
Biosimilar Therapeutic Peptides Market Report 2026
Global Outlook – By Type (Innovative, Generic), By Route Of Administration (Parenteral Route, Transdermal Route, Other Route Of Administrations), By Application (Cancer, Cardiovascular, Central Nervous Systems, Metabolic Disorders, Infection, Hematological Disorders, Gastrointestinal Disorders, Dermatology, Respiratory Disorders) – Market Size, Trends, Strategies, and Forecast to 2035
Biosimilar Therapeutic Peptides Market Overview
• Biosimilar Therapeutic Peptides market size has reached to $3.5 billion in 2025 • Expected to grow to $6.11 billion in 2030 at a compound annual growth rate (CAGR) of 11.9% • Growth Driver: Rising Prevalence Of Chronic Diseases Fuels Growth In The Biosimilar Therapeutic Peptide Market • Market Trend: Fujitsu Launches Biodrug Design Accelerator Platform To Revolutionize Peptide Drug Discovery • North America was the largest region in 2025 and Middle East is the fastest growing region.What Is Covered Under Biosimilar Therapeutic Peptides Market?
Biosimilar therapeutic peptides refer to a peptide drug that is used in place of or in addition to peptide hormones when endogenous levels tend to decline. Therapeutic peptides that are biosimilar have applications in the treatment of cancer, metabolic, pulmonary, cardiovascular, and other illnesses. The main types of biosimilar therapeutic peptides are innovative and generic. Biosimilar medications are frequently mistaken for generic drugs. Both are marketed as low-cost knockoffs of high-priced name-brand medications. The main distinction is that generics are duplicates of synthetic pharmaceuticals, whereas biosimilars are based on drugs that contain biological organisms as active components. The different routes of administration include the parenteral route, the transdermal route, and others and are used in cancer, cardiovascular disorders, central nervous system, metabolic disorders, infections, hematological disorders, gastrointestinal disorders, dermatology, and respiratory disorders.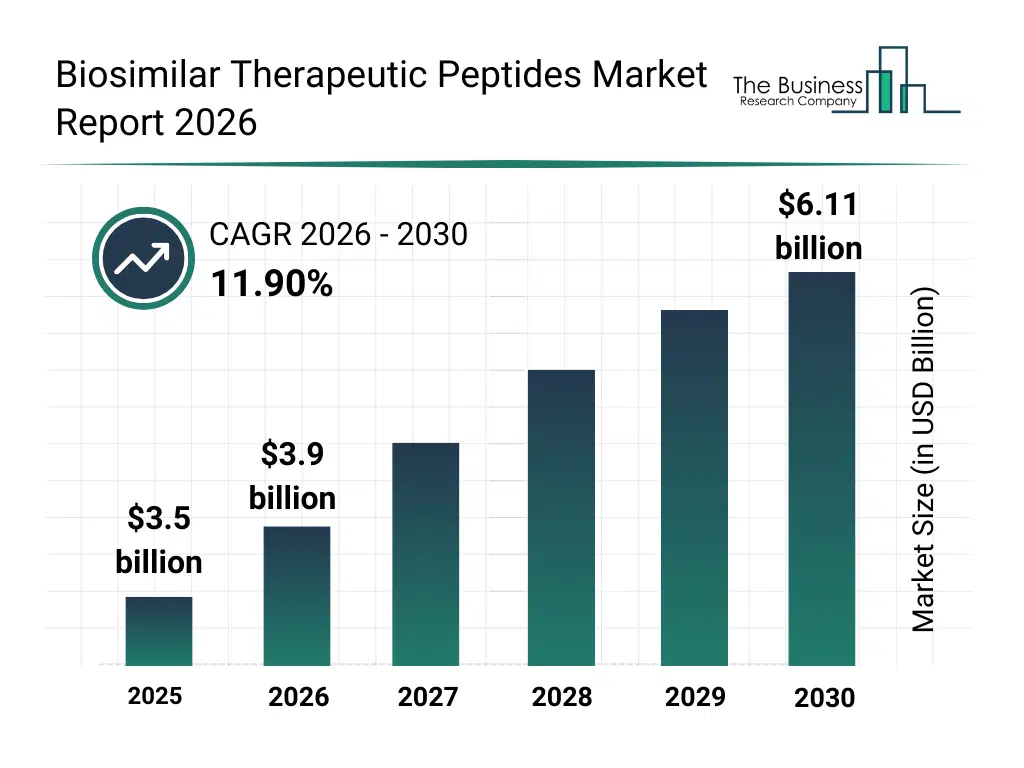
What Is The Biosimilar Therapeutic Peptides Market Size and Share 2026?
The biosimilar therapeutic peptides market size has grown rapidly in recent years. It will grow from $3.5 billion in 2025 to $3.9 billion in 2026 at a compound annual growth rate (CAGR) of 11.5%. The growth in the historic period can be attributed to limited availability of biosimilar peptides, reliance on innovative peptide therapies, growing prevalence of cancer and metabolic disorders, increasing awareness of peptide-based treatments, development of parenteral administration methods.What Is The Biosimilar Therapeutic Peptides Market Growth Forecast?
The biosimilar therapeutic peptides market size is expected to see rapid growth in the next few years. It will grow to $6.11 billion in 2030 at a compound annual growth rate (CAGR) of 11.9%. The growth in the forecast period can be attributed to launch of combination and long-acting peptides, expansion of generic peptide production, increasing adoption of transdermal delivery systems, rising investment in peptide research and development, growing healthcare expenditure on peptide therapeutics. Major trends in the forecast period include rising demand for biosimilar therapeutic peptides in cancer treatment, growth in peptide-based therapies for metabolic and cardiovascular disorders, increasing adoption of long-acting and combination peptides, expansion of parenteral and transdermal administration routes, focus on cost-effective generic peptides and accessibility in emerging markets.Global Biosimilar Therapeutic Peptides Market Segmentation
1) By Type: Innovative, Generic 2) By Route Of Administration: Parenteral Route, Transdermal Route, Other Route Of Administrations 3) By Application: Cancer, Cardiovascular, Central Nervous Systems, Metabolic Disorders, Infection, Hematological Disorders, Gastrointestinal Disorders, Dermatology, Respiratory Disorders Subsegments: 1) By Innovative: Long-Acting Peptides, Combination Peptides 2) By Generic: Monoclonal Antibodies, Hormones, EnzymesWhat Is The Driver Of The Biosimilar Therapeutic Peptides Market?
The prevalence of chronic diseases is expected to drive the biosimilar therapeutic peptide market. Chronic diseases are long-lasting medical conditions that require ongoing management and treatment, often lasting for years or a lifetime. Increasing chronic diseases drive greater demand for cost-effective treatments, making biosimilar therapeutic peptides an attractive option due to their affordability and potential to improve patient access and healthcare system sustainability. Regulatory support and clinical evidence further fuel market growth. For instance, in April 2025, according to the Centers for Disease Control and Prevention (CDC), a US-based federal agency. in 2023, approximately 194 million U.S. adults, or 76.4%, reported having at least one chronic condition. The prevalence was 59.5% among young adults, 78.4% among midlife adults, and 93.0% among older adults. Therefore, the prevalence of chronic diseases is expected to drive the growth of the biosimilar therapeutic peptides industry.Key Players In The Global Biosimilar Therapeutic Peptides Market
Major companies operating in the biosimilar therapeutic peptides market are Pfizer Inc., Merck & Co. Inc., Novartis AG, Sanofi SA, Takeda Pharmaceuticals Co. Ltd., Eli Lilly and Company, Novo Nordisk A/S, Apotex Inc., Teva Pharmaceuticals Industries Ltd., Sandoz International GmbH, Lonza Inc., Stada Arzneimittel AG, Ipsen S.A, Biocon Biopharmaceuticals Pvt. Ltd., Dr. Reddy's Laboratories Ltd., Alvogen Pharma US Inc., Corden Pharma GmbH, Bachem Holding AG, PolyPeptide Laboratories Ltd.Global Biosimilar Therapeutic Peptides Market Trends and Insights
Major companies in the biosimilar therapeutic peptide market are developing a research platform for optimizing peptide synthesis, enhancing production efficiency, and ensuring consistent quality, from discovery to commercialization. Biodrug Design Accelerator, a platform to manage research processes for peptide drug discovery, which many regard as a promising frontier in the field of new drug development. For instance, in May 2023, Fujitsu Limited, a US-based toy manufacturing and entertainment company, launched the Biodrug Design Accelerator platform to accelerate peptide drug discovery research. Biodrug Design Accelerator will support peptide drug discovery scientists in pharmaceutical companies to accelerate and improve the efficiency of peptide drug development, specifically through the cycles design, make, test, analyze (DMTA), ultimately enabling accelerated and informed design of peptide drug candidates as well as efficient communication among scientists regarding synthesis and testing.What Are Latest Mergers And Acquisitions In The Biosimilar Therapeutic Peptides Market?
In September 2024, Biocon Limited, an India-based biopharmaceutical company, formed a partnership with Tabuk Pharmaceuticals to commercialize its Glucagon-like peptide-1 (GLP-1) products in the Middle East region. The collaboration aims to expand the availability and adoption of Biocon’s GLP-1 therapies, enhancing treatment options for patients with metabolic disorders while driving growth and innovation in the Middle Eastern diabetes care market. Tabuk Pharmaceuticals is a Saudi Arabia-based pharmaceutical company focused on the distribution and commercialization of innovative healthcare products.Regional Insights
North America was the largest region in the biosimilar therapeutics peptides market in 2025. Middle East is expected to be the fastest growing region in the biosimilar therapeutic peptides market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biosimilar Therapeutic Peptides Market?
The biosimilar therapeutic peptides market consists of sales of pegfilgrastim, bevacizumab, and adalimumab. Values in this market are """"factory gate values,"""" that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biosimilar Therapeutic Peptides Market Report 2026?
The biosimilar therapeutic peptides market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biosimilar therapeutic peptides industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biosimilar Therapeutic Peptides Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.9 billion |
| Revenue Forecast In 2035 | $6.11 billion |
| Growth Rate | CAGR of 11.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Route Of Administration, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Merck & Co. Inc., Novartis AG, Sanofi SA, Takeda Pharmaceuticals Co. Ltd., Eli Lilly and Company, Novo Nordisk A/S, Apotex Inc., Teva Pharmaceuticals Industries Ltd., Sandoz International GmbH, Lonza Inc., Stada Arzneimittel AG, Ipsen S.A, Biocon Biopharmaceuticals Pvt. Ltd., Dr. Reddy's Laboratories Ltd., Alvogen Pharma US Inc., Corden Pharma GmbH, Bachem Holding AG, PolyPeptide Laboratories Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
