
Spinal Muscular Atrophy Treatment Market Report 2026
Global Outlook – By Treatment (Medication, Physical Therapy, Surgery, Other Treatments), By Type (Werdnig-Hoffmann Disease, Infant Spinal Muscular Atrophy, Kugelberg-Welander Disease, Adult Spinal Muscular Atrophy), By Route Of Administration (Oral, Parenteral), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Other Distribution Channels), By End User (Hospitals, Homecare, Specialty Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Spinal Muscular Atrophy Treatment Market Overview
• Spinal Muscular Atrophy Treatment market size has reached to $5.1 billion in 2025 • Expected to grow to $10.38 billion in 2030 at a compound annual growth rate (CAGR) of 15.2% • Growth Driver: Growing Focus On Gene Therapy Fuels Market Growth Due To Advances In Technology And Disease Prevalence • Market Trend: Advantages Of Oral SMN2 Splicing Modifier Tablets Over Traditional Treatments • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Spinal Muscular Atrophy Treatment Market?
Spinal muscular atrophy (SMA) treatment refers to medical therapies and interventions aimed at managing or improving muscle strength, motor function, and quality of life in individuals affected by spinal muscular atrophy, a genetic disorder characterized by the progressive loss of motor neurons in the spinal cord and lower brainstem. The main treatment types for spinal muscular atrophy include medication, physical therapy, surgery, and other treatments. Medication helps manage symptoms, enhance motor function, and slow disease progression. These treatments are used for various types of spinal muscular atrophy, such as Werdnig-Hoffmann disease, infantile spinal muscular atrophy, kugelberg-welander disease, and adult-onset spinal muscular atrophy. Drugs are administered through various routes, including oral and parenteral. They are distributed through hospital pharmacies, retail pharmacies, and other distribution channels and are used by various end users such as hospitals, homecare settings, specialty clinics, and others.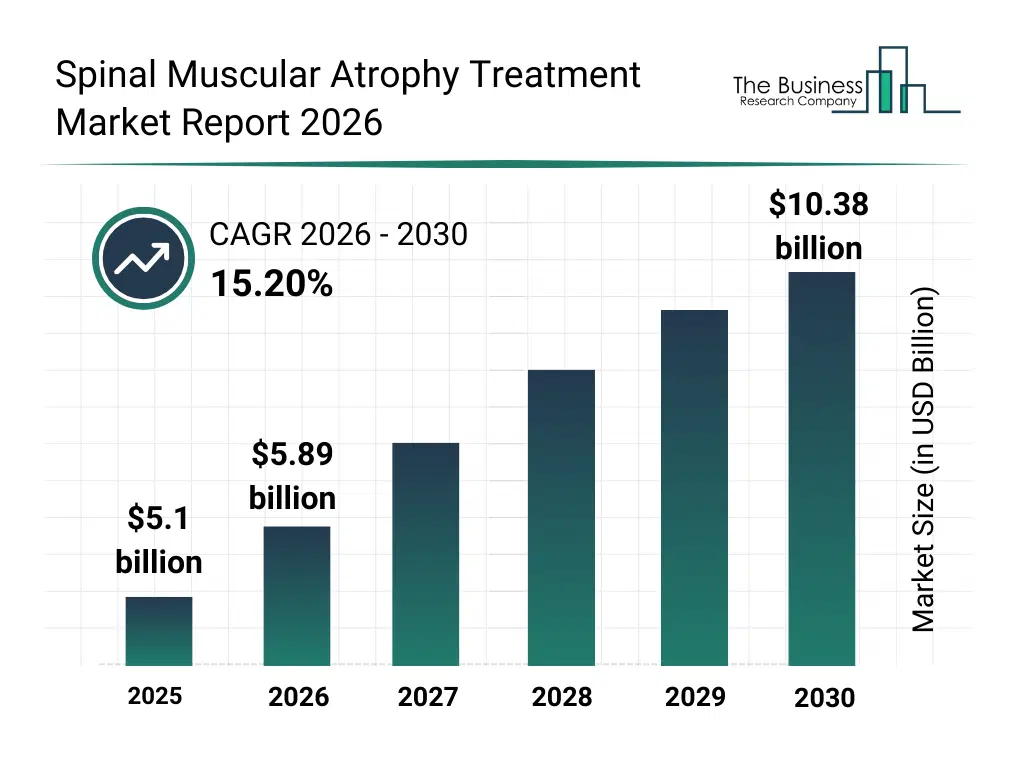
What Is The Spinal Muscular Atrophy Treatment Market Size and Share 2026?
The spinal muscular atrophy treatment market size has grown rapidly in recent years. It will grow from $5.1 billion in 2025 to $5.89 billion in 2026 at a compound annual growth rate (CAGR) of 15.5%. The growth in the historic period can be attributed to limited therapeutic options for sma, low awareness of genetic disorders, reliance on hospital-based treatment, slow adoption of innovative therapies, lack of early diagnosis programs.What Is The Spinal Muscular Atrophy Treatment Market Growth Forecast?
The spinal muscular atrophy treatment market size is expected to see rapid growth in the next few years. It will grow to $10.38 billion in 2030 at a compound annual growth rate (CAGR) of 15.2%. The growth in the forecast period can be attributed to development of advanced gene therapies, rising investment in sma research, increasing newborn screening initiatives, growth of specialty clinics and homecare services, integration of digital health and telemedicine solutions. Major trends in the forecast period include rising adoption of gene therapy and smn2 modifying treatments, increased focus on early diagnosis and newborn screening programs, growth in homecare and specialty clinic-based sma management, expansion of multidisciplinary care teams for sma patients, integration of telehealth and remote patient monitoring.Global Spinal Muscular Atrophy Treatment Market Segmentation
1) By Treatment: Medication, Physical Therapy, Surgery, Other Treatments 2) By Type: Werdnig-Hoffmann Disease, Infant Spinal Muscular Atrophy, Kugelberg-Welander Disease, Adult Spinal Muscular Atrophy 3) By Route Of Administration: Oral, Parenteral 4) By Distribution Channel: Hospital Pharmacies, Retail Pharmacies, Other Distribution Channels 5) By End User: Hospitals, Homecare, Specialty Clinics, Other End Users Subsegments: 1) By Medication: Nusinersen (Spinraza), Onasemnogene Abeparvovec-Xioi (Zolgensma), Risdiplam (Evrysdi), Antisense Oligonucleotides, Spinal Muscular Atrophy (SMN2) Splicing Modifiers, Corticosteroids, Supportive Medications 2) By Physical Therapy: Occupational Therapy, Respiratory Therapy, Aquatic Therapy, Orthotic Support, Assistive Mobility Devices, Range-Of-Motion Exercises 3) By Surgery: Spinal Fusion Surgery, Scoliosis Correction Surgery, Orthopedic Surgery, Gastrostomy Tube Placement, Tracheostomy 4) By Other Treatments: Nutritional Support, Respiratory Support, Psychosocial Support And Counseling, Genetic Counseling, Stem Cell TherapyWhat Is The Driver Of The Spinal Muscular Atrophy Treatment Market?
The growing focus on gene therapy is expected to propel the growth of the spinal muscular atrophy treatment market going forward. Gene therapy is a medical technique that involves modifying a person's genetic material, either DNA or RNA, to treat or prevent diseases. The growing focus on gene therapy is primarily due to its potential to treat or cure the root cause of genetic disorders by directly modifying or replacing faulty genes, offering long-term or permanent therapeutic effects compared to traditional treatments. Spinal muscular atrophy treatments are useful for gene therapy, such as onasemnogene abeparvovec (Zolgensma), providing a one-time, targeted approach that delivers a functional copy of the SMN1 gene, significantly improving motor function and quality of life for patients with SMA. For instance, in January 2024, according to the American Society of Gene and Cell Therapy, a US-based organization for gene and cell therapy, the number of gene therapies in Phase III trials increased by 10% in the fourth quarter of 2023 compared to the previous quarter. This marked the first such increase since the third quarter of 2022. Therefore, the growing focus on gene therapy is driving the growth of the spinal muscular atrophy treatment industry.Key Players In The Global Spinal Muscular Atrophy Treatment Market
Major companies operating in the spinal muscular atrophy treatment market are F. Hoffmann-La Roche Ltd, Novartis AG, Biogen Inc., Ionis Pharmaceuticals Inc., Chugai Pharmaceutical Co. Ltd, PTC Therapeutics Inc., Voyager Therapeutics Inc., Scholar Rock Holding Corporation, Genethon, Exegenesis Bio Inc., Alcyone Therapeutics Inc., Biophytis SA, Ractigen Therapeutics, Sarepta Therapeutics Inc., Astellas Pharma Inc., Pfizer Inc., AstraZeneca PLC, Roche Molecular Systems Inc., Avexis Inc., Cytokinetics Incorporated, Repligen Corporation, Neurogene Inc., Solid Biosciences Inc., Entrada Therapeutics Inc., Regenxbio Inc., Rocket Pharmaceuticals Inc., Asklepios BioPharmaceutical (AskBio) Inc., Ultragenyx Pharmaceutical Inc., Catalyst Pharmaceuticals Inc., NMD Pharma A/S, Biohaven Pharmaceutical Holding Company Ltd, Vybion Inc., Apteeus SAS, BIOCAD INDIA PRIVATE LIMITEDGlobal Spinal Muscular Atrophy Treatment Market Trends and Insights
Major companies operating in the spinal muscular atrophy treatment market are focusing on developing innovative formulations such as survival motor neuron 2 (SMN2) splicing modifier tablets to enhance motor function and slow disease progression. Survival motor neuron 2 (SMN2) splicing modifier tablets are oral medications designed to alter the splicing of the SMN2 gene, enabling the production of functional SMN protein that is critical for motor neuron survival. These tablets help treat spinal muscular atrophy (SMA) by increasing the levels of full-length SMN protein, thereby improving muscle function and slowing disease progression. For instance, in February 2025, Genentech, Inc., a US-based biotechnology company, received U.S. Food and Drug Administration approval for its Evrysdi (risdiplam) tablet, the first and only non-invasive SMN2 splicing modifier tablet for the treatment of SMA. The Evrysdi tablet offers comparable efficacy and safety to the existing oral solution, with added benefits such as room-temperature storage and simplified administration for patients aged 2 years and older who weigh more than 44 lbs (20 kgs), providing greater convenience and flexibility in disease management.What Are Latest Mergers And Acquisitions In The Spinal Muscular Atrophy Treatment Market?
In January 2024, Voyager Therapeutics Inc., a US-based biotechnology company, partnered with Novartis to advance gene therapies for neurological diseases. With this partnership, Voyager Therapeutics and Novartis aim to collaborate on discovering and developing novel gene therapies for Huntington’s disease and spinal muscular atrophy by combining Voyager’s TRACER capsid technology with Novartis’s expertise in gene therapy development and commercialization. Novartis, a Switzerland-based pharmaceutical company that specializes in providing drugs for spinal muscular atrophy (SMA).Regional Insights
North America was the largest region in the spinal muscular atrophy treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Spinal Muscular Atrophy Treatment Market?
The spinal muscular atrophy treatment market consists of revenues earned by entities by providing services such as gene delivery services, diagnostic services, and supportive care services. The market value includes the value of related goods sold by the service provider or included within the service offering. The spinal muscular atrophy treatment market also includes sales of spinraza, zolgensma, evrysdi, and supportive care products. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Spinal Muscular Atrophy Treatment Market Report 2026?
The spinal muscular atrophy treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the spinal muscular atrophy treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Spinal Muscular Atrophy Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.89 billion |
| Revenue Forecast In 2035 | $10.38 billion |
| Growth Rate | CAGR of 15.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment, Type, Route Of Administration, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Novartis AG, Biogen Inc., Ionis Pharmaceuticals Inc., Chugai Pharmaceutical Co. Ltd, PTC Therapeutics Inc., Voyager Therapeutics Inc., Scholar Rock Holding Corporation, Genethon, Exegenesis Bio Inc., Alcyone Therapeutics Inc., Biophytis SA, Ractigen Therapeutics, Sarepta Therapeutics Inc., Astellas Pharma Inc., Pfizer Inc., AstraZeneca PLC, Roche Molecular Systems Inc., Avexis Inc., Cytokinetics Incorporated, Repligen Corporation, Neurogene Inc., Solid Biosciences Inc., Entrada Therapeutics Inc., Regenxbio Inc., Rocket Pharmaceuticals Inc., Asklepios BioPharmaceutical (AskBio) Inc., Ultragenyx Pharmaceutical Inc., Catalyst Pharmaceuticals Inc., NMD Pharma A/S, Biohaven Pharmaceutical Holding Company Ltd, Vybion Inc., Apteeus SAS, BIOCAD INDIA PRIVATE LIMITED |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
