
Juvenile Idiopathic Arthritis Diagnostic Market Report 2026
Global Outlook – By Type (Serological Tests, Imaging Tests, Genetic Tests), By Technology (Molecular Diagnostics, Immunoassays), By Application (Hospital Laboratories, Diagnostic Centres) - Market Size, Trends, And Global Forecast 2026-2035
Juvenile Idiopathic Arthritis Diagnostic Market Overview
• Juvenile Idiopathic Arthritis Diagnostic market size has reached to $5.03 billion in 2025 • Expected to grow to $6.6 billion in 2030 at a compound annual growth rate (CAGR) of 5.6% • Growth Driver: Rising Prevalence Of Autoimmune Disorders Driving Growth Of The Market Due To Increasing Cases Of Immune-Mediated Joint Inflammation • Market Trend: Strategic Partnerships Driving Innovation In Autoimmune Diagnostics • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Juvenile Idiopathic Arthritis Diagnostic Market?
Juvenile idiopathic arthritis (JIA) diagnostic refers to the medical processes and tests used to identify JIA, a chronic autoimmune condition in children that causes persistent joint inflammation, pain, and stiffness, potentially leading to long-term joint damage and mobility issues. It is caused by an overactive immune system mistakenly attacking the body's own joint tissues due to genetic and environmental factors, triggering inflammation that, if left untreated, can result in growth complications, vision problems, and reduced quality of life. The main types of juvenile idiopathic arthritis diagnostics are serological tests, imaging tests, and genetic tests. Serological tests involve blood analysis to identify specific markers that indicate inflammation or autoimmune activity associated with JIA. Key technologies utilized in JIA diagnostics include molecular diagnostics, immunoassays, and flow cytometry. The services offered encompass diagnostic services, consultation services, and laboratory services and are applied across various settings, including hospital laboratories, diagnostic centers, and research institutes.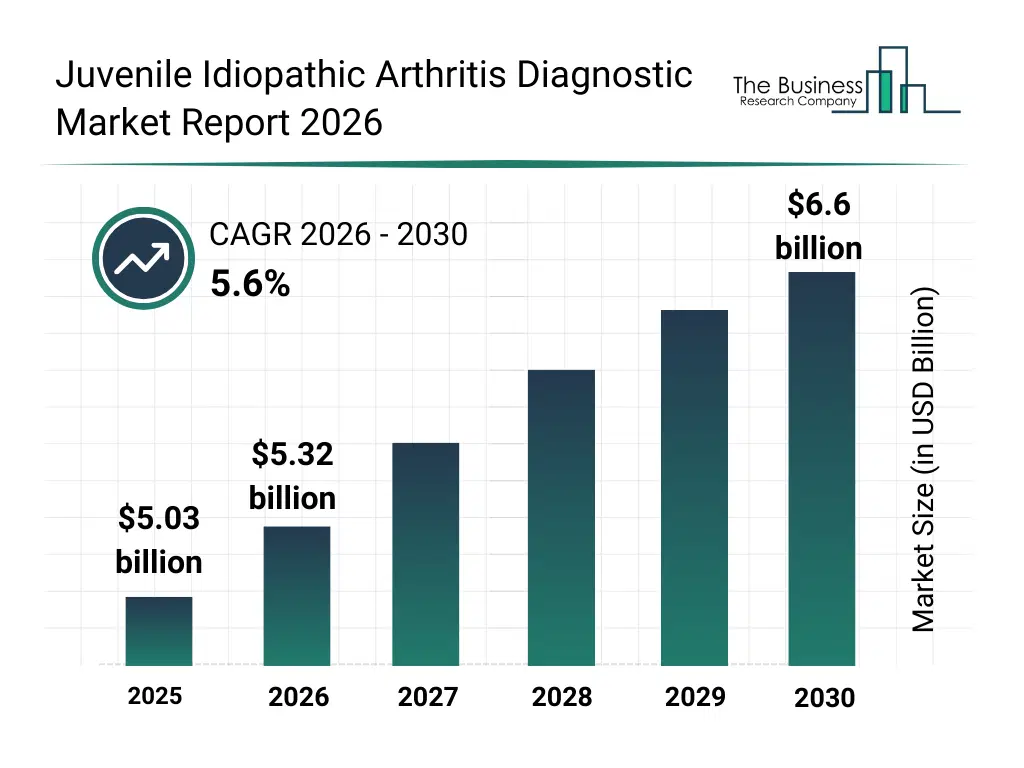
What Is The Juvenile Idiopathic Arthritis Diagnostic Market Size and Share 2026?
The juvenile idiopathic arthritis diagnostic market size has grown strongly in recent years. It will grow from $5.03 billion in 2025 to $5.32 billion in 2026 at a compound annual growth rate (CAGR) of 5.8%. The growth in the historic period can be attributed to increasing prevalence of autoimmune disorders in children, growing awareness of juvenile idiopathic arthritis, improved availability of serological and imaging tests, expansion of pediatric specialty care, rising use of laboratory diagnostics.What Is The Juvenile Idiopathic Arthritis Diagnostic Market Growth Forecast?
The juvenile idiopathic arthritis diagnostic market size is expected to see strong growth in the next few years. It will grow to $6.6 billion in 2030 at a compound annual growth rate (CAGR) of 5.6%. The growth in the forecast period can be attributed to advancement in molecular and genetic testing technologies, increasing focus on early disease diagnosis, rising investment in pediatric healthcare infrastructure, growing demand for personalized medicine approaches, expansion of diagnostic services in emerging markets. Major trends in the forecast period include early and accurate diagnosis of pediatric autoimmune disorders, growing use of advanced imaging in jia detection, rising adoption of genetic and biomarker-based testing, increasing demand for multimodal diagnostic approaches, expansion of specialized pediatric rheumatology diagnostics.Global Juvenile Idiopathic Arthritis Diagnostic Market Segmentation
1) By Type: Serological Tests, Imaging Tests, Genetic Tests 2) By Technology: Molecular Diagnostics, Immunoassays 3) By Application: Hospital Laboratories, Diagnostic Centres Subsegments: 1) By Serological Tests: Rheumatoid Factor (RF) Test, Antinuclear Antibody (ANA) Test, Erythrocyte Sedimentation Rate (ESR) Test, C-Reactive Protein (CRP) Test, Complete Blood Count (CBC) 2) By Imaging Tests: X-Ray, Magnetic Resonance Imaging (MRI), Ultrasound 3) By Genetic Tests: HLA-B27 Genetic Marker Test, Polymerase Chain Reaction (PCR)-Based TestingWhat Is The Driver Of The Juvenile Idiopathic Arthritis Diagnostic Market?
The increasing prevalence of autoimmune disorders is expected to propel the juvenile idiopathic arthritis diagnostic market going forward. Autoimmune disorders refer to conditions in which the immune system mistakenly attacks the body's healthy cells, causing inflammation, tissue damage, and chronic health complications. The rise in autoimmune diseases is attributed to a complex interplay of environmental triggers, genetic predispositions, and lifestyle changes. Autoimmune diseases cause juvenile idiopathic arthritis (JIA) by triggering the immune system to mistakenly attack the synovial tissue in joints, leading to inflammation and damage. For instance, in November 2023, according to the Multiple Sclerosis International Federation (MSIF), a UK-based international nonprofit organization focused on global MS research and advocacy, an estimated 2.9 million people worldwide were living with multiple sclerosis, a type of autoimmune disease in 2023. Therefore, the increasing prevalence of autoimmune disorders is driving the growth of the juvenile idiopathic arthritis diagnostic industry.Key Players In The Global Juvenile Idiopathic Arthritis Diagnostic Market
Major companies operating in the juvenile idiopathic arthritis diagnostic market are Thermo Fisher Scientific Inc., Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Danaher Corporation, Bio-Rad Laboratories, DiaSorin S.p.A., Sysmex Corporation, Beckman Coulter Inc., BD Biosciences, Agilent Technologies, Merck KGaA, Illumina Inc., Qiagen N.V., Hologic Inc., PerkinElmer Inc., Ortho Clinical Diagnostics, Grifols S.A., Fujirebio, Randox LaboratoriesGlobal Juvenile Idiopathic Arthritis Diagnostic Market Trends and Insights
Major companies operating in the juvenile idiopathic arthritis diagnostic market are focusing on strategic partnerships to expand their product offerings, enhance research capabilities, and strengthen their market presence globally. Strategic partnerships refer to collaborations between organizations aimed at advancing early detection and diagnosis. For instance, in November 2024, Augurex Life Sciences Corp., a Canada-based biotechnology company, partnered with Quest Diagnostics, a US-based diagnostic services company, to improve the diagnosis and management of rheumatoid arthritis (RA) using the 14-3-3η (eta) biomarker. As part of the collaboration, Quest Diagnostics will validate and offer a lab-developed test in the United States based on Augurex’s 14-3-3η biomarker. The test is intended to detect elevated levels of the 14-3-3η protein, which is linked to rheumatoid arthritis and associated with increased disease severity and risk of joint damage.What Are Latest Mergers And Acquisitions In The Juvenile Idiopathic Arthritis Diagnostic Market?
In September 2023, Globus Medical Inc., a US-based medical device company, merged with NuVasive Inc. to offer surgeons and patients a comprehensive range of musculoskeletal procedural solutions and advanced technologies, improving the overall continuum of care. The combined entity is expected to strengthen complementary global operations, broaden its market presence, and significantly expand its portfolio in arthritis and orthopedic treatment solutions. NuVasive, Inc. is a US-based medical device company that specializes in developing medical devices and procedures for orthopedic and idiopathic conditions.Regional Outlook
North America was the largest region in the juvenile idiopathic arthritis diagnostic market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Juvenile Idiopathic Arthritis Diagnostic Market?
The juvenile idiopathic arthritis diagnostic market consists of revenues earned by entities by providing services such as clinical evaluations, blood tests, ophthalmologic screenings, bone density scans, and synovial fluid analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. The juvenile idiopathic arthritis diagnostic market also includes sales of bone density scanners, AI-based diagnostic software, imaging equipment, and biomarker-based diagnostic panels. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Juvenile Idiopathic Arthritis Diagnostic Market Report 2026?
The juvenile idiopathic arthritis diagnostic market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the juvenile idiopathic arthritis diagnostic industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Juvenile Idiopathic Arthritis Diagnostic Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.32 billion |
| Revenue Forecast In 2035 | $6.6 billion |
| Growth Rate | CAGR of 5.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Technology, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Danaher Corporation, Bio-Rad Laboratories, DiaSorin S.p.A., Sysmex Corporation, Beckman Coulter Inc., BD Biosciences, Agilent Technologies, Merck KGaA, Illumina Inc., Qiagen N.V., Hologic Inc., PerkinElmer Inc., Ortho Clinical Diagnostics, Grifols S.A., Fujirebio, Randox Laboratories |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
