
Recombinant Proteins Manufacturing Services Market Report 2026
Global Outlook – By Service Type (Process Development, Upstream Manufacturing, Downstream Manufacturing, Analytical Development and Quality Control, Finish And Lyophilization), By Protein Type (Monoclonal Antibodies And Antibody Fragments, Vaccines And Viral Proteins, Enzymes And Growth Factors, Hormones And Cytokines, Other Therapeutic And Diagnostic Proteins), By Host Cell (Mammalian Cells, Bacterial Cells, Insect Cells, Yeast And Fungi, Other Host Cell), By Application (Therapeutics, Research And Development, Diagnostics, Other Applications), By End-User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes) – Market Size, Trends, Strategies, and Forecast to 2035
Recombinant Proteins Manufacturing Services Market Overview
• Recombinant Proteins Manufacturing Services market size has reached to $3.7 billion in 2025 • Expected to grow to $7.12 billion in 2030 at a compound annual growth rate (CAGR) of 14% • Growth Driver: Surge In Biopharmaceuticals And Biosimilars Fueling Market Growth Due To Rising Biologic Drug Approvals, Expanding Chronic Disease Burden, And Increasing Healthcare Spending On Biologics • Market Trend: Advancements In Scalable Mammalian Protein Expression Enhancing Recombinant Protein Manufacturing Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Recombinant Proteins Manufacturing Services Market?
Recombinant proteins manufacturing services refer to outsourced solutions that support the production of proteins created through Recombinant DNA Technology under controlled laboratory and industrial conditions. These services cover activities such as gene cloning, protein expression, purification, and quality testing to meet research, clinical, or commercial requirements. They help organizations access specialized expertise, infrastructure, and regulatory-compliant processes without building in-house manufacturing capabilities. The main service types of recombinant proteins manufacturing services are process development, upstream manufacturing, downstream manufacturing, analytical development and quality control, and finish and lyophilization. Process development refers to the design and optimization of production processes to efficiently produce recombinant proteins with desired quality and yield. These services cater to different protein types, including monoclonal antibodies and antibody fragments, vaccines and viral proteins, enzymes and growth factors, hormones and cytokines, and other therapeutic and diagnostic proteins, and utilize various host cells, such as mammalian cells, bacterial cells, insect cells, yeast and fungi, and other host cells. They are applied across multiple applications, including therapeutics, research and development, diagnostics, and other applications, and serve end-users, such as pharmaceutical and biotechnology companies and academic and research institutes.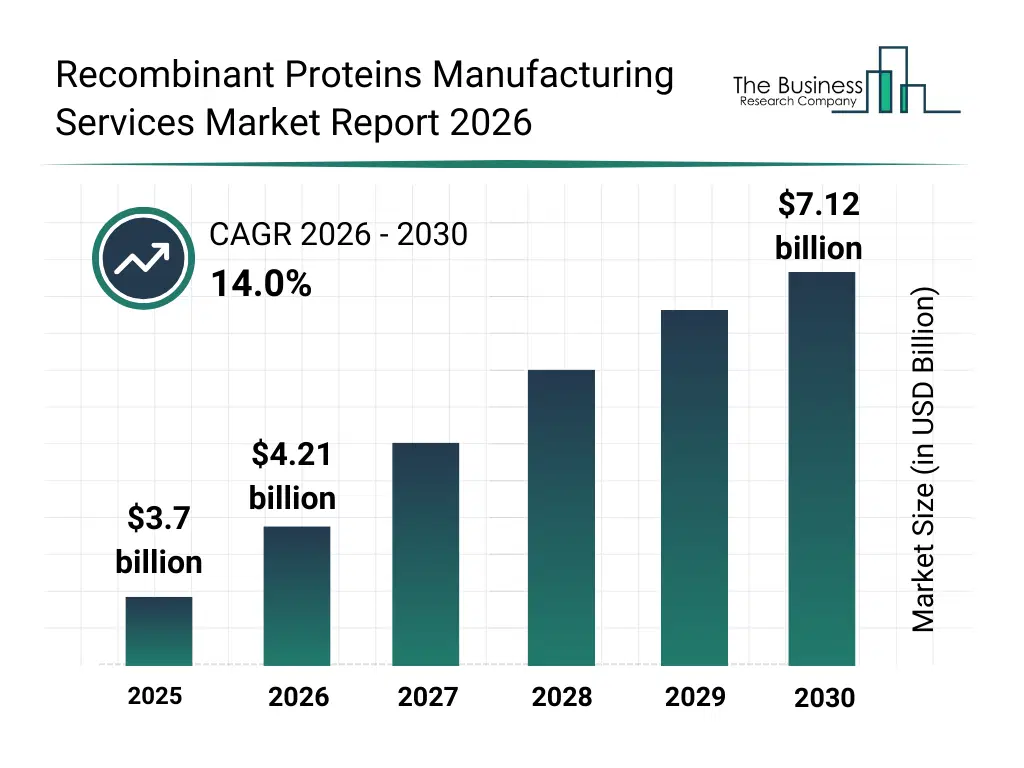
What Is The Recombinant Proteins Manufacturing Services Market Size and Share 2026?
The recombinant proteins manufacturing services market size has grown rapidly in recent years. It will grow from $3.7 billion in 2025 to $4.21 billion in 2026 at a compound annual growth rate (CAGR) of 13.8%. The growth in the historic period can be attributed to growth in biologics research programs, increasing academic protein studies, expansion of contract manufacturing models, rising monoclonal antibody development, historic growth in biotech startups.What Is The Recombinant Proteins Manufacturing Services Market Growth Forecast?
The recombinant proteins manufacturing services market size is expected to see rapid growth in the next few years. It will grow to $7.12 billion in 2030 at a compound annual growth rate (CAGR) of 14.0%. The growth in the forecast period can be attributed to increasing pipeline of protein therapeutics, rising outsourcing by pharma companies, higher demand for scalable bioprocessing, expansion of clinical stage biologics, growing need for regulatory compliant facilities. Major trends in the forecast period include expansion of outsourced protein production, growth in custom expression services, rising demand for GMP protein manufacturing, integrated end to end service models, higher use of single use bioprocess systems.Global Recombinant Proteins Manufacturing Services Market Segmentation
1) By Service Type: Process Development; Upstream Manufacturing; Downstream Manufacturing; Analytical Development and Quality Control; Finish And Lyophilization 2) By Protein Type: Monoclonal Antibodies And Antibody Fragments; Vaccines And Viral Proteins; Enzymes And Growth Factors; Hormones And Cytokines; Other Therapeutic And Diagnostic Proteins 3) By Host Cell: Mammalian Cells; Bacterial Cells; Insect Cells; Yeast And Fungi; Other Host Cell 4) By Application: Therapeutics; Research And Development; Diagnostics; Other Applications 5) By End-User: Pharmaceutical And Biotechnology Companies; Academic And Research Institutes Subsegments: 1) By Process Development: Cell Line Development; Expression System Optimization; Process Scale Up; Process Validation 2) By Upstream Manufacturing: Microbial Fermentation; Mammalian Cell Culture; Insect Cell Culture; Media And Feed Optimization 3) By Downstream Manufacturing: Protein Purification; Chromatography Processing; Filtration And Concentration; Viral Clearance 4) By Analytical Development And Quality Control: Method Development; Stability Testing; Purity And Potency Testing; Batch Release Testing 5) By Finish And Lyophilization: Sterile Filling; Freeze Drying; Packaging And Labeling; Storage And Distribution PreparationWhat Is The Driver Of The Recombinant Proteins Manufacturing Services Market?
The growing demand for biopharmaceuticals and biosimilars is expected to propel the growth of the recombinant proteins manufacturing services market going forward. Biopharmaceuticals and biosimilars refer to biologic medicines and their highly similar versions that are used to treat chronic, rare, and complex diseases, including cancer, autoimmune disorders, and metabolic conditions. The demand for biopharmaceuticals and biosimilars is increasing due to the rising prevalence of chronic and life-threatening diseases, advances in biologic drug development, and expanding patient access to targeted therapies across major healthcare markets. The recombinant proteins manufacturing services supports the development and commercialization of biopharmaceuticals and biosimilars by providing scalable, high-quality, and regulatory-compliant production of protein-based drugs, enabling pharmaceutical and biotechnology companies to accelerate development timelines and maintain consistent supply. For instance, according to GOV.UK, a UK-based government website, companies within the biopharmaceutical subsector reported a total turnover of $135.5 billion (£98.9 billion) during the 2023/2024 period, an increase of 15% compared to last year. Therefore, the growing demand for biopharmaceuticals and biosimilars is driving the growth of the recombinant proteins manufacturing services industry.Key Players In The Global Recombinant Proteins Manufacturing Services Market
Major companies operating in the recombinant proteins manufacturing services market are Thermo Fisher Scientific Inc., Lonza Group Ltd., Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Recipharm AB, Bio-Techne Corporation, FUJIFILM Diosynth Biotechnologies, AGC Biologics A/S, WuXi Biologics (Cayman) Inc., KBI Biopharma Inc., GenScript Biotech Corporation, Rentschler Biopharma SE, Abzena Ltd., ProBioGen AG, NorthX Biologics AB, Creative Biolabs Inc., evitria AG, Boster Biological Technology Co. Ltd., Icosagen Group ASGlobal Recombinant Proteins Manufacturing Services Market Trends and Insights
Major companies operating in the recombinant proteins manufacturing services market are focusing on technological advancements in high-yield protein expression and optimization techniques, such as research-grade and scalable mammalian recombinant protein manufacturing capabilities, to improve production efficiency, enhance protein quality, shorten development timelines, and increase flexibility in meeting diverse biopharmaceutical development requirements. Research-grade and scalable mammalian recombinant protein manufacturing capabilities refer to advancements in cell culture systems, expression technologies, and process controls that enable the efficient production of high-quality recombinant proteins at small research scales while allowing smooth transition to larger manufacturing volumes. For instance, in September 2024, Axio BioPharma, a U.S.-based biotechnology company, launched recombinant protein manufacturing services to support research-grade monoclonal antibody production and scalable mammalian protein expression solutions. The recombinant protein manufacturing services platform offers research-grade and scalable mammalian protein production capabilities that support high-quality monoclonal antibody and recombinant protein expression for early-stage development. With flexible production volumes ranging from small laboratory-scale batches to scalable systems, the platform enables reliable protein output while maintaining consistency and biological activity.What Are Latest Mergers And Acquisitions In The Recombinant Proteins Manufacturing Services Market?
In April 2024, Sino Biological Inc., a China-based provider of recombinant proteins, antibodies, antigens, assay kits, and life science research reagents, acquired SignalChem Biotech Inc. for an undisclosed amount. With this acquisition, Sino Biological aimed to strengthen its global presence and expand its product portfolio by enhancing its capabilities in kinase biology, signal transduction research, and drug discovery support, while deepening access to specialized enzymatic tools for pharmaceutical and biotechnology customers. SignalChem Biotech Inc. is a Canada-based company that offers custom recombinant protein manufacturing services as part of its expertise in bioactive enzymes and cell signaling proteins.Regional Insights
North America was the largest region in the recombinant proteins manufacturing services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Recombinant Proteins Manufacturing Services Market?
The recombinant proteins manufacturing services market includes revenues earned by entities through cell line development, protein expression and purification, quality control and analytical testing, regulatory compliance and documentation, and custom manufacturing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Recombinant Proteins Manufacturing Services Market Report 2026?
The recombinant proteins manufacturing services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the recombinant proteins manufacturing services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Recombinant Proteins Manufacturing Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.21 billion |
| Revenue Forecast In 2035 | $7.12 billion |
| Growth Rate | CAGR of 13.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Protein Type, Host Cell, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Lonza Group Ltd., Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Recipharm AB, Bio-Techne Corporation, FUJIFILM Diosynth Biotechnologies, AGC Biologics A/S, WuXi Biologics (Cayman) Inc., KBI Biopharma Inc., GenScript Biotech Corporation, Rentschler Biopharma SE, Abzena Ltd., ProBioGen AG, NorthX Biologics AB, Creative Biolabs Inc., evitria AG, Boster Biological Technology Co. Ltd., Icosagen Group AS |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
