
Respiratory Endotherapy Devices Market Report 2026
Global Outlook – By Product Type (Bronchoscopes, Endobronchial Ultrasound Systems, Biopsy Forceps, Guidewires, Catheters, Airway Stents, Endobronchial Valves, Ablation Devices, Other Endotherapy Accessories), By Diagnostic Devices (Trans Bronchial Aspiration, Biopsy Forceps, Cytology Brushes, Other Diagnostic Devices), By Patient Type (Adult Patients, Pediatric Patients), By Specialist (Hospitals, Diagnostic Centers, Ambulatory Surgical Centers), By End User (Peripheral Vascular Intervention, Endovascular Interventions, Cardiac, Interventional Radiology, Venous, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Respiratory Endotherapy Devices Market Overview
• Respiratory Endotherapy Devices market size has reached to $3.32 billion in 2025 • Expected to grow to $4.66 billion in 2030 at a compound annual growth rate (CAGR) of 7.1% • Growth Driver: Rising Adoption Of Robotic-Assisted Bronchoscopy Fueling The Growth Of The Market Due To Enhanced Precision And Minimally Invasive Pulmonary Interventions • Market Trend: Advancements In Minimally Invasive Endobronchial Valve–Based Lung Volume Reduction Therapies • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Respiratory Endotherapy Devices Market?
A respiratory endotherapy devices are medical devices designed to diagnose and treat respiratory conditions through minimally invasive procedures performed inside the airways and lungs. These devices are used to visualize, access, and deliver therapeutic interventions within the respiratory tract to manage conditions such as lung cancer, airway obstruction, chronic obstructive pulmonary disease, and pulmonary infections. Respiratory endotherapy devices are intended to improve patient outcomes by enabling precise, targeted treatment while reducing the need for open surgical procedures. The main product types of respiratory endotherapy devices include bronchoscopes, endobronchial ultrasound systems, biopsy forceps, guidewires, catheters, airway stents, endobronchial valves, ablation devices, and other endotherapy accessories. Bronchoscopes refer to instruments used to visually examine the airways and perform therapeutic or diagnostic procedures in the respiratory tract. These devices include diagnostic devices such as transbronchial aspiration tools, biopsy forceps, cytology brushes, and other diagnostic devices. They are used for patient types including adult patients and pediatric patients. They are utilized by specialists including hospitals, diagnostic centers, and ambulatory surgical centers, and are applied in procedures such as peripheral vascular intervention, endovascular interventions, cardiac procedures, interventional radiology, venous procedures, and other end-user applications.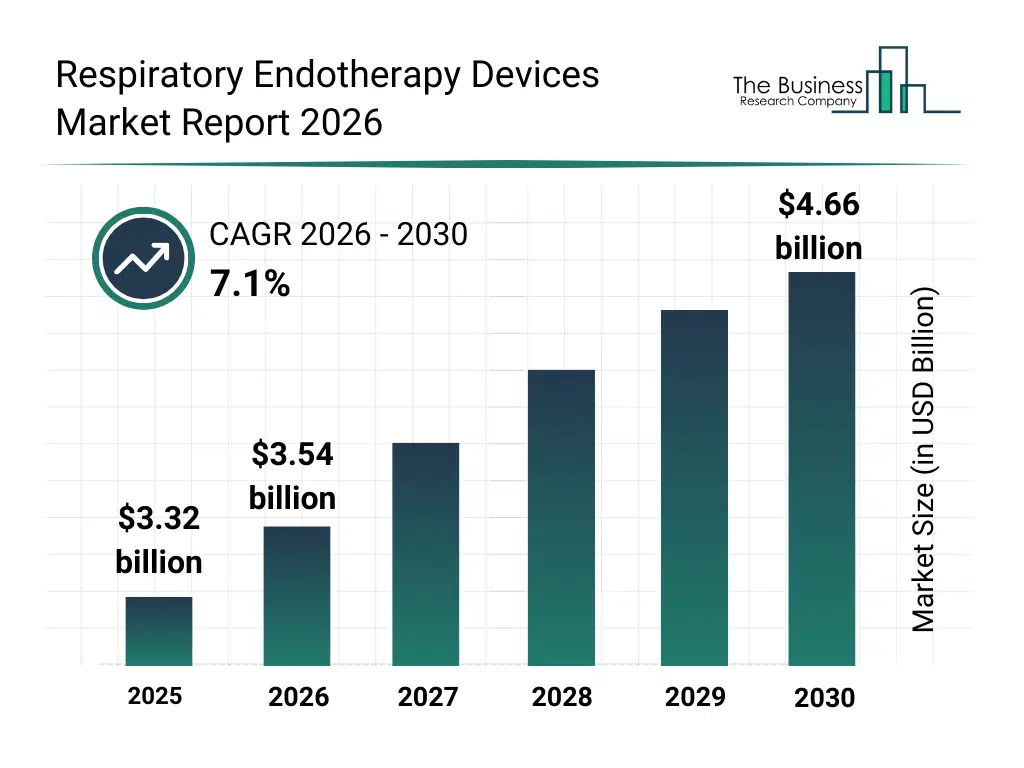
What Is The Respiratory Endotherapy Devices Market Size and Share 2026?
The respiratory endotherapy devices market size has grown strongly in recent years. It will grow from $3.32 billion in 2025 to $3.54 billion in 2026 at a compound annual growth rate (CAGR) of 6.9%. The growth in the historic period can be attributed to rise in chronic respiratory diseases, growth in lung cancer diagnosis, expansion of interventional pulmonology, hospital endoscopy unit growth, demand for minimally invasive procedures.What Is The Respiratory Endotherapy Devices Market Growth Forecast?
The respiratory endotherapy devices market size is expected to see strong growth in the next few years. It will grow to $4.66 billion in 2030 at a compound annual growth rate (CAGR) of 7.1%. The growth in the forecast period can be attributed to increase in early lung screening programs, robotic bronchoscopy adoption, outpatient pulmonary procedures growth, advanced imaging integration, precision airway therapy demand. Major trends in the forecast period include minimally invasive airway intervention devices, disposable bronchoscope adoption, image guided bronchoscopic procedures, advanced endobronchial ablation systems, smart navigation bronchoscopy tools.Global Respiratory Endotherapy Devices Market Segmentation
1) By Product Type: Bronchoscopes; Endobronchial Ultrasound Systems; Biopsy Forceps; Guidewires; Catheters; Airway Stents; Endobronchial Valves; Ablation Devices; Other Endotherapy Accessories 2) By Diagnostic Devices: Trans Bronchial Aspiration; Biopsy Forceps; Cytology Brushes; Other Diagnostic Devices 3) By Patient Type: Adult Patients; Pediatric Patients 4) By Specialist: Hospitals; Diagnostic Centers; Ambulatory Surgical Centers 5) By End User: Peripheral Vascular Intervention; Endovascular Interventions; Cardiac; Interventional Radiology; Venous; Other End Users Subsegments: 1) By Bronchoscopes: Flexible Bronchoscopes; Rigid Bronchoscopes; Video Bronchoscopes; Disposable Bronchoscopes; Therapeutic Bronchoscopes 2) By Endobronchial Ultrasound Systems: Radial Endobronchial Ultrasound Systems; Linear Endobronchial Ultrasound Systems; Portable Endobronchial Ultrasound Systems; Cart Based Endobronchial Ultrasound Systems; High Resolution Endobronchial Ultrasound Systems 3) By Biopsy Forceps: Flexible Biopsy Forceps; Rigid Biopsy Forceps; Disposable Biopsy Forceps; Alligator Jaw Biopsy Forceps; Cup Biopsy Forceps 4) By Guidewires: Hydrophilic Guidewires; Non Coated Guidewires; Steerable Guidewires; Flexible Tip Guidewires; High Strength Guidewires 5) By Catheters: Balloon Catheters; Aspiration Catheters; Delivery Catheters; Diagnostic Catheters; Therapeutic Catheters 6) By Airway Stents: Silicone Airway Stents; Metallic Airway Stents; Covered Airway Stents; Uncovered Airway Stents; Expandable Airway Stents 7) By Endobronchial Valves: One Way Endobronchial Valves; Permanent Endobronchial Valves; Temporary Endobronchial Valves; Adjustable Endobronchial Valves; Implantable Endobronchial Valves 8) By Ablation Devices: Radiofrequency Ablation Devices; Cryoablation Devices; Laser Ablation Devices; Thermal Ablation Devices; Microwave Ablation Devices 9) By Other Endotherapy Accessories: Suction Tubes; Specimen Collection Traps; Bronchoscopy Brushes; Biopsy Needles; Airway DilatorsWhat Is The Driver Of The Respiratory Endotherapy Devices Market?
The rising adoption of robotic-assisted bronchoscopy delivery is expected to propel the growth of the respiratory endotherapy devices market going forward. Robotic-assisted bronchoscopy systems are advanced technologies that enable precise navigation within the respiratory tract, improving diagnostic accuracy and therapeutic outcomes. The demand for these systems is increasing as healthcare providers seek minimally invasive solutions that reduce procedure time, enhance patient safety, and improve clinical efficiency. Respiratory endotherapy devices equipped with robotic assistance support complex interventions by providing real-time imaging, better maneuverability, and enhanced control. For instance, in September 2025, according to the European Respiratory Society International Congress, a Switzerland-based membership organization, the use of robotic-assisted bronchoscopy with integrated cone beam CT for peripheral lung lesions has risen to 84.6% in 2025, up from just 23.1% with conventional bronchoscopy in 2024. Therefore, the rising adoption of robotic-assisted bronchoscopy is driving the growth of the respiratory endotherapy devices industry.Key Players In The Global Respiratory Endotherapy Devices Market
Major companies operating in the respiratory endotherapy devices market are Johnson & Johnson, Medtronic plc, Siemens Healthineers, Stryker Corporation, GE Healthcare Technologies Inc., Boston Scientific Corporation, Intuitive Surgical Inc., Olympus Corporation, Hoya Corporation, Steris Healthcare, Teleflex Incorporated, Karl Storz SE & Co. KG, CookMedical, Merit Medical Systems Inc., Ambu A/S, Vygon, Richard Wolf GmbH, Vapotherm Inc, Scholly Fiberoptic GmbH, Clarus Medical LLC, ENDO-FLEX GmbH, The Lung Docs Inc., and Broncus Medical Inc.Global Respiratory Endotherapy Devices Market Trends and Insights
Major companies operating in the respiratory endotherapy devices market are focusing on developing advanced solutions, such as endobronchial valves, to minimize treatment risk while achieving targeted lung volume reduction without permanent tissue removal. An endobronchial valve is a minimally invasive implant placed via bronchoscopy into selected airways to block airflow to diseased lung regions, allowing healthier areas to function more effectively. For instance, in February 2024, Olympus Canada Inc., a Canada-based medical technology company, launched the Spiration Valve System for the treatment of severe emphysema. It is an umbrella-shaped device designed to be placed in the most severely diseased lung regions using a minimally invasive bronchoscopic procedure. It uniquely redirects airflow away from hyperinflated lung areas toward healthier tissue, supporting improved respiratory mechanics. As the only endobronchial valve therapy currently available in Canada, it addresses an unmet need in severe emphysema management. Clinical evidence from the EMPROVE trial demonstrates durable benefits through 24 months, including meaningful improvements in lung function, breathlessness, and patient quality of life.What Are Latest Mergers And Acquisitions In The Respiratory Endotherapy Devices Market?
In November 2023, Broncus Medical Inc., a US-based provider of interventional pulmonology devices, acquired Hangzhou Jingliang Medical Technology for an undisclosed amount. Through this acquisition, Broncus Medical aims to enhance its technological capabilities in fiber-optic navigation, flexible catheter systems, and robotic-assisted bronchoscopy, strengthening its product portfolio in respiratory endotherapy and minimally invasive lung procedures. Hangzhou Jingliang Medical Technology is a China-based medical technology company specializing in respiratory endotherapy devices.Regional Insights
North America was the largest region in the respiratory endotherapy devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Respiratory Endotherapy Devices Market?
The respiratory endotherapy devices market consists of sales of bronchoscopes, endobronchial ultrasound systems, biopsy forceps, guidewires, catheters, airway stents, endobronchial valves, ablation devices, and related endoscopic and interventional pulmonology accessories. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Respiratory Endotherapy Devices Market Report 2026?
The respiratory endotherapy devices market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the respiratory endotherapy devices industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Respiratory Endotherapy Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.54 billion |
| Revenue Forecast In 2035 | $4.66 billion |
| Growth Rate | CAGR of 6.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Diagnostic Devices, Patient Type, Specialist, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Medtronic plc, Siemens Healthineers, Stryker Corporation, GE Healthcare Technologies Inc., Boston Scientific Corporation, Intuitive Surgical Inc., Olympus Corporation, Hoya Corporation, Steris Healthcare, Teleflex Incorporated, Karl Storz SE & Co. KG, CookMedical, Merit Medical Systems Inc., Ambu A/S, Vygon, Richard Wolf GmbH, Vapotherm Inc, Scholly Fiberoptic GmbH, Clarus Medical LLC, ENDO-FLEX GmbH, The Lung Docs Inc., and Broncus Medical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
