
Viral Vector Affinity Resin Market Report 2026
Global Outlook – By Product Type (Adenoviral Vector Affinity Resin, Lentiviral Vector Affinity Resin, Adeno-Associated Viral (AAV) Vector Affinity Resin, Retroviral Vector Affinity Resin, Other Product Types), By Application (Gene Therapy, Vaccine Development, Cell Therapy, Other Applications), By End-User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Research Organizations) - Market Size, Trends, And Global Forecast 2026-2035
Viral Vector Affinity Resin Market Overview
• Viral Vector Affinity Resin market size has reached to $0.62 billion in 2025 • Expected to grow to $1.3 billion in 2030 at a compound annual growth rate (CAGR) of 15.9% • Growth Driver: Increasing Adoption Of Gene-Based Therapeutics Fueling The Growth Of The Market Due To Rising Demand For Efficient And High-Purity Viral Vector Purification • North America was the largest region in 2025.What Is Covered Under Viral Vector Affinity Resin Market?
Viral vector affinity resin is a specialized chromatography material designed to selectively bind viral vectors based on specific surface proteins or capsid structures. It helps to purify viral vectors efficiently from complex mixtures, removing impurities such as host cell proteins, DNA, and empty capsids, to achieve high-quality, high-titer viral preparations for research or therapeutic applications. The main product types of viral vector affinity resin are adenoviral vector affinity resin, lentiviral vector affinity resin, adeno-associated viral (AAV) vector affinity resin, retroviral vector affinity resin, and others. Adenoviral Vector Affinity Resin refers to a specialized chromatography resin designed to selectively capture and purify adenovirus vectors based on their surface proteins or capsid structures. It is applied in gene therapy, vaccine development, cell therapy, and others, serving end users that include pharmaceutical and biotechnology companies, academic and research institutes, and contract research organizations.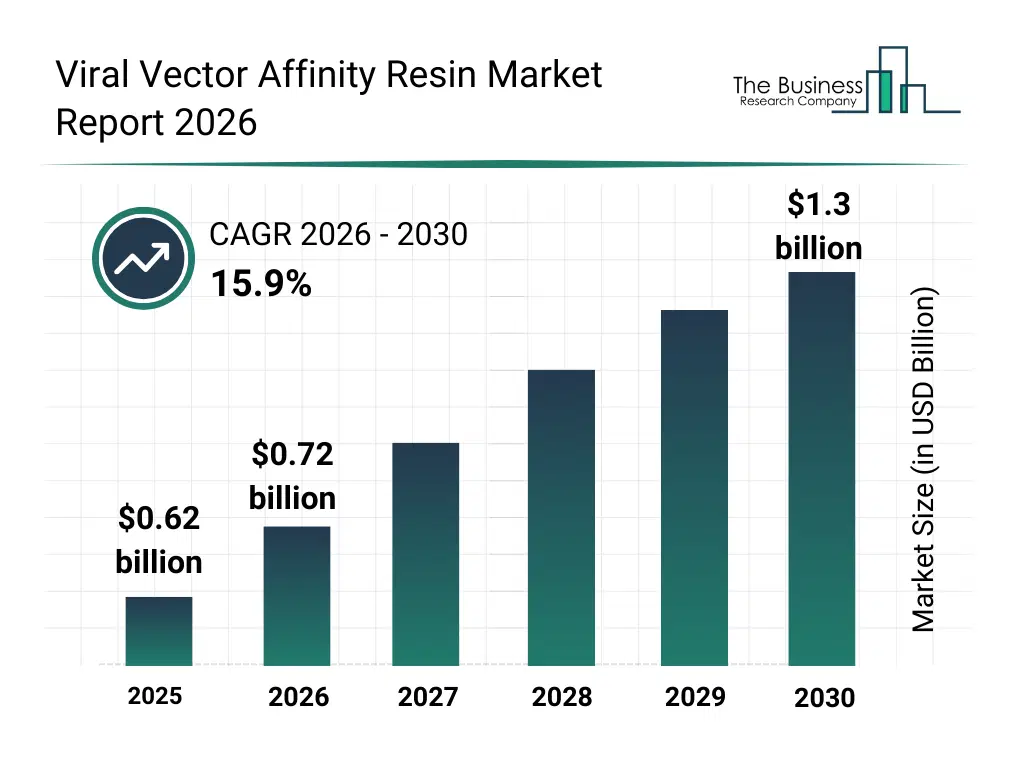
What Is The Viral Vector Affinity Resin Market Size and Share 2026?
The viral vector affinity resin market size has grown rapidly in recent years. It will grow from $0.62 billion in 2025 to $0.72 billion in 2026 at a compound annual growth rate (CAGR) of 16.2%. The growth in the historic period can be attributed to expansion of viral vector research activities, increasing use of chromatography in bioprocessing, growth in gene therapy development programs, rising demand for scalable purification solutions, improvements in resin manufacturing technologies.What Is The Viral Vector Affinity Resin Market Growth Forecast?
The viral vector affinity resin market size is expected to see rapid growth in the next few years. It will grow to $1.3 billion in 2030 at a compound annual growth rate (CAGR) of 15.9%. The growth in the forecast period can be attributed to increasing commercialization of gene therapies, rising demand for high-throughput purification platforms, expansion of vaccine development pipelines, growing focus on cost-efficient downstream processing, increasing adoption of customized affinity resins. Major trends in the forecast period include increasing adoption of high-capacity affinity resins, rising demand for aav and lentiviral purification solutions, growing use of single-use chromatography systems, expansion of ligand-specific resin development, enhanced focus on high-purity viral vector processing.Global Viral Vector Affinity Resin Market Segmentation
1) By Product Type: Adenoviral Vector Affinity Resin, Lentiviral Vector Affinity Resin, Adeno-Associated Viral (AAV) Vector Affinity Resin, Retroviral Vector Affinity Resin, Other Product Types 2) By Application: Gene Therapy, Vaccine Development, Cell Therapy, Other Applications 3) By End-User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Research Organizations Subsegments: 1) By Adenoviral Vector Affinity Resin: Cell Lysis-Based Adenoviral Vector Affinity Resin, Chromatography-Based Adenoviral Vector Affinity Resin, High-Capacity Adenoviral Vector Affinity Resin, Prepacked Column Adenoviral Vector Affinity Resin 2) By Lentiviral Vector Affinity Resin: High-Purity Lentiviral Vector Affinity Resin, Column-Based Lentiviral Vector Affinity Resin, Membrane Chromatography Lentiviral Vector Affinity Resin, Single-Use Lentiviral Vector Affinity Resin 3) By Adeno-Associated Viral Vector Affinity Resin: Serotype-Specific Adeno-Associated Viral Vector Affinity Resin, High-Capacity Adeno-Associated Viral Vector Affinity Resin, Prepacked Column Adeno-Associated Viral Vector Affinity Resin, Ligand-Based Adeno-Associated Viral Vector Affinity Resin 4) By Retroviral Vector Affinity Resin: High-Selectivity Retroviral Vector Affinity Resin, Column-Based Retroviral Vector Affinity Resin, Membrane-Based Retroviral Vector Affinity Resin, Prepacked Retroviral Vector Affinity Resin 5) By Other Product Types: Hybrid Viral Vector Affinity Resin, Multimodal Viral Vector Affinity Resin, Recombinant Ligand Affinity Resin, Customized Viral Vector Affinity ResinWhat Are The Drivers Of The Viral Vector Affinity Resin Market?
The increasing adoption of gene-based therapeutics is expected to propel the growth of the viral vector affinity resin market going forward. Gene-based therapeutics are advanced treatments that utilize DNA, RNA, or viral vectors to modify or regulate gene expression within a patient’s cells. The adoption of gene-based therapeutics is rising due to advancements in delivery systems, as they enable safe, efficient, and targeted transport of genetic material into cells. Viral vector affinity resin enhances gene-based therapeutics adoption by providing high-purity, scalable purification solutions, making them ideal for viral vector manufacturing. They reduce downstream processing challenges by efficiently isolating viral vectors from impurities, improving production efficiency and supporting broader clinical and commercial applications. For instance, in December 2024, according to the American Society of Gene & Cell Therapy (ASGCT), a US-based non-profit medical and scientific organization, the Food and Drug Administration (FDA) approved six gene therapy products in 2023, up from five approvals in 2022. Therefore, the increasing adoption of gene-based therapeutics is driving the growth of the viral vector affinity resin industry. The rising healthcare expenditure is expected to propel the growth of the viral vector affinity resin market going forward. Healthcare expenditure refers to the total amount of money spent on medical services, treatments, facilities, research, and public health initiatives to maintain or improve health outcomes. The increasing demand for healthcare expenditure is due to the rising prevalence of chronic diseases, which require long-term and costly treatments. Rising healthcare expenditure enhances the viral vector affinity resin market by driving investments in advanced biomanufacturing and purification technologies. Increased funding for gene and cell therapies boosts demand for high-quality viral vector production, where affinity resins ensure efficient purification, higher yields, and compliance with regulatory standards. For instance, in May 2024, according to the Office for National Statistics, a UK-based government department, total healthcare expenditure saw a 5.6% increase in nominal terms in 2023, an increase of 0.9% growth compared to 2022. Therefore, the rising healthcare expenditure is driving the growth of the viral vector affinity resin industry.Key Players In The Global Viral Vector Affinity Resin Market
Major companies operating in the viral vector affinity resin market are Thermo Fisher Scientific, GE Healthcare Technologies Inc., Repligen Corporation, Creative Biolabs, JNC Corporation, Aptamer Group, ChromaGenix Inc., Cytiva (Danaher Corporation), Merck KGaA, Sartorius AG, Purolite Life Sciences, Tosoh Bioscience, Bio‑Rad Laboratories, Avantor Inc., Pall Corporation, Takara Bio Inc., Cube Biotech, Agilent Technologies, Promega Corporation, Kaneka CorporationWhat Are Latest Mergers And Acquisitions In The Viral Vector Affinity Resin Market?
In July 2024, Repligen Corporation, a US-based biotechnology company, acquired Tantti Laboratory Inc. for an undisclosed amount. Through this acquisition, Repligen aims to enhance viral vector affinity resin capabilities and strengthen high-throughput purification solutions for emerging modalities such as viral vectors and nucleic acids by integrating Tantti’s advanced DuloCore bead technology with Avitide’s AVIPure affinity ligands. Tantti Laboratory Inc. is a Taiwan-based biomaterials engineering company that develops chromatography resins capable of purifying viral vectors.Regional Outlook
North America was the largest region in the viral vector affinity resin market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector Affinity Resin Market?
The viral vector affinity resin market consists of sales of viral vector purification resins, pre-packed chromatography columns, ancillary purification kits, and consumables. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector Affinity Resin Market Report 2026?
The viral vector affinity resin market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector affinity resin industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector Affinity Resin Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.72 billion |
| Revenue Forecast In 2035 | $1.3 billion |
| Growth Rate | CAGR of 16.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific, GE Healthcare Technologies Inc., Repligen Corporation, Creative Biolabs, JNC Corporation, Aptamer Group, ChromaGenix Inc., Cytiva (Danaher Corporation), Merck KGaA, Sartorius AG, Purolite Life Sciences, Tosoh Bioscience, Bio‑Rad Laboratories, Avantor Inc., Pall Corporation, Takara Bio Inc., Cube Biotech, Agilent Technologies, Promega Corporation, Kaneka Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
