
Human Leukocyte Antigen (HLA) Antibody Screening Market Report 2026
Global Outlook – By Technology (Enzyme-Linked Immunosorbent Assay (ELISA), Flow Cytometry, Luminex, Other Technologies), By Product Type (Reagents And Kits, Instruments, Software), By Target Class (Human Leukocyte Antigen Class I Antibodies, Human Leukocyte Antigen Class II Antibodies, Non- Human Leukocyte Antigen Antibodies), By Application (Pre-Transplant Screening, Post-Transplant Monitoring, Transfusion Medicine, Research Applications), By End-User (Hospitals And Transplant Centers, Diagnostic Laboratories, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Human Leukocyte Antigen (HLA) Antibody Screening Market Overview
• Human Leukocyte Antigen (HLA) Antibody Screening market size has reached to $0.98 billion in 2025 • Expected to grow to $1.47 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Surge In Organ Transplantation Rates Fueling The Growth Of The Market Due To Increasing Demand For Donor-specific Antibody Detection And Improved Graft Survival Outcomes • Market Trend: Innovations In Next-Generation HLA Research Tools Strengthen Antibody Characterization And Assay Development Accuracy • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Human Leukocyte Antigen (HLA) Antibody Screening Market?
Human leukocyte antigen (HLA) antibody screening is a laboratory testing process used to detect the presence of antibodies directed against human leukocyte antigen (HLA) molecules in an individual’s blood. These antibodies may develop due to prior exposure to foreign hla proteins through events such as blood transfusion, pregnancy, or organ transplantation. The human leukocyte antigen (HLA) antibody screening helps assess immunological risk by identifying sensitization that could lead to complications in transplant compatibility or immune-mediated reactions. The main technologies types of human leukocyte antigen (HLA) antibody screening include enzyme-linked immunosorbent assay (ELISA), flow cytometry, luminex, and others. Enzyme-linked immunosorbent assay (ELISA) is a laboratory technique that detects and quantifies specific proteins, antigens, or antibodies in a sample using enzyme-linked antibodies that produce a measurable color change. The various product types include reagents and kits, instruments, and software and these solutions target human leukocyte antigen class I antibodies, human leukocyte antigen class II antibodies, and non-human leukocyte antigen antibodies. They find application in pre-transplant screening, post-transplant monitoring, transfusion medicine, and research applications, serving end users including hospitals and transplant centers, diagnostic laboratories, research institutes, and other end-users.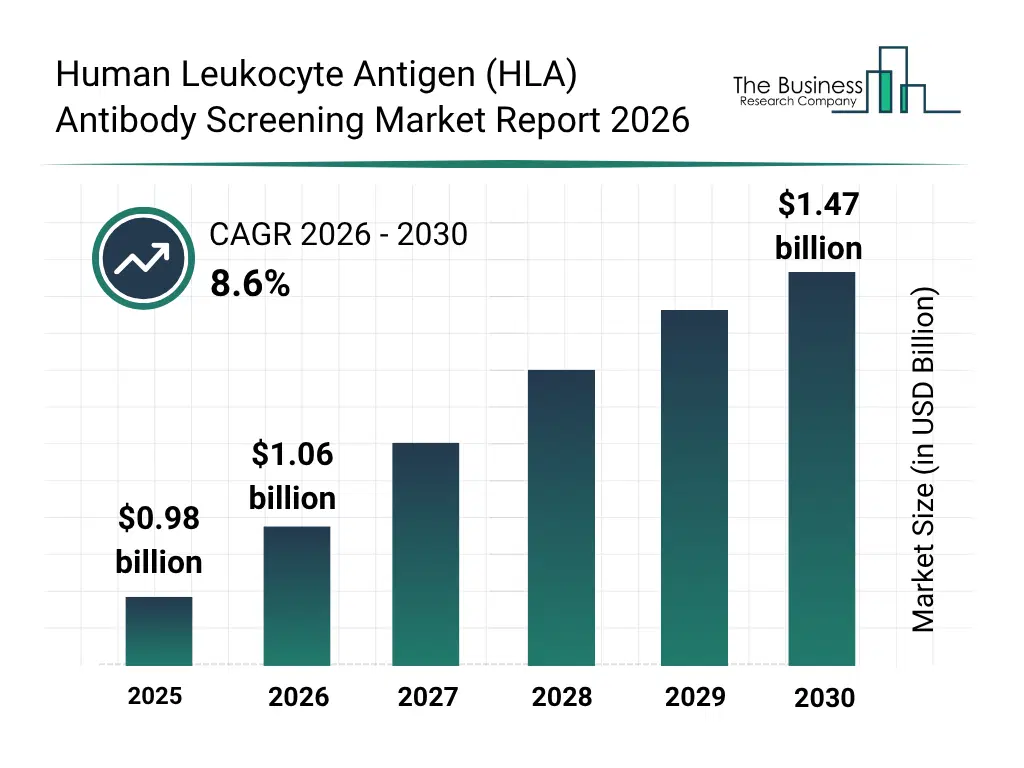
What Is The Human Leukocyte Antigen (HLA) Antibody Screening Market Size and Share 2026?
The human leukocyte antigen (HLA) antibody screening market size has grown strongly in recent years. It will grow from $0.98 billion in 2025 to $1.06 billion in 2026 at a compound annual growth rate (CAGR) of 8.4%. The growth in the historic period can be attributed to rising number of organ transplant procedures, increasing incidence of end-stage organ failure, growing awareness of transplant rejection risks, expansion of transfusion medicine practices, technological advancements in Immunoassay platforms.What Is The Human Leukocyte Antigen (HLA) Antibody Screening Market Growth Forecast?
The human leukocyte antigen (HLA) antibody screening market size is expected to see strong growth in the next few years. It will grow to $1.47 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to increasing demand for precision transplant matching, growing adoption of multiplex bead-based assays, rising investments in transplant immunology research, expansion of transplant programs in emerging markets, technological innovation in automated hla antibody screening systems. Major trends in the forecast period include increasing adoption of single antigen bead assays for enhanced sensitivity, rising demand for comprehensive pre-transplant immunological risk assessment, growing use of high-throughput flow cytometry in hla screening, expansion of post-transplant antibody monitoring protocols, increasing standardization of hla antibody detection methods.Global Human Leukocyte Antigen (HLA) Antibody Screening Market Segmentation
1) By Technology: Enzyme-Linked Immunosorbent Assay (ELISA), Flow Cytometry, Luminex, Other Technologies 2) By Product Type: Reagents And Kits, Instruments, Software 3) By Target Class: Human Leukocyte Antigen Class I Antibodies, Human Leukocyte Antigen Class II Antibodies, Non- Human Leukocyte Antigen Antibodies 4) By Application: Pre-Transplant Screening, Post-Transplant Monitoring, Transfusion Medicine, Research Applications 5) By End-User: Hospitals And Transplant Centers, Diagnostic Laboratories, Research Institutes, Other End-Users Subsegments: 1) By Enzyme Linked Immunosorbent Assay: Direct Enzyme Linked Immunosorbent Assay, Indirect Enzyme Linked Immunosorbent Assay, Sandwich Enzyme Linked Immunosorbent Assay, Competitive Enzyme Linked Immunosorbent Assay 2) By Flow Cytometry: Conventional Flow Cytometry, Bead Based Flow Cytometry, Imaging Flow Cytometry, High Throughput Flow Cytometry 3) By Luminex: Single Antigen Luminex Assay, Multi Antigen Luminex Assay, Bead Based Multiplex Luminex Assay, Custom Luminex Panels 4) By Other Technologies: Western Blotting, Radioimmunoassay, Complement Dependent Cytotoxicity Assay, Microarray Based AssayWhat Is The Driver Of The Human Leukocyte Antigen (HLA) Antibody Screening Market?
The rising organ transplantation rates are expected to propel the growth of the HLA antibody screening market going forward. Organ transplantation rates refer to the number of organ transplant procedures performed within a specific population and time, typically expressed per year or per million people. The rise in transplantation procedures is largely attributed to the increasing burden of chronic and end-stage diseases, as more patients advance to organ failure, the demand for organ replacement therapies correspondingly increases. HLA antibody screening supports higher organ transplantation success by ensuring better donor-recipient matching before surgery, thereby lowering the likelihood of immune-mediated rejection that reduces graft loss and complications, improving long-term transplant outcomes, and enabling more effective use of available donor organs. For instance, in January 2025, according to the Health Resources and Services Administration, a US-based Department of Health and Human Services agency, lung transplants experienced the highest proportional increase between 2023 and 2024, rising from 3,026 procedures to 3,340, which represents a 10.4% growth. Therefore, the rising organ transplantation rates are driving the growth of the HLA antibody screening market.Key Players In The Global Human Leukocyte Antigen (HLA) Antibody Screening Market
Major companies operating in the human leukocyte antigen (HLA) antibody screening market are Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Becton Dickinson Company, Laboratory Corporation of America Holdings, Quest Diagnostics Incorporated, Grifols S A, Beckman Coulter Inc., Bio Rad Laboratories Inc., QIAGEN N V, Takara Bio Inc., Fujirebio Inc., CareDx Inc., Histogenetics LLC, Creative Biolabs Inc., Biofortuna Ltd., Stanford Blood Center, GenDx B V, Omixon Biocomputing Ltd., BAG Diagnostics GmbH, Scisco Genetics Inc., TBG Diagnostics Ltd., American Red Cross.Global Human Leukocyte Antigen (HLA) Antibody Screening Market Trends and Insights
Major companies operating in the HLA antibody screening market are focusing on developing next-generation HLA research tools to improve immune checkpoint research, antibody validation, and assay development accuracy. Next-generation HLA research tools comprise innovative recombinant reagents, multiplexed analytical platforms, and high-resolution molecular technologies that allow accurate profiling of HLA molecules and their antibody interactions. For instance, in November 2025, HLA Protein Technologies, a US-based biotechnology company, launched its sHLA-G Monomer and Tetramer Portfolio, a next-generation HLA research tool that supports high-fidelity checkpoint biology studies, target validation, and assay development. The portfolio provides structurally validated recombinant HLA-G reagents optimized for immune receptor interaction studies, facilitating enhanced characterization of HLA-associated antibodies and advancing applications in transplant immunology, oncology, and immune tolerance research.What Are Latest Mergers And Acquisitions In The Human Leukocyte Antigen (HLA) Antibody Screening Market?
In June 2023, Innobiochips, a France-based biotechnology company, entered into a technology licensing agreement with BAG Diagnostics to advance transplantation diagnostics through expanded access to high-precision HLA antibody screening technologies. The partnership aims to strengthen Innobiochips’ position in the transplantation diagnostics market by licensing and commercializing BAG Diagnostics’ HLA antibody screening solutions, enhancing transplant compatibility testing capabilities. BAG Diagnostics is a Germany-based diagnostics company specializing in HLA typing and antibody screening assays used in transplantation medicine.Regional Insights
North America was the largest region in the human leukocyte antigen (HLA) antibody screening market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Human Leukocyte Antigen (HLA) Antibody Screening Market?
The human leukocyte antigen (HLA) antibody screening market consists of revenues earned by entities by providing services such as panel reactive antibody (PRA) testing, donor-specific antibody (DSA) identification, crossmatch testing, and antibody specificity and strength characterization. The market value includes the value of related goods sold by the service provider or included within the service offering. The human leukocyte antigen (HLA) antibody screening market also includes sales of automated ELISA processors, microplate washers, and fluorescence detection systems. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Human Leukocyte Antigen (HLA) Antibody Screening Market Report 2026?
The human leukocyte antigen (hla) antibody screening market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the human leukocyte antigen (hla) antibody screening industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Human Leukocyte Antigen (HLA) Antibody Screening Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.06 billion |
| Revenue Forecast In 2035 | $1.47 billion |
| Growth Rate | CAGR of 8.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Product Type, Target Class, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Becton Dickinson Company, Laboratory Corporation of America Holdings, Quest Diagnostics Incorporated, Grifols S A, Beckman Coulter Inc., Bio Rad Laboratories Inc., QIAGEN N V, Takara Bio Inc., Fujirebio Inc., CareDx Inc., Histogenetics LLC, Creative Biolabs Inc., Biofortuna Ltd., Stanford Blood Center, GenDx B V, Omixon Biocomputing Ltd., BAG Diagnostics GmbH, Scisco Genetics Inc., TBG Diagnostics Ltd., American Red Cross. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
