
Neurogenic Detrusor Overactivity Market Report 2026
Global Outlook – By Types (Non-Surgical Treatment, Surgical Treatment, Follow-Up Treatment), By Application (Adult, Child), By End-User (Hospitals, Urology Clinics, Rehabilitation Centers, Private Practices, Long-Term Care Facilities) – Market Size, Trends, Strategies, and Forecast to 2035
Neurogenic Detrusor Overactivity Market Overview
• Neurogenic Detrusor Overactivity market size has reached to $1.82 billion in 2025 • Expected to grow to $2.63 billion in 2030 at a compound annual growth rate (CAGR) of 7.6% • Growth Driver: Rising Incidence Of Neurological Disorders Drives Growth Of Market Due To Higher Demand For Effective Bladder Dysfunction Treatments • Market Trend: Advanced Generic Therapies Improving Pediatric NDO Treatment Outcomes • North America was the largest region in 2025.What Is Covered Under Neurogenic Detrusor Overactivity Market?
Neurogenic detrusor overactivity (NDO) is a bladder dysfunction caused by neurological conditions that disrupt the normal nerve signals between the brain, spinal cord, and the detrusor muscle of the bladder. This disruption leads to increased or involuntary contractions of the detrusor muscle, resulting in symptoms such as urinary urgency, frequency, urge incontinence, and sometimes incomplete bladder emptying. The main treatment types for neurogenic detrusor overactivity (NDO) include non-surgical treatment, surgical treatment, and follow-up treatment. Non-surgical treatment refers to medical care or therapies that manage or cure a condition without the need for invasive surgical procedures. These treatments are used for different applications, including adults and children, for varied end-users such as hospitals, urology clinics, rehabilitation centers, private practices, and long-term care facilities.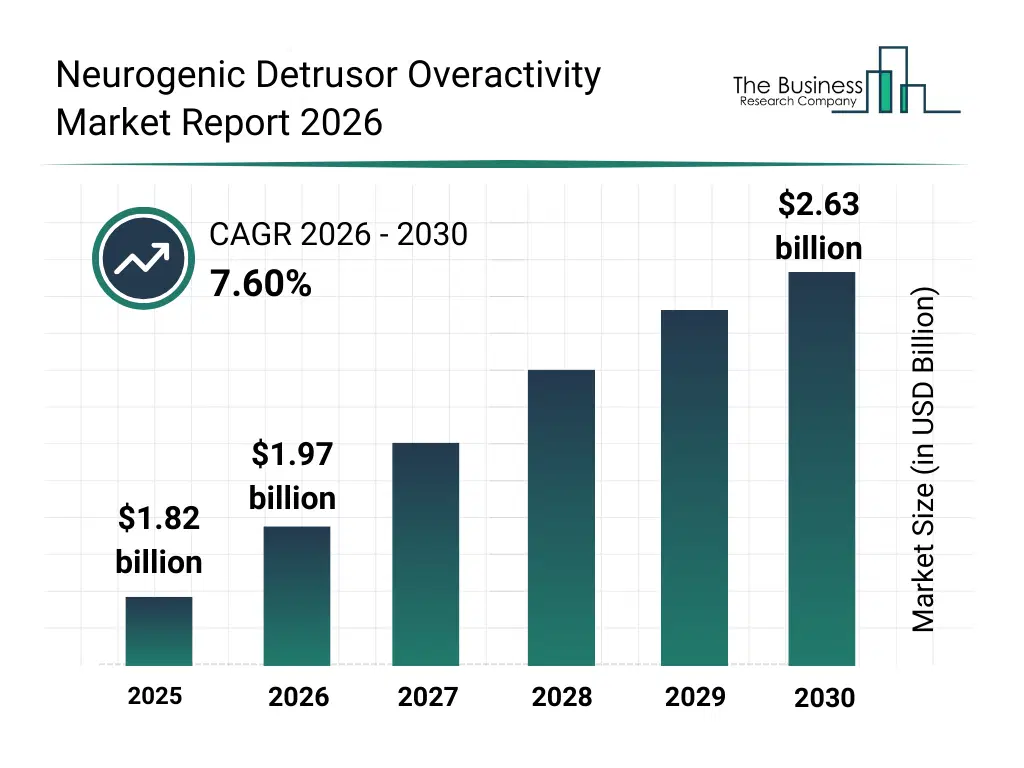
What Is The Neurogenic Detrusor Overactivity Market Size and Share 2026?
The neurogenic detrusor overactivity market size has grown strongly in recent years. It will grow from $1.82 billion in 2025 to $1.97 billion in 2026 at a compound annual growth rate (CAGR) of 7.9%. The growth in the historic period can be attributed to limited treatment options for NDO, reliance on conventional surgical interventions, growing prevalence of neurogenic bladder disorders, increasing hospital and urology clinic infrastructure, lack of patient and caregiver education.What Is The Neurogenic Detrusor Overactivity Market Growth Forecast?
The neurogenic detrusor overactivity market size is expected to see strong growth in the next few years. It will grow to $2.63 billion in 2030 at a compound annual growth rate (CAGR) of 7.6%. The growth in the forecast period can be attributed to development of novel pharmacological therapies, advancement in minimally invasive surgical procedures, integration of neuromodulation devices, increasing adoption of continuous patient monitoring technologies, rising investment in long-term care and rehabilitation services. Major trends in the forecast period include rising adoption of antimuscarinic and beta-3 adrenergic agonists, increased use of neuromodulation and minimally invasive therapies, expansion of behavioral therapy and patient training programs, growing focus on urodynamic and ultrasound monitoring, increasing awareness of quality of life assessment and long-term care.Global Neurogenic Detrusor Overactivity Market Segmentation
1) By Types: Non-Surgical Treatment, Surgical Treatment, Follow-Up Treatment 2) By Application: Adult, Child 3) By End-User: Hospitals, Urology Clinics, Rehabilitation Centers, Private Practices, Long-Term Care Facilities Subsegments: 1) By Non-Surgical Treatment: Antimuscarinic Drugs, Beta-3 Adrenergic Agonists, Behavioral Therapy, Neuromodulation, Clean Intermittent Catheterization, Intravesical Therapies 2) By Surgical Treatment: Botulinum Toxin Injection, Augmentation Cystoplasty, Urinary Diversion, External Sphincterotomy, Artificial Urinary Sphincter Implantation 3) By Follow-Up Treatment: Urodynamic Evaluation, Ultrasound Monitoring, Laboratory Tests, Ongoing Catheterization, Repeat Botulinum Toxin Injections, Patient and Caregiver Training, Quality of Life AssessmentWhat Is The Driver Of The Neurogenic Detrusor Overactivity Market?
The rising incidence of neurological disorders is expected to propel the growth of the neurogenic detrusor overactivity market going forward. Neurological disorders refer to medical conditions that affect the brain, spinal cord, or nerves, leading to problems with movement, sensation, cognition, or other bodily functions. Neurological disorders are rising as people are living longer, which increases the chances of age-related brain degeneration. Neurogenic detrusor overactivity (NDO) is a bladder dysfunction commonly associated with neurological disorders, such as spinal cord injury, multiple sclerosis, and Parkinson’s disease. These conditions disrupt normal nerve signaling between the brain and bladder, leading to involuntary bladder contractions. For instance, in April 2024, according to a study published by the American Academy of Neurology (AAN), a US-based neurology and neuroscience organization, there will be an estimated 4.9 billion cases of brain disorders globally by 2050, a 22% increase from approximately 4 billion cases in 2021. Therefore, the rising incidence of neurological disorders is driving growth in the neurogenic detrusor overactivity industry.Key Players In The Global Neurogenic Detrusor Overactivity Market
Major companies operating in the neurogenic detrusor overactivity market are Astellas Pharma Inc., Medtronic plc, Ipsen, Coloplast A/S, Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Viatris Inc., Apotex Inc., Intas Pharmaceuticals Ltd., Macleods Pharmaceuticals Ltd., Aurobindo Pharma Limited, Hugel Inc., Abbott Laboratories, Boston Scientific Corporation, Cook Medical, Convatec Group plc, B. Braun Melsungen AG, Teleflex Incorporated, Olympus Corporation, Karl Storz SE & Co. KG, Laborie Medical Technologies, Wellspect HealthCare (DENTSPLY Sirona), Hollister Incorporated, Uroplasty Inc., Endo Pharmaceuticals Inc., Ferring Pharmaceuticals, Hikma Pharmaceuticals PLC, Dr. Reddy’s Laboratories Ltd., Lupin Limited, Zydus Lifesciences Ltd.Global Neurogenic Detrusor Overactivity Market Trends and Insights
Major companies operating in the neurogenic detrusor overactivity (NDO) treatment market are focusing on developing advanced generic therapeutics such as beta-3 adrenergic receptor agonists to deliver effective bladder control solutions at reduced costs. Beta-3 adrenergic receptor agonists, such as mirabegron, function by relaxing the detrusor muscle in the bladder, thereby increasing bladder capacity and reducing symptoms such as urgency and incontinence. For instance, in April 2024, Lupin Ltd., an India-based pharmaceutical company, received approval from the U.S. Food and Drug Administration (USFDA) to launch Mirabegron, the generic version of Myrbetriq, in the United States. As an advanced once-daily oral therapy, Mirabegron offers a convenient and effective treatment option for patients with overactive bladder (OAB) and neurogenic detrusor overactivity (NDO) in pediatric populations. This approval reinforces Lupin’s commitment to expanding access to high-quality, affordable generic medications in critical therapeutic areas.What Are Latest Mergers And Acquisitions In The Neurogenic Detrusor Overactivity Market?
In November 2024, Boston Scientific Corporation, a US-based medical device company, acquired Axonics, Inc. for an undisclosed amount. Through this acquisition, Boston Scientific aims to broaden its neuromodulation and urology offerings by incorporating Axonics' advanced sacral neuromodulation technologies, strengthen its position in treating patients with overactive bladder and other urinary and bowel dysfunctions, and enhance its ability to deliver comprehensive solutions for pelvic health conditions. Axonics Inc. is a US-based medical technology company that provides sacral neuromodulation (SNM) systems that improve the control of an overactive bladder.Regional Insights
North America was the largest region in the neurogenic detrusor overactivity market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Neurogenic Detrusor Overactivity Market?
The neurogenic detrusor overactivity market includes revenues earned by entities by providing services such as medication management, behavioral and lifestyle interventions, physiotherapy, absorbent products and skin care, and catheterization. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Neurogenic Detrusor Overactivity Market Report 2026?
The neurogenic detrusor overactivity market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the neurogenic detrusor overactivity industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Neurogenic Detrusor Overactivity Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.97 billion |
| Revenue Forecast In 2035 | $2.63 billion |
| Growth Rate | CAGR of 7.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Types, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Astellas Pharma Inc., Medtronic plc, Ipsen, Coloplast A/S, Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Viatris Inc., Apotex Inc., Intas Pharmaceuticals Ltd., Macleods Pharmaceuticals Ltd., Aurobindo Pharma Limited, Hugel Inc., Abbott Laboratories, Boston Scientific Corporation, Cook Medical, Convatec Group plc, B. Braun Melsungen AG, Teleflex Incorporated, Olympus Corporation, Karl Storz SE & Co. KG, Laborie Medical Technologies, Wellspect HealthCare (DENTSPLY Sirona), Hollister Incorporated, Uroplasty Inc., Endo Pharmaceuticals Inc., Ferring Pharmaceuticals, Hikma Pharmaceuticals PLC, Dr. Reddy’s Laboratories Ltd., Lupin Limited, Zydus Lifesciences Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
