
Viral And Non-Viral Vector Manufacturing Market Report 2026
Global Outlook – By Vector Type (Viral Vector, Non-Viral Vector), By Disease (Cancer, Genetic Disease, Infectious Disease, Cardiovascular Disease, Other Diseases), By Application (Gene Therapy, Vaccinology, Cell Therapy, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Viral And Non-Viral Vector Manufacturing Market Overview
• Viral And Non-Viral Vector Manufacturing market size has reached to $10.28 billion in 2025 • Expected to grow to $26.83 billion in 2030 at a compound annual growth rate (CAGR) of 21.3% • Growth Driver: Clinical Trial Surges Fuel Viral And Non-Viral Vector Manufacturing Growth • Market Trend: Advancing Vector Technology For Enhanced Gene And Cell Therapies • North America was the largest region in 2025.What Is Covered Under Viral And Non-Viral Vector Manufacturing Market?
Viral and non-viral vector manufacturing refers to producing viral and non-viral vectors, tools that deliver genetic material into cells. Viral vectors treat human diseases and are used in anticancer and gene therapies. Non-viral vectors are utilized for a variety of gene therapy applications, particularly for the creation of vaccines and the treatment of cancer. The main types of viral and non-viral vector manufacturing vectors are viral vectors and non-viral vectors. A viral vector refers to a method commonly employed in gene therapy and gene editing to add therapeutic genes or change how existing genes are expressed in living things. The various diseases are cancer, genetic disease, infectious disease, cardiovascular disease, and others applied in gene therapy, vaccinology, cell therapy, and others.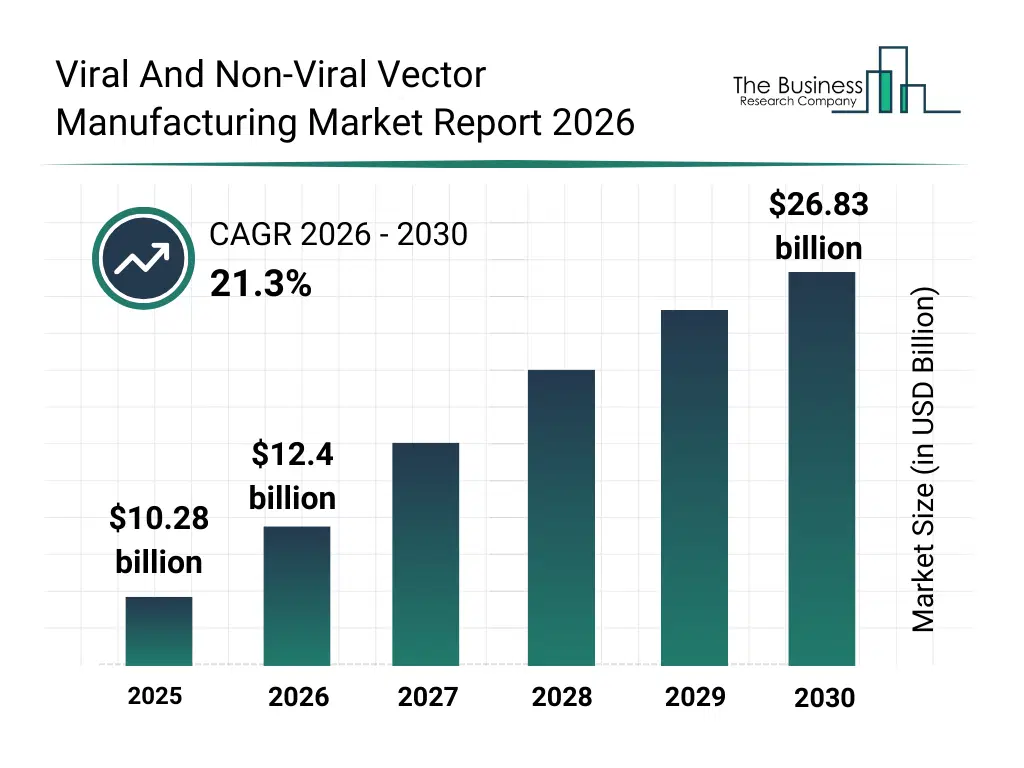
What Is The Viral And Non-Viral Vector Manufacturing Market Size and Share 2026?
The viral and non-viral vector manufacturing market size has grown exponentially in recent years. It will grow from $10.28 billion in 2025 to $12.4 billion in 2026 at a compound annual growth rate (CAGR) of 20.7%. The growth in the historic period can be attributed to advancements in gene therapy research, increasing clinical trial activity, expansion of biopharmaceutical manufacturing facilities, rising demand for viral vectors in oncology, availability of specialized manufacturing expertise.What Is The Viral And Non-Viral Vector Manufacturing Market Growth Forecast?
The viral and non-viral vector manufacturing market size is expected to see exponential growth in the next few years. It will grow to $26.83 billion in 2030 at a compound annual growth rate (CAGR) of 21.3%. The growth in the forecast period can be attributed to increasing commercialization of gene and cell therapies, rising investments in vaccine development, expansion of CDMO partnerships, growing focus on scalable and flexible manufacturing, increasing regulatory approvals for advanced therapies. Major trends in the forecast period include increasing scale-up of viral vector production capacity, rising demand for non-viral delivery systems, growing adoption of modular manufacturing platforms, expansion of contract vector manufacturing services, enhanced focus on process standardization and quality control.Global Viral And Non-Viral Vector Manufacturing Market Segmentation
1) By Vector Type: Viral Vector, Non-Viral Vector 2) By Disease: Cancer, Genetic Disease, Infectious Disease, Cardiovascular Disease, Other Diseases 3) By Application: Gene Therapy, Vaccinology, Cell Therapy, Other Applications Subsegments: 1) By Viral Vector: Adenoviral Vectors, Lentiviral Vectors, AAV (Adeno-Associated Virus) Vectors, Retroviral Vectors, Other Viral Vectors 2) By Non-Viral Vector: Plasmid DNA, Lipid-Based Vectors, Nanoparticles, Electroporation-Based Vectors, Other Non-Viral VectorsWhat Is The Driver Of The Viral And Non-Viral Vector Manufacturing Market?
The rising number of clinical trials is expected to propel the growth of the viral and non-viral vector manufacturing markets going forward. Clinical trials are research studies conducted with human participants to evaluate the safety, efficacy, and potential side effects of a medical intervention, treatment, or drug. Viral and non-viral vector manufacturing is indispensable in supporting gene therapy clinical trials by providing the necessary tools for delivering therapeutic genes to patients and ensuring the safety, quality, and scalability of these vectors throughout the trial phases. For instance, in December 2023, according to ClinicalTrials.gov, a US-based web-based resource maintained by the US National Library of Medicine, providing information on clinical trials in the year 2022, a total of 437,515 clinical trials were registered, and among them, 56,561 were completed. Moving into 2023, as of December 20, there has been an increase in both registration and completion figures. Specifically, 476,626 clinical trials have been registered, with 61,504 trials already completed. Therefore, the rising number of clinical trials is driving the growth of the viral and non-viral vector manufacturing markets.Key Players In The Global Viral And Non-Viral Vector Manufacturing Market
Major companies operating in the viral and non-viral vector manufacturing market are Novartis AG, Thermo Fisher Scientific Inc., Merck KGaA, FUJIFILM Holdings Corporation, Regeneron Pharmaceuticals Inc., Astellas Pharma Inc., Lonza Group AG, WuXi AppTec Co Ltd., Catalent Inc., Sartorius AG, Charles River Laboratories Inc., Sarepta Therapeutics Inc., GenScript ProBio Co. Ltd., TAKARA BIO Inc., Oxford BioMedica plc, Beam Therapeutics Inc., Intellia Therapeutics Inc., MaxCyte Inc., Voyager Therapeutics Inc., Genethon, MeiraGTx Holdings plc, Evox Therapeutics Ltd., Entos Pharmaceuticals Inc., LogicBio Therapeutics Inc., bluebird bio Inc., 4D Molecular Therapeutics Inc., Precision NanoSystems Inc., GenSight Biologics S.A., Abeona Therapeutics Inc., Generation Bio Co.Global Viral And Non-Viral Vector Manufacturing Market Trends and Insights
Major companies operating in the viral and non-viral vector manufacturing market are focused on partnerships to increase their profitability in the market. Collaborative partnerships in viral and non-viral vector manufacturing involve biopharmaceutical companies, CDMOs, research institutions, and technology providers working together to enhance the efficiency, safety, and scalability of non-viral vector development and production. For instance, in September 2023, NecstGen, a Netherland-based CDMO providing comprehensive support for all developers in the field of Cell and Gene Therapy collaborated with ProteoNic BioSciences, a Netherland-based technology provider for the production of complex therapeutic proteins and launched ProteoNic's 2G UNic premium vector technology. The successful application of this technology has enhanced the development of lentiviral (LV) vectors, boosting functional viral particle titers and directly improving process efficiency, thereby increasing the cost-effectiveness and accessibility of therapies. This versatile technology extends to various applications, including gene therapy viral vector production, mRNA therapeutics, and transient protein production. By advancing AAV and LV viral vector manufacturing technology for both transient systems and stable producer cell lines, this development has the potential to benefit viral vector manufacturing for gene therapies and non-vector processes, offering potential benefits across diverse applications.What Are Latest Mergers And Acquisitions In The Viral And Non-Viral Vector Manufacturing Market?
In March 2023, Sartorius, a Germany based pharmaceutical and laboratory equipment supplier, acquired Polyplus for approximately $2.59 billion. Through this acquisition Sartorius aims to strengthen its position in the gene therapy and viral vector production markets by expanding its cell and gene therapy capabilities. Polyplus is a France based biotechnology company that specializes in the manufacturing of both viral and non-viral vectors for gene therapy applications.Regional Outlook
North America was the largest region in the viral and non-viral vector manufacturing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral And Non-Viral Vector Manufacturing Market?
The viral and non-viral vector manufacturing market includes revenues earned by entities by providing adeno-associated viral vectors, lentiviral vectors, retroviral vectors, adenoviral vectors, particle-based vectors, and chemical-based vectors. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral And Non-Viral Vector Manufacturing Market Report 2026?
The viral and non-viral vector manufacturing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral and non-viral vector manufacturing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral And Non-Viral Vector Manufacturing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $12.4 billion |
| Revenue Forecast In 2035 | $26.83 billion |
| Growth Rate | CAGR of 20.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Vector Type, Disease, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Novartis AG, Thermo Fisher Scientific Inc., Merck KGaA, FUJIFILM Holdings Corporation, Regeneron Pharmaceuticals Inc., Astellas Pharma Inc., Lonza Group AG, WuXi AppTec Co Ltd., Catalent Inc., Sartorius AG, Charles River Laboratories Inc., Sarepta Therapeutics Inc., GenScript ProBio Co. Ltd., TAKARA BIO Inc., Oxford BioMedica plc, Beam Therapeutics Inc., Intellia Therapeutics Inc., MaxCyte Inc., Voyager Therapeutics Inc., Genethon, MeiraGTx Holdings plc, Evox Therapeutics Ltd., Entos Pharmaceuticals Inc., LogicBio Therapeutics Inc., bluebird bio Inc., 4D Molecular Therapeutics Inc., Precision NanoSystems Inc., GenSight Biologics S.A., Abeona Therapeutics Inc., Generation Bio Co. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
