is driving the growth of the balloon ablation catheters industry.Key Players In The Global Balloon Ablation Catheters Market
Major companies operating in the balloon ablation catheters market are Johnson & Johnson, Abbott Laboratories, Medtronic plc, Toray Industries Inc., Boston Scientific Corporation, Cook Medical LLC, Biotronik SE & Co. KG, Merit Medical Systems Inc., CONMED Corporation, Lepu Medical Technology (Beijing) Co. Ltd., MicroPort Scientific Corporation, Avanos Medical Inc., Japan Lifeline Co. Ltd., AngioDynamics Inc., Imricor Medical Systems Inc., Kardium Inc., B. Braun SE, ERBE Elektromedizin GmbH, CathVision A/S, Osypka AG, CathRx Ltd., Pulse Biosciences Inc., CardioNXT Inc., Shanghai MicroPort EP MedTech Co. Ltd., Aurigen Medical Inc.Global Balloon Ablation Catheters Market Trends and Insights
Major companies operating in the balloon ablation catheters market are focusing on developing innovative products such as, next-generation pulsed field ablation catheters to enhance safety, reduce procedure time, and improve patient outcomes. Next-generation pulsed field ablation catheters are advanced balloon-based devices that deliver precise electrical pulses to treat abnormal heart rhythms, helping reduce procedure time, minimize tissue damage, and improve patient recovery. For instance, in December 2025, Abbott Laboratories, a US-based medical device company, received U.S. Food and Drug Administration approval for Volt Pulsed Field Ablation System, an innovative balloon‑in‑basket pulsed field ablation (PFA) catheter integrated with the EnSite X electroanatomical mapping system. The device enables clinicians to map, pace, and ablate cardiac tissue using a single catheter, delivering precise, non-thermal energy to create targeted lesions while minimizing damage to surrounding tissue. Its balloon‑in‑basket design, featuring multiple active splines, ensures optimal tissue contact and uniform energy delivery, enhancing procedural efficiency and safety compared with traditional thermal ablation catheters. Designed for treating both paroxysmal and persistent atrial fibrillation, the system reduces procedure times and supports faster patient recovery.What Are Latest Mergers And Acquisitions In The Balloon Ablation Catheters Market?
In October 2025, Merit Medical Systems Inc., a US-based healthcare technology company, acquired the C2 CryoBalloon device and related technology from PENTAX Medical for an undisclosed amount. With this acquisition, Merit Medical aims to expand its endoscopy product portfolio and strengthen its presence in the gastroenterology market by offering minimally invasive treatment options for conditions such as Barrett’s esophagus and other gastrointestinal disorders. PENTAX Medical is a Japan-based healthcare technology company that specializes in providing the C2 cryoballoon ablation system.Regional Insights
North America was the largest region in the balloon ablation catheters market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Balloon Ablation Catheters Market?
The balloon ablation catheters market consists of sales of cryoballoon ablation catheters, radiofrequency balloon ablation catheters, laser balloon ablation catheters, single-use balloon catheters, and multi-electrode balloon catheters. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Balloon Ablation Catheters Market Report 2026?
The balloon ablation catheters market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the balloon ablation catheters industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Balloon Ablation Catheters Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.4 billion |
| Revenue Forecast In 2035 | $2.04 billion |
| Growth Rate | CAGR of 9.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Johnson & Johnson, Abbott Laboratories, Medtronic plc, Toray Industries Inc., Boston Scientific Corporation, Cook Medical LLC, Biotronik SE & Co. KG, Merit Medical Systems Inc., CONMED Corporation, Lepu Medical Technology (Beijing) Co. Ltd., MicroPort Scientific Corporation, Avanos Medical Inc., Japan Lifeline Co. Ltd., AngioDynamics Inc., Imricor Medical Systems Inc., Kardium Inc., B. Braun SE, ERBE Elektromedizin GmbH, CathVision A/S, Osypka AG, CathRx Ltd., Pulse Biosciences Inc., CardioNXT Inc., Shanghai MicroPort EP MedTech Co. Ltd., Aurigen Medical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
