Omics-Based Clinical Trials Market Report 2026

Omics-Based Clinical Trials Market Report 2026
Global Outlook – By Type (Genomics, Proteomics, Metabolomics, Transcriptomics), By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Interventional Studies, Observational Studies, Expanded Access Studies), By Application (Oncology, Cardiovascular, Neurology, Infectious Diseases, Metabolic Disorders), By End-User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Research Organizations (CROs), Hospitals And Diagnostic Laboratories) - Market Size, Trends, And Global Forecast 2026-2035
Omics-Based Clinical Trials Market Overview
• Omics-Based Clinical Trials market size has reached to $31.71 billion in 2025 • Expected to grow to $47.78 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Rising Demand For Personalized Medicine Accelerates Growth Of The Market Due To Genomic Advancements And Targeted Treatment Approaches • Market Trend: Advancements In HiFi Sequencing Boosting Trial Precision • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Omics-Based Clinical Trials Market?
Omics-based clinical trials refer to clinical studies that incorporate omics technologies such as genomics, proteomics, transcriptomics, metabolomics, and epigenomics to understand disease mechanisms, identify biomarkers, predict therapeutic responses, and personalize medical treatments. These trials use high-throughput data from biological molecules to guide patient selection, stratification, and treatment optimization, ultimately aiming to improve outcomes and reduce adverse effects through precision medicine approaches. The main types of omics-based clinical trials are genomics, proteomics, metabolomics, and transcriptomics. Genomics focuses on analyzing an organism’s entire DNA sequence to understand gene structure, function, and their role in health and disease. These trials progress through phases such as phase I, phase II, phase III, and phase IV and typically follow study designs such as interventional studies, observational studies, and expanded access studies. They are widely applied in areas such as oncology, cardiovascular, neurology, infectious diseases, and metabolic disorders, and cater to several end users, including pharmaceutical and biotechnology companies, academic and research institutes, contract research organizations (CROs), and hospitals and diagnostic laboratories.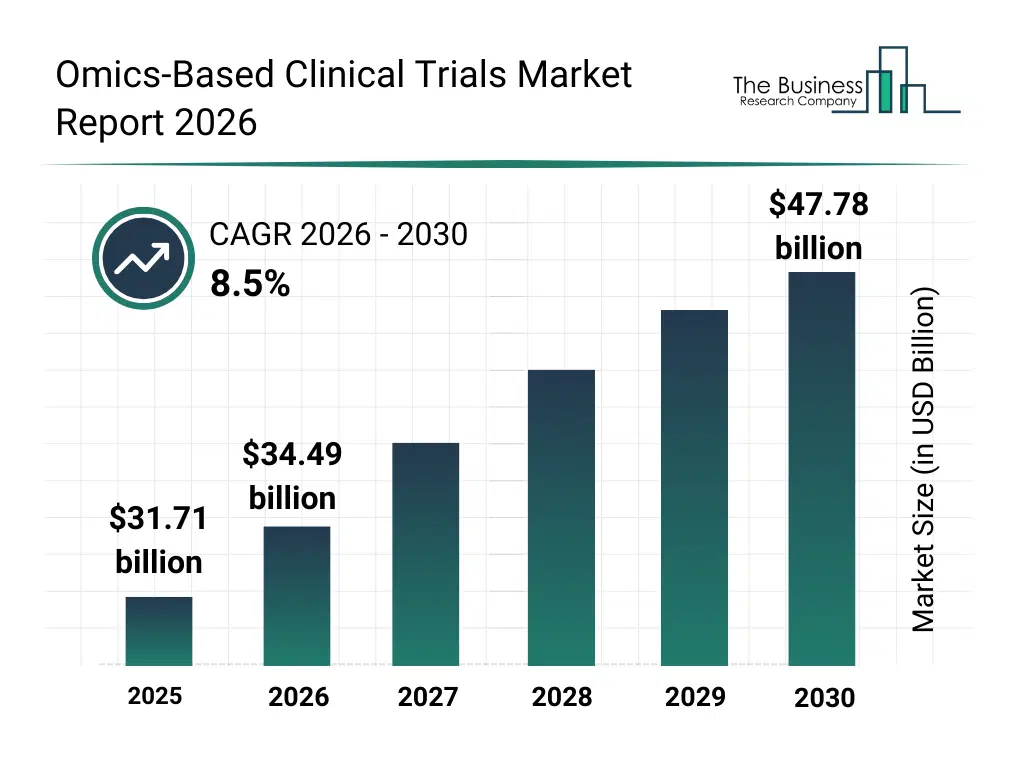
What Is The Omics-Based Clinical Trials Market Size and Share 2026?
The omics-based clinical trials market size has grown strongly in recent years. It will grow from $31.71 billion in 2025 to $34.49 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to increasing complexity of clinical trial designs, growth in genomic and proteomic research, rising demand for personalized therapies, expansion of clinical research infrastructure, increasing collaboration between pharma and academic institutions.What Is The Omics-Based Clinical Trials Market Growth Forecast?
The omics-based clinical trials market size is expected to see strong growth in the next few years. It will grow to $47.78 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to increasing adoption of precision medicine approaches, rising investments in data-driven clinical trials, expansion of ai-enabled patient recruitment, growing regulatory acceptance of omics-based endpoints, increasing demand for efficient trial outcomes. Major trends in the forecast period include increasing adoption of biomarker-driven trial designs, rising use of multi-omics patient stratification, growing integration of real-world evidence analytics, expansion of precision oncology clinical trials, enhanced focus on data-centric trial optimization.Global Omics-Based Clinical Trials Market Segmentation
1) By Type: Genomics, Proteomics, Metabolomics, Transcriptomics 2) By Phase: Phase I, Phase II, Phase III, Phase IV 3) By Study Design: Interventional Studies, Observational Studies, Expanded Access Studies 4) By Application: Oncology, Cardiovascular, Neurology, Infectious Diseases, Metabolic Disorders 5) By End-User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Research Organizations (CROs), Hospitals And Diagnostic Laboratories Subsegments: 1) By Genomics: Whole Genome Sequencing, Whole Exome Sequencing, Targeted Gene Sequencing 2) By Proteomics: Protein Expression Profiling, Post-Translational Modifications Analysis, Protein-Protein Interaction Mapping 3) By Metabolomics: Targeted Metabolomics, Untargeted Metabolomics, Metabolic Flux Analysis 4) By Transcriptomics: Microarray Analysis, Single-Cell Transcriptomics, Gene Expression ProfilingWhat Is The Driver Of The Omics-Based Clinical Trials Market?
The increasing demand for personalized medicine is expected to propel the growth of the omics-based clinical trials market going forward. Personalized medicine refers to a medical approach that tailors disease prevention, diagnosis, and treatment to the individual characteristics of each patient. Personalized medicine demand is rising due to advances in genomic technologies that allow for faster, cost-effective analysis of individual genetic profiles to guide targeted treatments. Omics-based clinical trials support personalized medicine by enabling the identification of individual genetic, proteomic, and metabolic profiles, thereby allowing for more targeted and effective treatment strategies tailored to each patient. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit research organization, the US Food and Drug Administration approved 16 new personalized treatments for rare disease patients in 2023, up from six approvals in 2022, with the 2023 approvals also encompassing seven oncology therapies and three treatments targeting various other conditions. Therefore, the increasing demand for personalized medicine is driving the growth of the omics-based clinical trials industry.Key Players In The Global Omics-Based Clinical Trials Market
Major companies operating in the omics-based clinical trials market are Pfizer Inc., F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., GlaxoSmithKline Plc, Eli Lilly and Company, Novo Nordisk A/S, BioNTech SE, IQVIA Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, Syneos Health Inc., Illumina Inc., Charles River Laboratories International Inc., Parexel International Corporation, Bruker Corporation, Fulgent Genetics Inc., Signios Biosciences, BioAro Inc.Global Omics-Based Clinical Trials Market Trends and Insights
Major companies operating in the omics-based clinical trials market are focusing on innovative solutions, such as HiFi sequencing technology, to enhance the accuracy, speed, and depth of multi-omics data analysis, ultimately improving trial outcomes and personalized treatment strategies. HiFi sequencing refers to a next-generation sequencing (NGS) technology that produces long, highly accurate reads using circular consensus sequencing (CCS), enabling comprehensive and precise analysis of genomes, transcriptomes, and epigenomes. For instance, in October 2024, Pacific Biosciences of California Inc., a US-based biotechnology company, in collaboration with the Agency for Science, Technology, and Research (A*STAR), a Singapore-based research and product development agency, and Macrogen Inc., a South Korea-based biotechnology company, launched a state-of-the-art lab to accelerate genomics innovation in Singapore with cutting-edge long-read sequencing technology. The facility enables researchers to conduct advanced genomic studies with greater precision and speed. It supports a wide range of applications, including whole genome sequencing, transcriptomics, epigenetics, and metagenomics. The lab also fosters collaboration among global scientists and local institutions, aiming to accelerate discoveries in personalized medicine, rare disease diagnostics, and precision oncology. Additionally, it offers training programs and shared access to high-throughput sequencing infrastructure, promoting capacity-building and innovation within the Asia-Pacific region.What Are Latest Mergers And Acquisitions In The Omics-Based Clinical Trials Market?
In June 2025, Bruker Corporation, a US-based manufacturer of scientific instruments and diagnostic tests, acquired Biocrates Life Sciences AG for an undisclosed amount. With this acquisition, Bruker Corporation aims to enhance its multiomics solutions by integrating Biocrates mass spectrometry-based metabolomics technologies to deliver standardized, reproducible, and comprehensive quantitative workflows for life sciences and clinical research. Biocrates Life Sciences AG is an Austria-based biotechnology company that offers omics-based clinical trial solutions.Regional Outlook
North America was the largest region in the omics-based clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Omics-Based Clinical Trials Market?
The omics-based clinical trials market consists of revenues earned by entities by providing services such as genomics analysis, transcriptomics profiling, proteomics studies, and metabolomics assessments. The market value includes the value of related goods sold by the service provider or included within the service offering. The omics-based clinical trials market also includes sales of next-generation sequencing (NGS) platforms, mass spectrometry instruments, microarrays, bioinformatics software, reagents, and kits. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Omics-Based Clinical Trials Market Report 2026?
The omics-based clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the omics-based clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Omics-Based Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $34.49 billion |
| Revenue Forecast In 2035 | $47.78 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Phase, Study Design, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., GlaxoSmithKline Plc, Eli Lilly and Company, Novo Nordisk A/S, BioNTech SE, IQVIA Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, Syneos Health Inc., Illumina Inc., Charles River Laboratories International Inc., Parexel International Corporation, Bruker Corporation, Fulgent Genetics Inc., Signios Biosciences, BioAro Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Omics-Based Clinical Trials market was valued at $31.71 billion in 2025, increased to $34.49 billion in 2026, and is projected to reach $47.78 billion by 2030.
request a sample hereThe global Omics-Based Clinical Trials market is expected to grow at a CAGR of 8.5% from 2026 to 2035 to reach $47.78 billion by 2035.
request a sample hereSome Key Players in the Omics-Based Clinical Trials market Include, Pfizer Inc., F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., GlaxoSmithKline Plc, Eli Lilly and Company, Novo Nordisk A/S, BioNTech SE, IQVIA Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, Syneos Health Inc., Illumina Inc., Charles River Laboratories International Inc., Parexel International Corporation, Bruker Corporation, Fulgent Genetics Inc., Signios Biosciences, BioAro Inc. .
request a sample hereMajor trend in this market includes: Advancements In HiFi Sequencing Boosting Trial Precision. For further insights on this market.
request a sample hereNorth America was the largest region in the omics-based clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the omics-based clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here