
Exon Skipping Therapies Market Report 2026
Global Outlook – By Product Type (Antisense Oligonucleotides, Synthetic Ribonucleic Acid Molecules, Exon-Targeted Gene Therapy Kits, Combination Therapy Formulations, Other Product Types), By Route Of Administration (Oral, Intravenous, Subcutaneous, Other Route Of Administration), By Distribution Channel (Direct Sales, Distributors, Online Pharmacies, Other Distribution Channel), By Application (Duchenne Muscular Dystrophy, Becker Muscular Dystrophy, Spinal Muscular Atrophy, Other Inherited Diseases, Other Applications), By End-User (Hospitals And Clinics, Research Institutes, Biopharmaceutical Companies, Specialty Genetic Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Exon Skipping Therapies Market Overview
• Exon Skipping Therapies market size has reached to $2.34 billion in 2025 • Expected to grow to $5.9 billion in 2030 at a compound annual growth rate (CAGR) of 20.4% • Growth Driver: Rising Prevalence Of Genetic Disorders Driving The Market Growth Due To Growing Need For Targeted Gene Therapies • Market Trend: Innovations In Targeted Therapies For Genetic Disorders Driving Enhance Treatment Outcomes And Patient Care • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Exon Skipping Therapies Market?
Exon skipping therapies are RNA-based genetic treatments that work by altering pre-mRNA splicing to bypass disease-causing exons, enabling cells to produce a shorter but partially functional protein. These therapies typically use antisense oligonucleotides (ASOs) that bind to specific sequences in the pre-mRNA, masking the mutated exon so it is excluded during RNA processing. The main product types of exon skipping therapies include antisense oligonucleotides, synthetic ribonucleic acid molecules, exon-targeted Gene Therapy kits, combination therapy formulations, and other product types. Antisense oligonucleotides refer to short, synthetic strands designed to bind specific ribonucleic acid sequences and modify gene expression to restore functional protein production. The routes of administration include oral, intravenous, subcutaneous, and other routes of administration and the distribution channels include direct sales, distributors, online pharmacies, and other distribution channels. The key applications of Duchenne muscular dystrophy, Becker muscular dystrophy, spinal muscular atrophy, other inherited diseases, and other applications, while the end-use includes hospitals and clinics, research institutes, biopharmaceutical companies, specialty genetic centers, and other end users.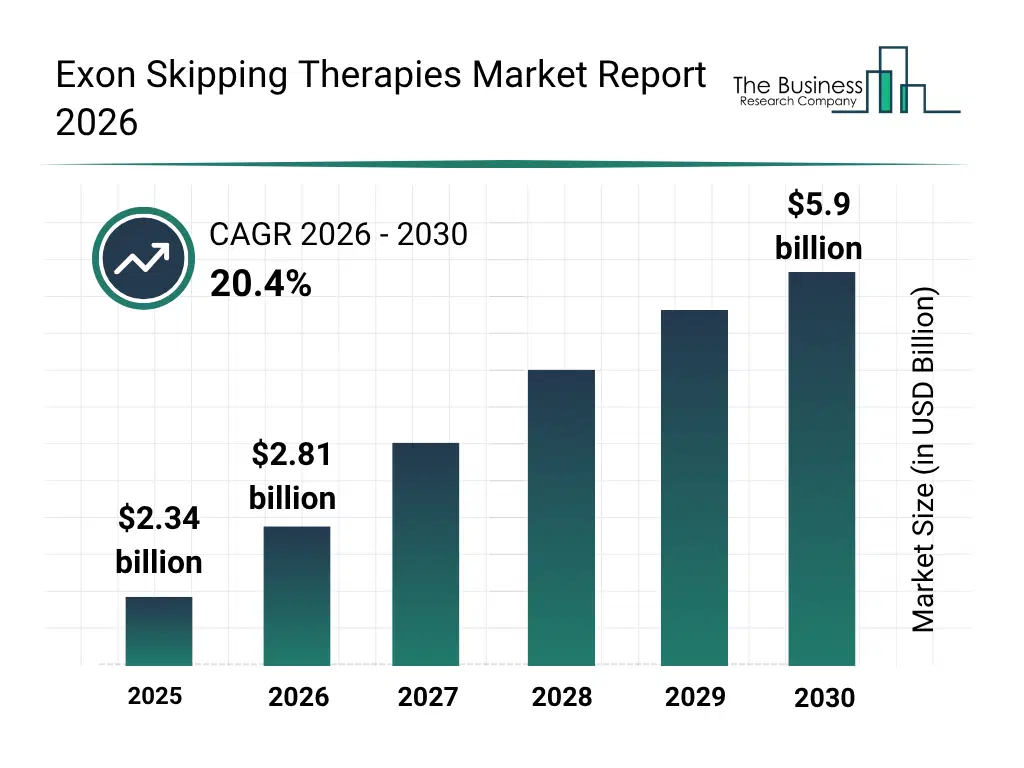
What Is The Exon Skipping Therapies Market Size and Share 2026?
The exon skipping therapies market size has grown exponentially in recent years. It will grow from $2.34 billion in 2025 to $2.81 billion in 2026 at a compound annual growth rate (CAGR) of 20.1%. The growth in the historic period can be attributed to increasing prevalence of duchenne muscular dystrophy, growing research in RNA splicing mechanisms, early regulatory approvals for antisense therapies, rising funding for rare disease research, expanding biotechnology partnerships.What Is The Exon Skipping Therapies Market Growth Forecast?
The exon skipping therapies market size is expected to see exponential growth in the next few years. It will grow to $5.9 billion in 2030 at a compound annual growth rate (CAGR) of 20.4%. The growth in the forecast period can be attributed to adoption of personalized genetic medicine, increasing pipeline of exon-targeted therapies, rising investment in advanced delivery systems, expanding approvals across inherited neuromuscular disorders, increasing collaboration between research institutes and biopharmaceutical companies. Major trends in the forecast period include advancements in antisense oligonucleotide chemistry, increasing focus on rare genetic disorders, expansion of combination genetic therapy approaches, growing investment in RNA-based therapeutics pipelines, rising clinical trials for exon-specific mutations.Global Exon Skipping Therapies Market Segmentation
1) By Product Type: Antisense Oligonucleotides, Synthetic Ribonucleic Acid Molecules, Exon-Targeted Gene Therapy Kits, Combination Therapy Formulations, Other Product Types 2) By Route Of Administration: Oral, Intravenous, Subcutaneous, Other Route Of Administration 3) By Distribution Channel: Direct Sales, Distributors, Online Pharmacies, Other Distribution Channel 4) By Application: Duchenne Muscular Dystrophy, Becker Muscular Dystrophy, Spinal Muscular Atrophy, Other Inherited Diseases, Other Applications 5) By End-User: Hospitals And Clinics, Research Institutes, Biopharmaceutical Companies, Specialty Genetic Centers, Other End Users Subsegments: 1) By Antisense Oligonucleotides: Phosphorodiamidate Morpholino Oligonucleotides, Peptide Conjugated Oligonucleotides, Gapmer Oligonucleotides, Steric Blocking Oligonucleotides, Splice Modulating Oligonucleotides 2) By Synthetic Ribonucleic Acid Molecules: Messenger Ribonucleic Acid Molecules, Small Interfering Ribonucleic Acid Molecules, Guide Ribonucleic Acid Molecules, Modified Ribonucleic Acid Molecules, Therapeutic Ribonucleic Acid Constructs 3) By Exon-Targeted Gene Therapy Kits: Viral Vector Delivery Kits, Non-Viral Delivery Kits, Plasmid-Based Gene Therapy Kits, Targeted Editing Delivery Kits, Tissue Specific Delivery Kits 4) By Combination Therapy Formulations: Gene Therapy And Drug Combination, Oligonucleotide And Small Molecule Combination, Multi-Target Genetic Therapy, Supportive Protein Therapy Combination, Sequential Genetic Therapy Formulations 5) By Other Product Types: Peptide Based Genetic Modulators, Nanoparticle Delivery Formulations, Lipid Based Delivery Systems, Targeted Molecular Complexes, Personalized Genetic Therapy FormatsWhat Is The Driver Of The Exon Skipping Therapies Market?
The rising prevalence of genetic disorders is expected to drive the growth of the exon skipping therapies market over the forecast period. Genetic disorders are conditions caused by abnormalities in an individual’s genetic material (DNA), including single-gene mutations and chromosomal alterations that can result in inherited or acquired diseases. The apparent increase in the prevalence of genetic disorders is largely attributed to advancements in diagnostic technologies and the expanding use of genetic testing and newborn screening programs, which are enabling earlier and more accurate identification of previously undiagnosed cases. Exon skipping therapies are designed to address specific genetic mutations by selectively excluding faulty exons during messenger RNA (mRNA) processing, restoring the production of partially or fully functional proteins. This targeted therapeutic approach directly addresses the underlying molecular causes of certain genetic disorders, particularly rare and previously untreatable conditions, creating significant clinical and commercial interest. For instance, in October 2024, according to the National Health Service (NHS) England, a UK-based government department, a pioneering newborn screening initiative was launched across NHS hospitals to test hundreds of newborns for more than 200 rare genetic conditions, with plans to expand screening to up to 100,000 babies across England. Therefore, the rising prevalence of genetic disorders is driving the growth of the exon skipping therapies industry.Key Players In The Global Exon Skipping Therapies Market
Major companies operating in the exon skipping therapies market are Daiichi Sankyo Company Limited, BioMarin Pharmaceutical Inc., Sarepta Therapeutics, Nippon Shinyaku Co. Ltd., Ionis Pharmaceuticals Inc., PTC Therapeutics, Ionis Pharmaceuticals Inc., Entrada Therapeutics Inc., Wave Life Sciences, PepGen Inc., ProQR Therapeutics NV, NS Pharma Inc., Genethon, Astellas Gene Therapies, SQY Therapeutics, AUM Biotech Inc., Cure Rare Disease Inc., Autotelic Bio Inc., Dyne Therapeutics, Skip Therapeutics.Global Exon Skipping Therapies Market Trends and Insights
Major companies operating in the exon skipping therapies market are increasingly focusing on the development of innovative treatment modalities, such as investigational antibody-oligonucleotide conjugate (AOC) therapies, to improve targeted delivery and enhance dystrophin production in patients with Duchenne muscular dystrophy (DMD). AOC therapies represent a next-generation approach that combines a monoclonal antibody with a synthetic oligonucleotide, enabling the targeted delivery of exon-skipping molecules directly to muscle cells. For instance, in July 2025, Avidity Biosciences Inc., a U.S.-based biotechnology company, received Breakthrough Therapy designation from the U.S. Food and Drug Administration (FDA) for delpacibart zotadirsen (del-zota), an investigational AOC therapy for the treatment of DMD in patients with mutations amenable to exon 44 skipping. Del-zota consists of a proprietary monoclonal antibody conjugated to phosphorodiamidate morpholino oligomers (PMOs), which promote exon 44 skipping during mRNA processing. The therapy is currently in Phase 2 clinical development, with plans for regulatory submission supported by robust biomarker and clinical efficacy data. Such advancements underscore the growing emphasis on differentiated delivery platforms within the exon skipping therapies market and highlight how AOC technologies are shaping the future of precision treatments for genetic neuromuscular disorders.What Are Latest Mergers And Acquisitions In The Exon Skipping Therapies Market?
In November 2025, Biogen Inc., a US-based healthcare biotechnology company, acquired Alcyone Therapeutics for an undisclosed amount. With this acquisition, Biogen aimed to expand its drug delivery technology capabilities by integrating alcyone’s thecaflex DRx intrathecal delivery platform to enhance patient centric administration of neurologic therapies and strengthen its pipeline execution. Alcyone Therapeutics is a US-based clinical stage biotechnology company that specializes in developing exon skipping therapies.Regional Insights
North America was the largest region in the exon skipping therapies market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Exon Skipping Therapies Market?
The exon skipping therapies market consists of sales of eteplirsen, golodirsen, viltolarsen, and casimersen. Values in this market are “factory gate” values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services such as formulation development, regulatory compliance support, clinical trial assistance, and technical guidance sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Exon Skipping Therapies Market Report 2026?
The exon skipping therapies market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the exon skipping therapies industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Exon Skipping Therapies Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.81 billion |
| Revenue Forecast In 2035 | $5.9 billion |
| Growth Rate | CAGR of 20.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Route Of Administration, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Daiichi Sankyo Company Limited, BioMarin Pharmaceutical Inc., Sarepta Therapeutics, Nippon Shinyaku Co. Ltd., Ionis Pharmaceuticals Inc., PTC Therapeutics, Ionis Pharmaceuticals Inc., Entrada Therapeutics Inc., Wave Life Sciences, PepGen Inc., ProQR Therapeutics NV, NS Pharma Inc., Genethon, Astellas Gene Therapies, SQY Therapeutics, AUM Biotech Inc., Cure Rare Disease Inc., Autotelic Bio Inc., Dyne Therapeutics, Skip Therapeutics. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
