
Rescue Airway Devices Market Report 2026
Global Outlook – By Product Type (Supraglottic Airway Devices (SGA), Laryngeal Mask Airways (LMA), Intubating Laryngeal Mask Airways (ILMA), Laryngeal Tubes, I-Gel Airway Devices, Other Product Types), By Material (Silicone, Polyvinyl Chloride (PVC), Thermoplastic Elastomers (TPE), Other Materials), By Anatomical Placement (Simple Adjuncts (Upper Airway), Supraglottic Or Extraglottic Airway Devices (SADs Or EGDs), Infraglottic Or Tracheal Devices (Definitive Airway), Airway Access Or Visualization Tools, Ventilation Or Resuscitation Devices), By End User (Hospitals, Ambulatory Surgery Centers (ASC), Clinics, Emergency Medical Services (EMS) Providers, Military And Government Medical Units, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Rescue Airway Devices Market Overview
• Rescue Airway Devices market size has reached to $2.76 billion in 2025 • Expected to grow to $4.49 billion in 2030 at a compound annual growth rate (CAGR) of 10.2% • Growth Driver: Rising Number Of Emergency Cases Fueling The Growth Of The Market Due To Increasing Need For Rapid And Effective Airway Management • Market Trend: Advanced Visualization Tools Enhance Intubation Accuracy And Patient Safety • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Rescue Airway Devices Market?
Rescue airway devices are medical tools designed to maintain or restore a patient’s airway during emergencies, such as respiratory distress or obstruction. Their primary purpose is to ensure adequate ventilation and oxygenation, prevent airway collapse, and support life until definitive airway management can be established. The main product types of rescue airway devices are supraglottic airway devices, laryngeal mask airways, intubating laryngeal mask airways, laryngeal tubes, i-gel airway devices, and other supraglottic devices. Supraglottic airway devices are emergency airway management tools designed to maintain airway patency without tracheal intubation during resuscitation and acute care scenarios. These devices are manufactured using materials such as silicone, polyvinyl chloride, thermoplastic elastomers, and others, designed for anatomical placement including simple adjuncts, supraglottic or extraglottic airway devices, infraglottic or tracheal devices, airway access or visualization tools, and ventilation or resuscitation devices, among end users such as hospitals, ambulatory surgery centers, clinics, emergency medical services providers, military and government medical units, and others.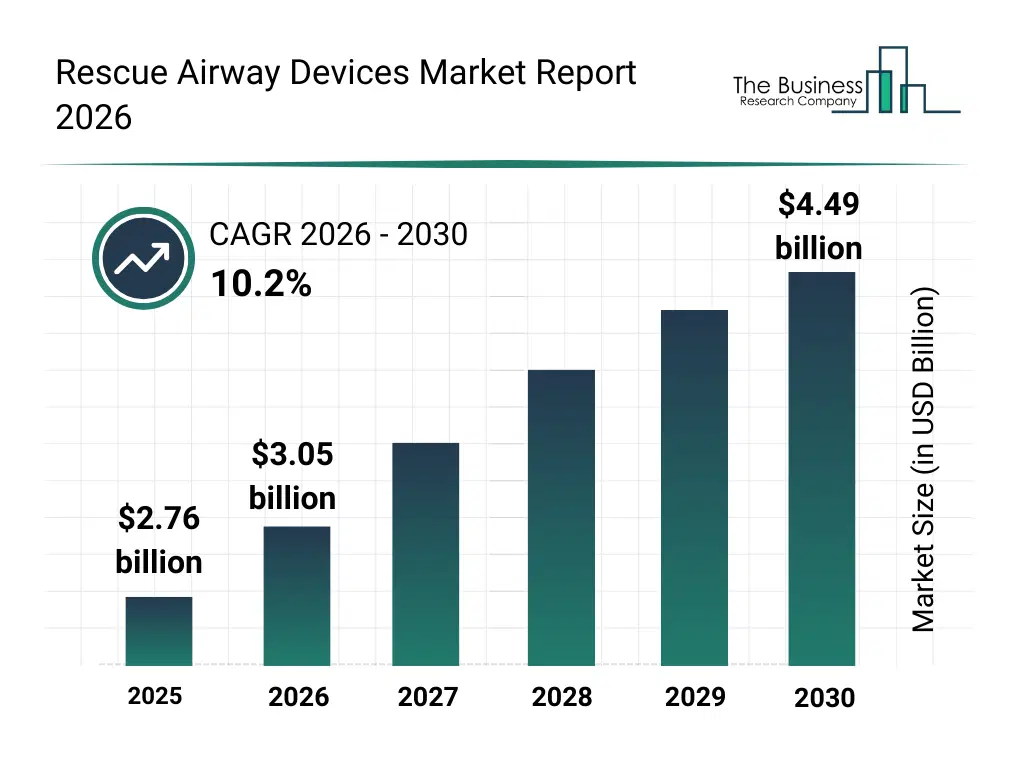
What Is The Rescue Airway Devices Market Size and Share 2026?
The rescue airway devices market size has grown rapidly in recent years. It will grow from $2.76 billion in 2025 to $3.05 billion in 2026 at a compound annual growth rate (CAGR) of 10.4%. The growth in the historic period can be attributed to increasing mobile connectivity, rising sensor integration, expanding iot adoption, growing data analytics usage, and increasing indoor positioning demandWhat Is The Rescue Airway Devices Market Growth Forecast?
The rescue airway devices market size is expected to see rapid growth in the next few years. It will grow to $4.49 billion in 2030 at a compound annual growth rate (CAGR) of 10.2%. The growth in the forecast period can be attributed to growing context aware automation, rising real time localization needs, increasing smart infrastructure expansion, expanding ai enabled processing, and growing multi modal sensing adoption. Major trends in the forecast period include advancements in adaptive sensing, innovations in ambient automation, developments in multimodal fusion, research in predictive intelligence, and technology upgrades in spatial computingGlobal Rescue Airway Devices Market Segmentation
1) By Product Type: Supraglottic Airway Devices (SGA), Laryngeal Mask Airways (LMA), Intubating Laryngeal Mask Airways (ILMA), Laryngeal Tubes, I-Gel Airway Devices, Other Product Types 2) By Material: Silicone, Polyvinyl Chloride (PVC), Thermoplastic Elastomers (TPE), Other Materials 3) By Anatomical Placement: Simple Adjuncts (Upper Airway), Supraglottic Or Extraglottic Airway Devices (SADs Or EGDs), Infraglottic Or Tracheal Devices (Definitive Airway), Airway Access Or Visualization Tools, Ventilation Or Resuscitation Devices 4) By End User: Hospitals, Ambulatory Surgery Centers (ASC), Clinics, Emergency Medical Services (EMS) Providers, Military And Government Medical Units, Other End Users Subsegments: 1) By Supraglottic Airway Devices (SGA): First Generation Supraglottic Devices, Second Generation Supraglottic Devices, Reusable Supraglottic Devices, Disposable Supraglottic Devices 2) By Laryngeal Mask Airways (LMA): Reusable Laryngeal Mask Airways, Disposable Laryngeal Mask Airways, First Generation Laryngeal Mask Airways, Second Generation Laryngeal Mask Airways 3) By Intubating Laryngeal Mask Airways (ILMA): Reusable Intubating Laryngeal Mask Airways, Disposable Intubating Laryngeal Mask Airways, Silicone Intubating Mask Airways, Polyvinyl Chloride Intubating Mask Airways 4) By Laryngeal Tubes: Single Lumen Laryngeal Tubes, Dual Lumen Laryngeal Tubes, Reusable Laryngeal Tubes, Disposable Laryngeal Tubes 5) By I-Gel Airway Devices: Standard I-Gel Airway Devices, Pediatric I-Gel Airway Devices, Adult I-Gel Airway Devices, Disposable I-Gel Airway Devices 6) By Other Product Types: Pharyngeal Airway Devices, Oropharyngeal Airway Devices, Nasopharyngeal Airway Devices, Cuffed Pharyngeal Airway DevicesWhat Are The Drivers Of The Rescue Airway Devices Market?
The rising number of emergency cases is expected to propel the growth of the rescue airway devices market going forward. Emergency cases refer to acute medical situations such as road accidents, cardiac arrests, respiratory failure, and severe injuries that require immediate lifesaving interventions. The number of emergency cases is rising due to growing road traffic accidents and violence-related injuries in increasingly urbanized and motorized populations which drives people to seek urgent hospital care. Rescue airway devices help manage emergency cases by enabling quick, reliable, and effective airway management in pre-hospital and hospital settings, ensuring adequate oxygenation and reducing the risk of complications during emergency care. For instance, in October 2025, according to the NHS England, a UK-based government health agency, there were around 401,800 admissions to hospital in an emergency via all accident and emergency departments in September 2025, averaging 13,400 people per day, with a further 133,800 emergency admissions that did not come via accident and emergency, averaging 4,500 per day. Therefore, the rising number of emergency cases is driving the growth of the rescue airway devices industry. The rising incidence of cardiac arrest cases is expected to propel the growth of the rescue airway devices market going forward. Cardiac arrest refers to a sudden loss of blood flow resulting from the failure of the heart to pump effectively, often caused by an electrical malfunction in the heart. The incidence of cardiac arrest cases is rising largely due to growing rates of heart disease, obesity, hypertension, and diabetes, which contribute to weakened heart function and abnormal heart rhythms. Rescue airway devices help manage cardiac arrest cases by enabling rapid airway control and effective oxygenation during emergency resuscitation efforts. They support better survival outcomes by maintaining uninterrupted ventilation until definitive care is provided. For instance, in February 2025, according to Hato Hone St John, a New Zealand-based charitable organization providing ambulance services, 2,547 people were treated for cardiac arrest in 2024, representing a 3.6 percent increase from 2023, with 72 percent of cases occurring at home. Therefore, the rising incidence of cardiac arrest cases is driving the growth of the rescue airway devices industry.Key Players In The Global Rescue Airway Devices Market
Major companies operating in the rescue airway devices market are Medtronic plc, Medline Industries LP, GE HealthCare Technologies Inc., Becton, Dickinson and Company, Olympus Corporation, Smiths Group plc, Drägerwerk AG & Co. KGaA, Teleflex Incorporated, Cook Medical LLC, Nihon Kohden Corporation, Fisher & Paykel Healthcare Corporation Limited, Ambu A/S, Armstrong Medical Ltd., Vyaire Medical Inc., Vygon S.A., Intersurgical Ltd., Mercury Enterprises LLC, SunMed LLC, Verathon Inc., SourceMark LLCGlobal Rescue Airway Devices Market Trends and Insights
Major companies operating in the rescue airway devices market are focusing on developing advanced visual technologies, such as integrated video laryngoscopy solutions, to improve clinician visibility, procedural accuracy, and patient safety during airway management in both routine and complex clinical scenarios. Integrated video laryngoscopy solutions refer to devices that combine high-resolution video imaging with ergonomic controls and software connectivity, enabling clinicians to visually guide intubation in real time and share imaging data seamlessly with hospital information systems. For instance, in January 2025, Ambu A/S, a Denmark-based medical technology company, launched the Ambu SureSight Connect video laryngoscopy solution, designed to facilitate efficient endotracheal intubation in operating rooms and intensive care units. The solution integrates with Ambu’s digital platforms, including Ambu aView 2 Advance and Ambu aBox 2, to simplify workflow, reduce task complexity, and provide clinicians with comprehensive airway visualization tools. It is the only airway visualization offering that features a one-lung ventilation solution and includes all devices recommended for airway assessment and intubation, broadening its utility across clinical settings.Regional Insights
North America was the largest region in the rescue airway devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Rescue Airway Devices Market?
The rescue airway devices market consists of sales of endotracheal tubes, laryngeal mask airways (LMAs), oropharyngeal and nasopharyngeal airways, supraglottic airway devices, and tracheostomy kits. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Rescue Airway Devices Market Report 2026?
The rescue airway devices market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the rescue airway devices industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Rescue Airway Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.05 billion |
| Revenue Forecast In 2035 | $4.49 billion |
| Growth Rate | CAGR of 10.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Material, Anatomical Placement, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic plc, Medline Industries LP, GE HealthCare Technologies Inc., Becton, Dickinson and Company, Olympus Corporation, Smiths Group plc, Drägerwerk AG & Co. KGaA, Teleflex Incorporated, Cook Medical LLC, Nihon Kohden Corporation, Fisher & Paykel Healthcare Corporation Limited, Ambu A/S, Armstrong Medical Ltd., Vyaire Medical Inc., Vygon S.A., Intersurgical Ltd., Mercury Enterprises LLC, SunMed LLC, Verathon Inc., SourceMark LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
1) By Product Type: Supraglottic Airway Devices (SGA), Laryngeal Mask Airways (LMA), Intubating Laryngeal Mask Airways (ILMA), Laryngeal Tubes, I-Gel Airway Devices, Other Product Types
2) By Material: Silicone, Polyvinyl Chloride (PVC), Thermoplastic Elastomers (TPE), Other Materials
3) By Anatomical Placement: Simple Adjuncts (Upper Airway), Supraglottic Or Extraglottic Airway Devices (SADs Or EGDs), Infraglottic Or Tracheal Devices (Definitive Airway), Airway Access Or Visualization Tools, Ventilation Or Resuscitation Devices
4) By End User: Hospitals, Ambulatory Surgery Centers (ASC), Clinics, Emergency Medical Services (EMS) Providers, Military And Government Medical Units, Other End Users Subsegments:
1) By Supraglottic Airway Devices (SGA): First Generation Supraglottic Devices, Second Generation Supraglottic Devices, Reusable Supraglottic Devices, Disposable Supraglottic Devices
2) By Laryngeal Mask Airways (LMA): Reusable Laryngeal Mask Airways, Disposable Laryngeal Mask Airways, First Generation Laryngeal Mask Airways, Second Generation Laryngeal Mask Airways
3) By Intubating Laryngeal Mask Airways (ILMA): Reusable Intubating Laryngeal Mask Airways, Disposable Intubating Laryngeal Mask Airways, Silicone Intubating Mask Airways, Polyvinyl Chloride Intubating Mask Airways
4) By Laryngeal Tubes: Single Lumen Laryngeal Tubes, Dual Lumen Laryngeal Tubes, Reusable Laryngeal Tubes, Disposable Laryngeal Tubes
5) By I-Gel Airway Devices: Standard I-Gel Airway Devices, Pediatric I-Gel Airway Devices, Adult I-Gel Airway Devices, Disposable I-Gel Airway Devices
6) By Other Product Types: Pharyngeal Airway Devices, Oropharyngeal Airway Devices, Nasopharyngeal Airway Devices, Cuffed Pharyngeal Airway Devices
