
Temporary Mechanical Circulatory Support Device Market Report 2026
Global Outlook – By Type (Left Ventricular Support Devices, Right Support Devices, Biventricular Support Devices), By Technology (Pneumatic Devices, Mechanical Pumps, Biomimetic Devices, Hybrid Systems, Novel Wearable Devices), By Application (Cardiogenic Shock, Bridge To Transplantation, Bridge To Recovery, High-Risk Cardiac Procedures, Other Applications), By End User (Hospitals, Cardiac Surgery Centers, Outpatient Surgical Facilities, Research And Academic Institutions, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Temporary Mechanical Circulatory Support Device Market Overview
• Temporary Mechanical Circulatory Support Device market size has reached to $2.66 billion in 2025 • Expected to grow to $4.1 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Increasing Incidence Of Cardiovascular Diseases Is Fueling Market Growth Due To Aging Populations And Rising Patient Numbers • Market Trend: Strategic Partnership Formed To Advance Next-Generation Percutaneous Ventricular Assist Device • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Temporary Mechanical Circulatory Support Device Market?
A temporary mechanical circulatory support (MCS) device is a specialized medical device designed to provide short-term assistance to the heart’s pumping function in patients experiencing acute cardiac dysfunction or failure by mechanically augmenting blood flow and stabilizing hemodynamics until recovery, transplantation, or more permanent intervention can be pursued. These devices are used in critical care settings to support patients during high-risk cardiac procedures, manage cardiogenic shock, or bridge them to heart transplantation or durable ventricular assist device implantation. The main types of temporary mechanical circulatory support devices are left ventricular support devices, right support devices, and biventricular support devices. Left ventricular support devices are mechanical machines that assist or take over the pumping function of the left ventricle to help maintain adequate blood flow in patients with heart failure. The multiple technologies are pneumatic devices, mechanical pumps, biomimetic devices, hybrid systems, and novel wearable devices. The various applications involved are cardiogenic shock, bridge to transplantation, bridge to recovery, high-risk cardiac procedures, and others, and they are used by several end-users such as hospitals, cardiac surgery centers, outpatient surgical facilities, research and academic institutions, and others.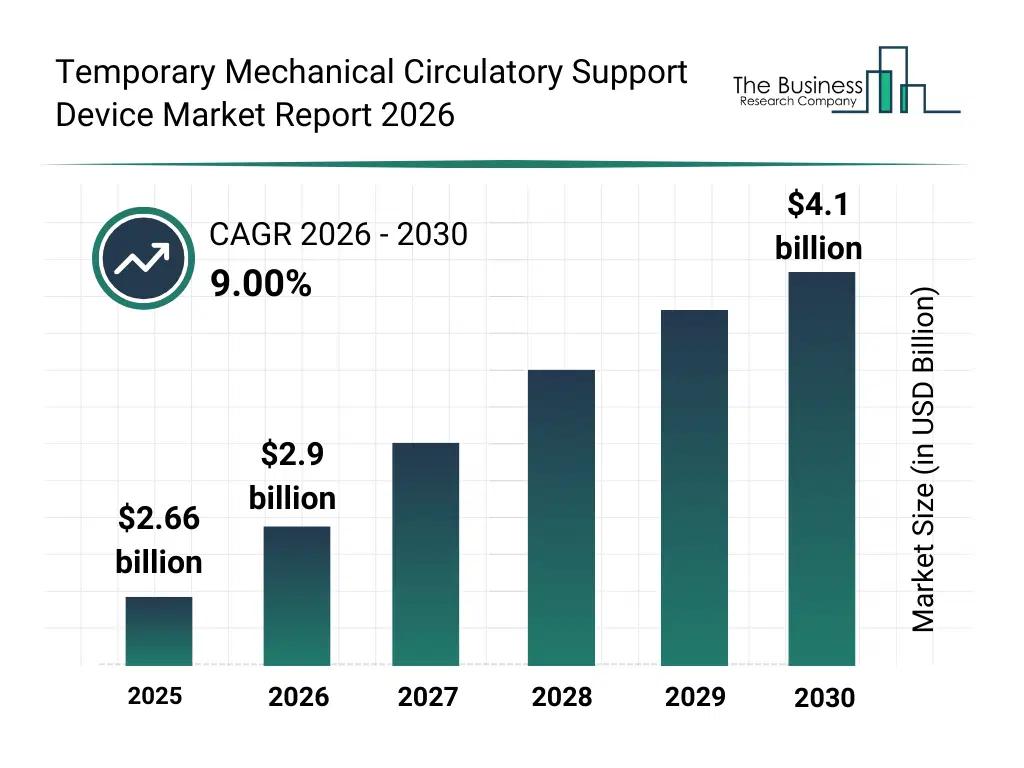
What Is The Temporary Mechanical Circulatory Support Device Market Size and Share 2026?
The temporary mechanical circulatory support device market size has grown strongly in recent years. It will grow from $2.66 billion in 2025 to $2.9 billion in 2026 at a compound annual growth rate (CAGR) of 9.0%. The growth in the historic period can be attributed to limited availability of temporary mcs devices, reliance on invasive open-heart procedures, high mortality rates from cardiogenic shock, growing demand for acute cardiac care, lack of portable and wearable solutions.What Is The Temporary Mechanical Circulatory Support Device Market Growth Forecast?
The temporary mechanical circulatory support device market size is expected to see strong growth in the next few years. It will grow to $4.1 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to technological advancements in biomimetic and hybrid devices, increasing adoption of minimally invasive mcs, rising cardiac surgery volumes, expansion of outpatient cardiac centers, integration of digital monitoring and smart hemodynamic management. Major trends in the forecast period include rising adoption of percutaneous ventricular assist devices, increased use of extracorporeal membrane oxygenation (ecmo), growth in high-risk cardiac procedures, expansion of bridge-to-transplant and bridge-to-recovery applications, focus on minimally invasive and portable mcs devices.Global Temporary Mechanical Circulatory Support Device Market Segmentation
1) By Type: Left Ventricular Support Devices, Right Support Devices, Biventricular Support Devices 2) By Technology: Pneumatic Devices, Mechanical Pumps, Biomimetic Devices, Hybrid Systems, Novel Wearable Devices 3) By Application: Cardiogenic Shock, Bridge To Transplantation, Bridge To Recovery, High-Risk Cardiac Procedures, Other Applications 4) By End User: Hospitals, Cardiac Surgery Centers, Outpatient Surgical Facilities, Research And Academic Institutions, Other End-Users Subsegments: 1) By Left Ventricular Support Devices: Intra-Aortic Balloon Pump, Percutaneous Ventricular Assist Device, Extracorporeal Membrane Oxygenation 2) By Right Support Devices: Right Ventricular Assist Device, Extracorporeal Membrane Oxygenation, Percutaneous Right Ventricular Assist Device 3) By Biventricular Support Devices: Biventricular Assist Device, Temporary Total Artificial Heart, Extracorporeal Membrane OxygenationWhat Is The Driver Of The Temporary Mechanical Circulatory Support Device Market?
The increasing incidence of cardiovascular diseases is expected to propel the growth of the temporary mechanical circulatory support device market going forward. Cardiovascular diseases (CVD) are a group of disorders that affect the heart and blood vessels, leading to conditions such as heart attacks, strokes, and hypertension. The rise in cardiovascular diseases is due to aging populations, as older age increases the risk of heart and blood vessel conditions. Temporary mechanical circulatory support devices help patients with cardiovascular diseases by temporarily supporting heart function and improving blood flow during critical conditions such as heart failure or cardiogenic shock.For instance, in December 2024, GOV.UK, a UK-based government agency, reported that in March 2024, nearly 2.24 million adults in England—about 4.4% of those aged 18 and over—had a GP-recorded diagnosis of CKD. This reflects an increase from 2.09 million cases the previous year. Therefore, the increasing incidence of cardiovascular diseases is driving growth in the temporary mechanical circulatory support device industry.Key Players In The Global Temporary Mechanical Circulatory Support Device Market
Major companies operating in the temporary mechanical circulatory support device market are Abbott Laboratories, Medtronic Plc, Boston Scientific Corporation, Terumo Corporation, Getinge AB, LivaNova PLC, Magenta Medical Ltd., CorWave SA, Procyrion Inc., CardioBridge GmbH, Abiomed Inc., Berlin Heart GmbH, Thoratec Corporation, PulseCath BV, TandemLife Inc., SynCardia Systems LLC, CardiacAssist Inc., Xenios AG, Nihon Kohden Corporation, Eurosets Srl, Senko Medical Instrument Manufacturing Co Ltd, Nipro Medical Corporation, Calon Cardio‑Technology Ltd., Windmill Cardiovascular Systems Inc.Global Temporary Mechanical Circulatory Support Device Market Trends and Insights
Major companies in the temporary mechanical circulatory support (tMCS) device market are forming strategic partnerships to develop advanced technologies. For instance, in November 2025, VenstraMedical, an Australia-based cardiovascular medical device company, partnered with Highcroft Capital, a US-based venture capital firm, securing strategic investment to accelerate the development of its next-generation percutaneous ventricular assist device (pVAD). This low-profile, catheter-based pump is designed to provide full ventricular unloading and hemodynamic support for patients in cardiogenic shock or undergoing high-risk coronary procedures. Protected by multiple patents, the device enables minimally invasive insertion and can deliver up to 7 liters per minute of cardiac output. The investment highlights VenstraMedical’s aim to transform temporary mechanical circulatory support in a market valued over $2 billion, with plans to move toward clinical trials and commercialization.What Are Latest Mergers And Acquisitions In The Temporary Mechanical Circulatory Support Device Market?
In February 2023, Abbott Laboratories, a US-based pharmaceutical company, acquired Cardiovascular Systems, Inc. (CSI) for an undisclosed amount. With this acquisition, Abbott’s aim is to strengthen and diversify its vascular-device portfolio by integrating CSI’s specialized atherectomy technology, thereby enhancing its treatment offerings for complex peripheral and coronary artery disease. CSI is a US-based medical device company that provides temporary mechanical circulatory support devices.Regional Outlook
North America was the largest region in the temporary mechanical circulatory support device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Temporary Mechanical Circulatory Support Device Market?
The temporary mechanical circulatory support device market consists of sales of intra-aortic balloon pump, extracorporeal membrane oxygenation device, percutaneous ventricular assist device, and centrifugal pumps. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Temporary Mechanical Circulatory Support Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.9 billion |
| Revenue Forecast In 2035 | $4.1 billion |
| Growth Rate | CAGR of 9.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic Plc, Boston Scientific Corporation, Terumo Corporation, Getinge AB, LivaNova PLC, Magenta Medical Ltd., CorWave SA, Procyrion Inc., CardioBridge GmbH, Abiomed Inc., Berlin Heart GmbH, Thoratec Corporation, PulseCath BV, TandemLife Inc., SynCardia Systems LLC, CardiacAssist Inc., Xenios AG, Nihon Kohden Corporation, Eurosets Srl, Senko Medical Instrument Manufacturing Co Ltd, Nipro Medical Corporation, Calon Cardio‑Technology Ltd., Windmill Cardiovascular Systems Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
