
Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Report 2026
Global Outlook – By Technology (Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Digital Polymerase Chain Reaction (dPCR)), By Source (Plasma, Serum, Urine), By Application (Oncology, Non-Invasive Prenatal Testing, Transplantation, Other Applications), By End-User (Hospitals, Diagnostic Laboratories, Academic And Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Overview
• Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection market size has reached to $2.58 billion in 2025 • Expected to grow to $5.64 billion in 2030 at a compound annual growth rate (CAGR) of 16.9% • Growth Driver: Increasing Demand For Personalized Medicine Fueling The Growth Of The Due To Advancements In Genomics And Tailored Cancer Treatments • Market Trend: Advanced Kits Enhance Accuracy And Sensitivity In Cancer Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market?
Cell free tumor deoxyribonucleic acid (DNA) detection is a non-invasive diagnostic approach that identifies and analyzes small DNA fragments released into the bloodstream by cancer cells. It provides real-time insights into genetic mutations, tumor burden, and disease progression without requiring tissue biopsies. The primary purpose is to enable early cancer detection, monitor treatment response, and track minimal residual disease or relapse. The main technologies used in cell free tumor deoxyribonucleic acid (DNA) detection are polymerase chain reaction (PCR), next-generation sequencing (NGS), and digital polymerase chain reaction (dPCR). Polymerase chain reaction (PCR) refers to a technique that rapidly amplifies specific DNA segments, making it possible to detect even small amounts of genetic material, including tumor DNA.The various source types include plasma, serum, and urine and is applied in oncology, non-invasive prenatal testing, transplantation, and others, serving end-users such as hospitals, diagnostic laboratories, academic and research institutes, and others.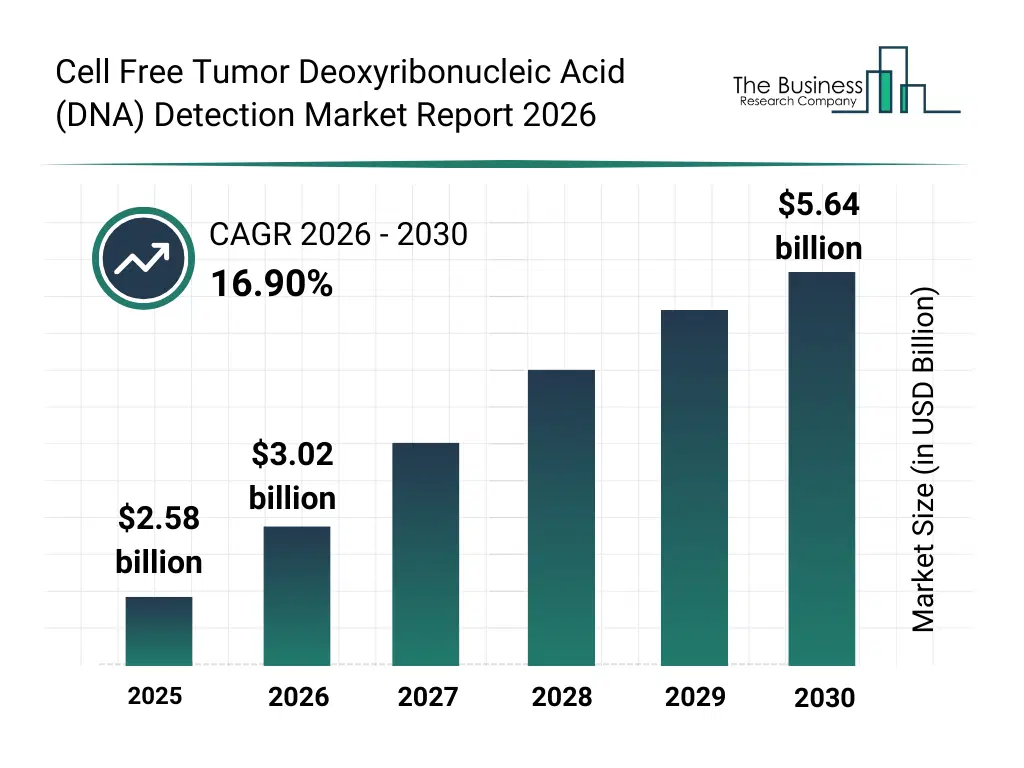
What Is The Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Size and Share 2026?
The cell free tumor deoxyribonucleic acid (dna) detection market size has grown rapidly in recent years. It will grow from $2.58 billion in 2025 to $3.02 billion in 2026 at a compound annual growth rate (CAGR) of 17.2%. The growth in the historic period can be attributed to growing cancer prevalence, advancements in molecular diagnostics, rising demand for non-invasive testing, adoption of PCR and NGS techniques, increasing clinical research studies.What Is The Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Growth Forecast?
The cell free tumor deoxyribonucleic acid (dna) detection market size is expected to see rapid growth in the next few years. It will grow to $5.64 billion in 2030 at a compound annual growth rate (CAGR) of 16.9%. The growth in the forecast period can be attributed to expansion of precision medicine, technological innovations in ctDNA analysis, integration with AI-based diagnostics, rising healthcare investments in emerging economies, growth in early cancer detection programs. Major trends in the forecast period include liquid biopsy adoption, integration with genomic data platforms, real-time tumor monitoring, non-invasive cancer diagnostics, advanced ctdna detection technologies.Global Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Segmentation
1) By Technology: Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Digital Polymerase Chain Reaction (dPCR) 2) By Source: Plasma, Serum, Urine 3) By Application: Oncology, Non-Invasive Prenatal Testing, Transplantation, Other Applications 4) By End-User: Hospitals, Diagnostic Laboratories, Academic And Research Institutes, Other End-Users Subsegments: 1) By Polymerase Chain Reaction (PCR): Real-Time Polymerase Chain Reaction (qPCR), Multiplex Polymerase Chain Reaction (PCR), Allele-Specific Polymerase Chain Reaction (PCR), High-Resolution Melting (HRM) Analysis, Beaming Polymerase Chain Reaction (PCR) 2) By Next-Generation Sequencing (NGS): Whole Genome Sequencing (WGS), Whole Exome Sequencing (WES), Targeted Gene Panels, Ribonucleic Acid Sequencing (RNA-seq), Methylation Sequencing 3) By Digital Polymerase Chain Reaction (dPCR): Droplet Digital Polymerase Chain Reaction (ddPCR), Chip-Based Digital Polymerase Chain Reaction (PCR), Nanofluidic Digital Polymerase Chain Reaction (PCR)What Is The Driver Of The Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market?
The increasing demand for personalized medicine is expected to propel the growth of the cell free tumor deoxyribonucleic acid (DNA) detection market going forward. Personalized medicine refers to a medical approach that tailors treatment and prevention strategies to an individual’s unique genetic makeup, lifestyle, and environment. The increasing adoption of personalized medicine is primarily due to advancements in genomics, which enable precise identification of genetic variations and tailored treatments for individuals. Cell free tumor deoxyribonucleic acid (DNA) detection enhances personalized medicine by enabling non-invasive, real-time insights into a patient’s tumor genetics. It allows clinicians to tailor treatments based on individual mutation profiles, monitor therapy response, and detect disease recurrence early, improving patient outcomes and precision in cancer care. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the U.S. Food and Drug Administration (FDA) approved 26 new personalized medicines in 2023, a significant increase from the 12 approved in 2022. Therefore, the increasing demand for personalized medicine is driving the growth of the cell free tumor deoxyribonucleic acid (DNA) detection industry.Key Players In The Global Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market
Major companies operating in the cell free tumor deoxyribonucleic acid (dna) detection market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Labcorp Holdings Inc., Illumina Inc., Sysmex Inostics Inc., Bio-Rad Laboratories Inc., QIAGEN N.V., Natera Inc., Guardant Health Inc., Invitae Corporation, Foundation Medicine Inc, Adaptive Biotechnologies Corporation, Freenome Holdings Inc., Grail Inc., Biodesix Inc, Personal Genome Diagnostics Inc., Agena Bioscience Inc., Menarini Silicon Biosystems SpA, Exosome Diagnostics Inc., Lucence Health Inc.Global Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Trends and Insights
Major companies operating in the cell free tumor deoxyribonucleic acid (DNA) detection market are focusing on developing advanced products, such as library preparation kits, to enhance accuracy, sensitivity, and speed in cancer testing. Library preparation kits are reagent kits that prepare DNA or RNA samples for sequencing by fragmenting, tagging, and amplifying them for accurate analysis. For instance, in February 2024, Twist Bioscience Corporation, a US-based biotechnology company, launched the cfDNA Library Preparation Kit designed to enhance the sensitivity and precision of liquid biopsy applications. The kit enables high-conversion cfDNA library generation for next-generation sequencing on Illumina platforms, effectively addressing challenges posed by low-input and degraded cfDNA samples. This innovation is intended to improve the reliability and efficiency of liquid biopsy testing, supporting earlier and more accurate cancer detection as well as better monitoring of treatment response.What Are Latest Mergers And Acquisitions In The Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market?
In April 2023, Quest Diagnostics Incorporated, a US-based provider of diagnostic testing, information, and services, acquired Haystack Oncology Inc. for an undisclosed amount. With this acquisition, Quest Diagnostics aims to enhance its oncology portfolio by integrating Haystack’s sensitive liquid biopsy technology, which is designed to improve the accuracy of cancer detection and personalized treatment strategies. Haystack Oncology, Inc. is a US-based precision oncology company specializing in cell-free tumor DNA (cfDNA) detection and liquid biopsy diagnostics.Regional Outlook
North America was the largest region in the cell free tumor deoxyribonucleic acid (DNA) detection market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market?
The cell free tumor deoxyribonucleic acid (DNA) detection market consists of revenues earned by entities by providing services such as early cancer screening, liquid biopsy testing, genomic profiling, treatment response monitoring, and personalized therapy selection. The market value includes the value of related goods sold by the service provider or included within the service offering. The cell free tumor deoxyribonucleic acid (DNA) detection market also includes sales of liquid biopsy kits, next-generation sequencing (NGS) platforms, target enrichment kits, bioinformatics and data analysis software, microfluidics-based detection platforms, and automated liquid handling systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Cell Free Tumor Deoxyribonucleic Acid (DNA) Detection Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.02 billion |
| Revenue Forecast In 2035 | $5.64 billion |
| Growth Rate | CAGR of 17.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Source, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Labcorp Holdings Inc., Illumina Inc., Sysmex Inostics Inc., Bio-Rad Laboratories Inc., QIAGEN N.V., Natera Inc., Guardant Health Inc., Invitae Corporation, Foundation Medicine Inc, Adaptive Biotechnologies Corporation, Freenome Holdings Inc., Grail Inc., Biodesix Inc, Personal Genome Diagnostics Inc., Agena Bioscience Inc., Menarini Silicon Biosystems SpA, Exosome Diagnostics Inc., Lucence Health Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
