
Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Report 2026
Global Outlook – By Service Type (Process Development, Manufacturing, Analytical and Quality Control, Fill-Finish, Other Service Types), By Therapy Type (Cell Therapy, Gene Therapy, Combination Therapy), By Phase (Preclinical, Clinical, Commercial), By End-User (Pharmaceutical and Biotechnology Companies, Academic and Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Overview
• Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy market size has reached to $2.49 billion in 2025 • Expected to grow to $5.88 billion in 2030 at a compound annual growth rate (CAGR) of 18.9% • Growth Driver: Increasing Number Of Cell And Gene Therapy Clinical Trials Fueling The Growth Of The Market Due To Expanding Demand For Compliant Manufacturing Support • Market Trend: Expansion Of Advanced Cell Therapy Manufacturing Capabilities To Support Clinical Development • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
Good manufacturing practice (GMP) services for cell and gene therapy refer to regulated manufacturing and quality-control activities that ensure cell- and gene-based therapies are produced safely, consistently, and in compliance with global regulatory standards. These services include GMP-compliant facilities, cell and viral vector processing, testing, documentation, and regulatory support for clinical and commercial use. The main service types of good manufacturing practice (GMP) services for cell and gene therapy include process development, manufacturing, analytical and quality control, fill-finish, and other service types. Process development refers to the design, optimization, and scale-up of cell and gene therapy production processes to ensure consistency, safety, and regulatory compliance. These services are applied across therapy types including cell therapy, gene therapy, and combination therapy, and phases including preclinical, clinical, and commercial. They are used by end users including pharmaceutical and biotechnology companies, academic and research institutes, and other end users.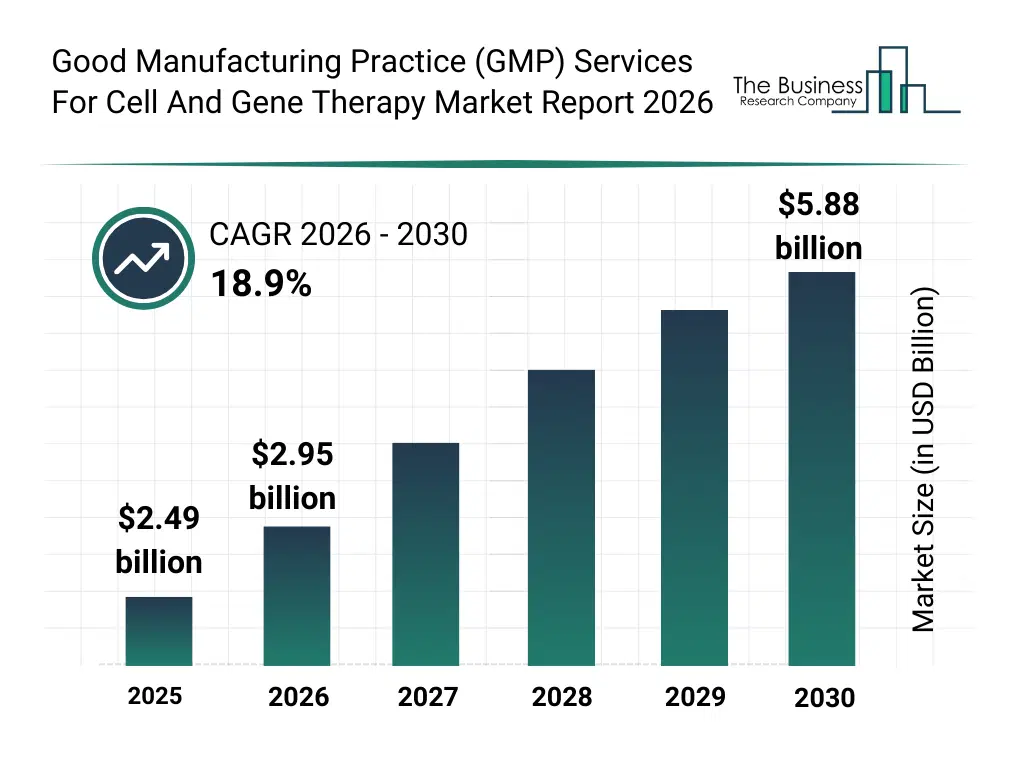
What Is The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Size and Share 2026?
The good manufacturing practice (gmp) services for cell and gene therapy market size has grown rapidly in recent years. It will grow from $2.49 billion in 2025 to $2.95 billion in 2026 at a compound annual growth rate (CAGR) of 18.6%. The growth in the historic period can be attributed to growth in clinical trials for advanced therapies, expansion of cell therapy research programs, increasing regulatory standardization, development of early-stage gmp facilities, rising investment in biopharmaceutical manufacturing.What Is The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Growth Forecast?
The good manufacturing practice (gmp) services for cell and gene therapy market size is expected to see rapid growth in the next few years. It will grow to $5.88 billion in 2030 at a compound annual growth rate (CAGR) of 18.9%. The growth in the forecast period can be attributed to increasing commercialization of gene therapies, rising demand for scalable manufacturing capacity, expansion of personalized medicine pipelines, growing outsourcing to cdmos, increasing adoption of automated quality control systems. Major trends in the forecast period include increasing demand for commercial-scale cell and gene therapy manufacturing, rising adoption of viral vector production services, growing focus on gmp-compliant automation, expansion of fill-finish and cold chain services, enhanced emphasis on regulatory compliance.Global Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Segmentation
1) By Service Type: Process Development; Manufacturing; Analytical and Quality Control; Fill-Finish; Other Service Types 2) By Therapy Type: Cell Therapy; Gene Therapy; Combination Therapy 3) By Phase: Preclinical; Clinical; Commercial 4) By End-User: Pharmaceutical and Biotechnology Companies; Academic and Research Institutes; Other End-Users Subsegments: 1) By Process Development: Vector Design Optimization; Cell Line Development; Upstream Process Optimization; Downstream Purification Development; Process Scale Up Studies 2) By Manufacturing: Clinical Trial Material Production; Commercial Scale Manufacturing; Viral Vector Manufacturing; Plasmid Production Services; Cell Therapy Manufacturing 3) By Analytical and Quality Control: Method Development And Validation; Potency And Efficacy Testing; Safety And Sterility Testing; Stability And Shelf Life Studies; Release Testing Services 4) By Fill-Finish: Aseptic Filling Services; Vial And Syringe Filling; Lyophilization Services; Packaging And Labeling; Cold Chain Handling 5) By Other Service Types: Regulatory Support Services; Technology Transfer Services; Supply Chain Management; Storage And Distribution; Post Manufacturing SupportWhat Is The Driver Of The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
The increasing number of cell and gene therapy clinical trials is expected to propel the growth of the good manufacturing practice (GMP) services for cell and gene therapy market going forward. Cell and gene therapy trials are regulated studies evaluating the safety, efficacy, and quality of advanced therapies that use genetically modified cells or living cells to treat conditions such as cancer, rare genetic disorders, and autoimmune diseases. The growth in these trials is being fueled by expanded research and development in advanced therapies, increased investment from biotechnology and pharmaceutical companies, and rising demand for innovative treatments for unmet medical needs. As many trial sponsors lack in-house GMP manufacturing capabilities, they increasingly rely on specialized GMP service providers for compliant manufacturing, analytical testing, quality control, and quality assurance. These services are critical to ensuring the safe, consistent, and timely supply of clinical-grade materials across trial phases. For instance, in January 2026, according to Bioindustry Association (BIA) a UK-based national trade association, the UK had 193 ongoing advanced therapy clinical trials, with over half in early stages and more than 80% focused on gene therapies. Therefore, the increasing number of cell and gene therapy clinical trials is driving the growth of the good manufacturing practice (GMP) services for cell and gene therapy industry.Key Players In The Global Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market
Major companies operating in the good manufacturing practice (gmp) services for cell and gene therapy market are Novartis AG, Boehringer Ingelheim BioXcellence GmbH, Batavia Biosciences B.V., FUJIFILM Diosynth Biotechnologies, AGC Biologics Inc., Lonza Group AG, WuXi AppTec Co. Ltd., BioNTech Innovative Manufacturing Services, Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Miltenyi Biotec GmbH, SK pharmteco Co. Ltd., KBI Biopharma Inc., Oxford Biomedica plc, OmniaBio Inc., RoslinCT Ltd., Pluri CDMO, SCTbio Inc., Made Scientific Inc., ENCell Co. Ltd.Global Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Trends and Insights
Major companies operating in the good manufacturing practice (GMP) services for cell and gene therapy market are increasingly focused on expanding advanced manufacturing capabilities, including cell therapy manufacturing, to support regional clinical trials and accelerate therapy development. Cell therapy manufacturing involves GMP-compliant processes for the collection, modification, expansion, and formulation of therapeutic cells to ensure product safety, quality, and clinical effectiveness. For instance, in July 2025, AGC Biologics, a US-based contract development and manufacturing organization (CDMO), launched cell therapy process development and clinical manufacturing services at its Yokohama Technical Center in Japan. The facility supports GMP-compliant production for preclinical and clinical trials, including CAR-T therapies, induced pluripotent stem cells, and other stem cell platforms. This expansion strengthens AGC Biologics’ global GMP footprint across Europe, North America, and Asia, enabling faster and more compliant advancement of cell and gene therapies toward commercialization.What Are Latest Mergers And Acquisitions In The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
In May 2025, Altaris LLC, a US-based healthcare investment firm, acquired Minaris Regenerative Medicine GmbH for an undisclosed amount. Through this acquisition, Altaris, LLC aims to expand its global GMP capabilities in cell and gene therapy by integrating Minaris Regenerative Medicine’s expertise in compliant manufacturing of advanced therapy products. Minaris Regenerative Medicine GmbH is a Germany-based contract development and manufacturing organization (CDMO) specializing in GMP-compliant cell and gene therapy manufacturing.Regional Insights
North America was the largest region in the good manufacturing practice (GMP) services for cell and gene therapy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
The good manufacturing practice (GMP) services for cell and gene therapy market includes revenues earned by entities through clinical manufacturing support, commercial scale manufacturing support, fill-finish services, cell banking services, raw material management services, quality control testing services, analytical testing services, regulatory support services, and quality assurance and compliance services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Report 2026?
The good manufacturing practice (gmp) services for cell and gene therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the good manufacturing practice (gmp) services for cell and gene therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.95 billion |
| Revenue Forecast In 2035 | $5.88 billion |
| Growth Rate | CAGR of 18.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Therapy Type, Phase, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Novartis AG, Boehringer Ingelheim BioXcellence GmbH, Batavia Biosciences B.V., FUJIFILM Diosynth Biotechnologies, AGC Biologics Inc., Lonza Group AG, WuXi AppTec Co. Ltd., BioNTech Innovative Manufacturing Services, Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Miltenyi Biotec GmbH, SK pharmteco Co. Ltd., KBI Biopharma Inc., Oxford Biomedica plc, OmniaBio Inc., RoslinCT Ltd., Pluri CDMO, SCTbio Inc., Made Scientific Inc., ENCell Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
