
Medical Device Labeling Market Report 2026
Global Outlook – By Type (Pressure Sensitive Labels, Glue Applied Labels, Sleeve Labels, In Mold Labels, Other Types), By Device Type (Diagnostic Devices, Therapeutic Devices, Surgical Devices, Monitoring Devices, Assistive Devices), By Material (Paper, Plastic, Other Materials), By Technology (Traditional Labeling, Smart Labeling Systems, Interactive Labels, Quick Response Code And Barcoding Technologies, Digital Printing Technologies), By End User (Hospitals, Clinics, Home Healthcare, Diagnostic Laboratories, Research Institutions) – Market Size, Trends, Strategies, and Forecast to 2035
Medical Device Labeling Market Overview
• Medical Device Labeling market size has reached to $1.23 billion in 2025 • Expected to grow to $1.72 billion in 2030 at a compound annual growth rate (CAGR) of 7% • Growth Driver: The Rise In Product Recalls Drives The Growth Of The Market Due To Increasing Need For Accurate Device Identification And Traceability • Market Trend: Innovative Adhesive Technologies Enhance Safety, Durability, And Compliance In Healthcare Identification • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Labeling Market?
Medical device labeling refers to all written, printed, or graphic information that accompanies a medical device, including the label on the device itself, packaging, instructions for use, manuals, and promotional materials. It provides essential information such as the device's name, intended use, instructions, warnings, manufacturer details, and regulatory symbols. Medical device labeling plays a key role in regulatory compliance, helping to ensure that devices meet the standards set by authorities. The main types of medical device labeling are pressure-sensitive labels, glue-applied labels, sleeve labels, in-mold labels, and others. Pressure-sensitive medical device labeling refers to labels with adhesive backing that stick to medical devices when pressure is applied, without the need for heat or water. These labels are used across various device categories, such as diagnostic devices, therapeutic devices, surgical devices, monitoring devices, and assistive devices. Labeling materials typically include paper, plastic, and others are these labels incorporate a range of technologies, such as traditional labeling, smart labeling systems, interactive labels, quick response codes and barcoding technologies, and digital printing technologies. Cater to various end users, including hospitals, clinics, home healthcare, diagnostic laboratories, and research institutions.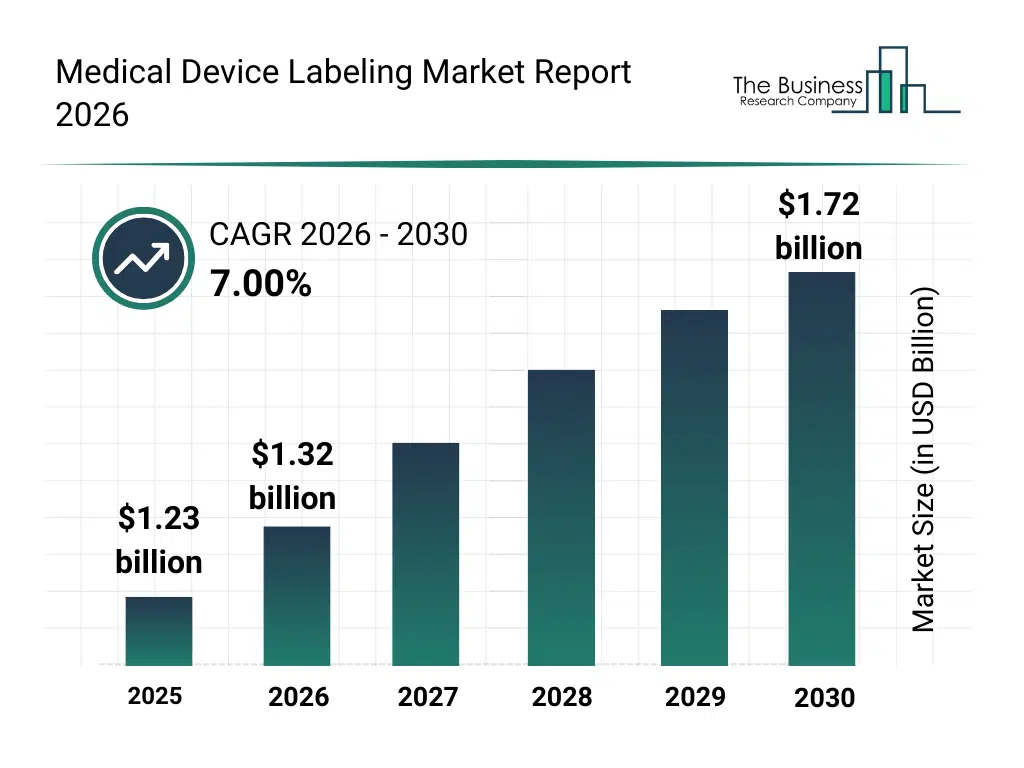
What Is The Medical Device Labeling Market Size and Share 2026?
The medical device labeling market size has grown strongly in recent years. It will grow from $1.23 billion in 2025 to $1.32 billion in 2026 at a compound annual growth rate (CAGR) of 7.3%. The growth in the historic period can be attributed to reliance on traditional labeling methods, manual labeling processes, limited adoption of smart labels, growing regulatory requirements, increasing medical device production volumes.What Is The Medical Device Labeling Market Growth Forecast?
The medical device labeling market size is expected to see strong growth in the next few years. It will grow to $1.72 billion in 2030 at a compound annual growth rate (CAGR) of 7.0%. The growth in the forecast period can be attributed to integration of digital printing and smart labeling technologies, adoption of interactive and qr code-enabled labels, increasing demand for patient safety and anti-counterfeiting solutions, expansion of home healthcare and diagnostic devices, rising investment in automation and connected labeling systems. Major trends in the forecast period include rising adoption of smart and interactive labels, increasing use of qr codes and barcoding technologies, growth in digital printing for medical devices, focus on tamper-evident and high-security labeling, regulatory compliance and standardization in labeling practices.Global Medical Device Labeling Market Segmentation
1) By Type: Pressure Sensitive Labels, Glue Applied Labels, Sleeve Labels, In Mold Labels, Other Types 2) By Device Type: Diagnostic Devices, Therapeutic Devices, Surgical Devices, Monitoring Devices, Assistive Devices 3) By Material: Paper, Plastic, Other Materials 4) By Technology: Traditional Labeling, Smart Labeling Systems, Interactive Labels, Quick Response Code And Barcoding Technologies, Digital Printing Technologies 5) By End User: Hospitals, Clinics, Home Healthcare, Diagnostic Laboratories, Research Institutions Subsegments: 1) By Pressure Sensitive Labels: Removable Labels, Permanent Labels, High-Temperature Labels, Freezer-Grade Labels, Tamper-Evident Labels 2) By Glue Applied Labels: Cut-And-Stack Labels, Wrap-Around Labels, Wet Glue Labels, Cold Glue Labels, Gummed Labels 3) By Sleeve Labels: Shrink Sleeve Labels, Stretch Sleeve Labels, Full-Body Sleeve Labels, Partial Sleeve Labels, Tamper-Evident Sleeve Labels 4) By In Mold Labels: Blow Molding Labels, Injection Molding Labels, Thermoforming Labels, Polypropylene Labels, High-Density Polyethylene Labels 5) By Other Types: Laser Labels, Thermal Transfer Labels, Direct Thermal Labels, Smart Labels, Radio Frequency Identification LabelsWhat Is The Driver Of The Medical Device Labeling Market?
The increasing product recalls are expected to propel the growth of the medical device labeling market going forward. Product recalls refer to the process of retrieving defective or unsafe products from consumers, manufacturers, or distributors to prevent harm and ensure compliance with safety standards. Product recalls are increasing due to rising consumer safety concerns, as people demand higher standards for product quality and protection against health or safety risks. Medical device labeling helps product recalls by providing accurate identification, usage instructions, and traceability details to efficiently locate and remove affected devices from the market. For instance, in February 2024, according to Sedgwick LP, a US-based software company, in 2023, U.S. product recalls reached a seven-year high, rising 11% compared to 2022, with a total of 975 medical-device recall events. Therefore, the increasing product recalls are driving the growth of the medical device labeling industry.Key Players In The Global Medical Device Labeling Market
Major companies operating in the medical device labeling market are Avery Dennison Corporation, Trumpf SE + Co. KG, Zebra Technologies Corporation, CCL Industries Inc., Brady Corporation, SATO Holdings Corporation, Herma Holding GmbH + Co. KG, Weber Packaging Solutions Inc., Tapecon Inc., Denny Bros Ltd., JH Bertrand Inc., Royal Label Co. Inc., Schreiner Group GmbH & Co. KG, Sidco Labeling Systems, Amcor Limited, Mondi Group Plc, Lintec Corporation, Huhtamaki Oyj, UPM Raflatac, WS Packaging Group Inc., Resource Label Group LLC, Faubel & Co Nachf GmbH, Label SourceGlobal Medical Device Labeling Market Trends and Insights
Major companies operating in the medical device labeling market are focusing on developing advanced adhesive solutions, such as permanent solvent-based biocompatible adhesive, to enhance label durability, ensure patient safety, and comply with stringent regulatory standards for long-term medical device identification. Permanent solvent-based biocompatible adhesive refers to a medical-grade adhesive formulation that uses solvent chemistry to create a long-lasting bond while being safe for contact with human tissue or medical devices. For instance, in December 2023, Flexcon Company Inc., a US-based manufacturing company, launched the Flexcon MedFlex product line of polyester printable films and overlaminates. This line is designed for lifetime labeling and tracking of Food and Drug Administration (FDA) Class I, II, and III medical devices used outside the body. MedFlex products meet all FDA UDI performance criteria and include UL-recognized, biocompatible adhesive solutions with sustainability features. This aims to simplify product selection and enhance durability, safety, and compliance in medical device labelling.What Are Latest Mergers And Acquisitions In The Medical Device Labeling Market?
In July 2023, Resource Label Group LLC, a US-based labels and packaging company, acquired Pharmaceutic Litho and Label Company for an undisclosed amount. With this acquisition, Resource Label Group aims to strengthen its position in the pharmaceutical and medical device labeling market by expanding its capabilities and geographic reach. Pharmaceutic Litho And Label Company Inc is a US-based pharmaceutical labeling company and manufacturer of printed inserts, labels, and packaging components for pharmaceutical, biotech, and medical device manufacturers.Regional Outlook
North America was the largest region in the medical device labeling market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Medical Device Labeling Market?
The medical device labeling market consists of revenues earned by entities by providing services such as design and development of labels, regulatory compliance services, printing and packaging services, labeling software solutions, and multilingual and localization services. The market value includes the value of related goods sold by the service provider or included within the service offering. The medical device labeling market also includes sales of sterilization indicator labels, instructional labels, compliance labels, and branding and logo labels. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Medical Device Labeling Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.32 billion |
| Revenue Forecast In 2035 | $1.72 billion |
| Growth Rate | CAGR of 7.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Device Type, Material, Technology, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Avery Dennison Corporation, Trumpf SE + Co. KG, Zebra Technologies Corporation, CCL Industries Inc., Brady Corporation, SATO Holdings Corporation, Herma Holding GmbH + Co. KG, Weber Packaging Solutions Inc., Tapecon Inc., Denny Bros Ltd., JH Bertrand Inc., Royal Label Co. Inc., Schreiner Group GmbH & Co. KG, Sidco Labeling Systems, Amcor Limited, Mondi Group Plc, Lintec Corporation, Huhtamaki Oyj, UPM Raflatac, WS Packaging Group Inc., Resource Label Group LLC, Faubel & Co Nachf GmbH, Label Source |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
