
Cardiac Biomarkers Testing Market Report 2026
Global Outlook – By Biomarkers Type (Creatine Kinase (CK-MB), Troponins, Myoglobin, Natriuretic Peptides (BNP And NT-proBNP), Ischemia Modified Albumin (IMA), Other Biomarkers Types), By Application (Congestive Heart Failure, Acute Coronary Syndrome, Myocardial Infarction, Atherosclerosis, Other Applications), By End-User (Hospitals, Specialty Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Cardiac Biomarkers Testing Market Overview
• Cardiac Biomarkers Testing market size has reached to $14.95 billion in 2025 • Expected to grow to $25.92 billion in 2030 at a compound annual growth rate (CAGR) of 11.6% • Growth Driver: Rising Prevalence Of Cardiovascular Diseases Fuels Growth In The Cardiac Biomarkers Testing Market • Market Trend: Strategic Partnerships Enhance Early Detection And Management Of Heart Conditions • North America was the largest region in 2025.What Is Covered Under Cardiac Biomarkers Testing Market?
Cardiac biomarkers testing refers to the tests to evaluate the quantities of cardiac biomarkers in blood. It is used to diagnose, assess, and keep track of people who may have acute coronary syndrome (ACS). The main types of biomarkers in cardiac biomarkers testing are creatine kinase (CK-MB), troponins, myoglobin, natriuretic peptides (BNP and NT-proBNP), ischemia-modified albumin (IMA) and others. Creatine kinase (CK-MB) refers to a blood test that looks for a particular enzyme. The various applications involved are congestive heart failure, acute coronary syndrome, myocardial infarction, atherosclerosis and others, with hospitals and specialty clinic end-users.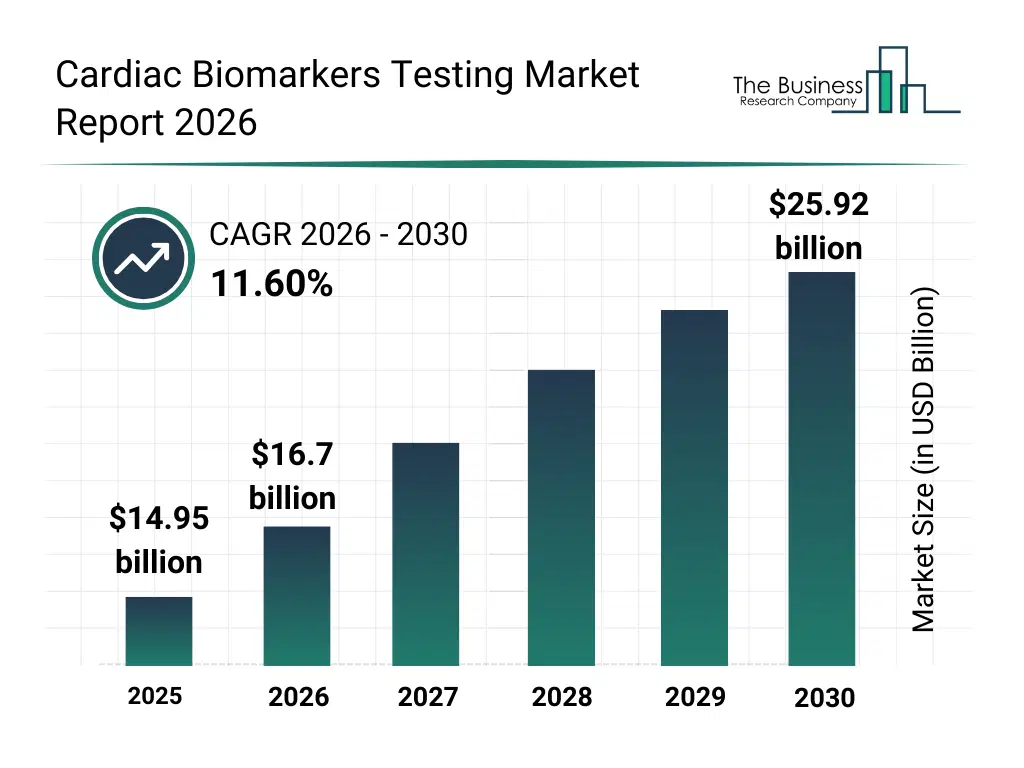
What Is The Cardiac Biomarkers Testing Market Size and Share 2026?
The cardiac biomarkers testing market size has grown rapidly in recent years. It will grow from $14.95 billion in 2025 to $16.7 billion in 2026 at a compound annual growth rate (CAGR) of 11.7%. The growth in the historic period can be attributed to rising incidence of cardiovascular diseases, increasing awareness about cardiac health, advancements in laboratory diagnostics, growing hospital infrastructure, increasing government healthcare initiatives.What Is The Cardiac Biomarkers Testing Market Growth Forecast?
The cardiac biomarkers testing market size is expected to see rapid growth in the next few years. It will grow to $25.92 billion in 2030 at a compound annual growth rate (CAGR) of 11.6%. The growth in the forecast period can be attributed to adoption of AI in diagnostics, expansion of home-based and remote monitoring, growth of precision medicine, development of multi-biomarker panels, rising demand for real-time cardiac monitoring. Major trends in the forecast period include point-of-care testing adoption, miniaturized & portable diagnostic devices, biomarker multiplexing technologies, personalized cardiac risk profiling, integration of wearables for cardiac monitoring.Global Cardiac Biomarkers Testing Market Segmentation
1) By Biomarkers Type: Creatine Kinase (CK-MB), Troponins, Myoglobin, Natriuretic Peptides (BNP And NT-proBNP), Ischemia Modified Albumin (IMA), Other Biomarkers Types 2) By Application: Congestive Heart Failure, Acute Coronary Syndrome, Myocardial Infarction, Atherosclerosis, Other Applications 3) By End-User: Hospitals, Specialty ClinicsWhat Is The Driver Of The Cardiac Biomarkers Testing Market?
The increasing prevalence of cardiovascular diseases is expected to boost the growth of the cardiac biomarkers testing market going forward. Cardiovascular disease refers to a type of condition that affects the heart or blood vessels. Cardiac biomarker tests assist in determining the levels of biomarkers to immediately determine the severity of a heart attack and its magnitude. Hence, an increase in the prevalence of cardiovascular diseases will lead to an increase in the demand for cardiac biomarker testing solutions. For instance, in January 2024, according to the American Heart Association, a US-based non-profit organization, the age-adjusted death rate from cardiovascular disease increased to 233.3 per 100,000 in 2024, up 4.0% from 224.4 per 100,000 in 2023. Therefore, an increase in the prevalence of cardiovascular diseases is driving the growth of the cardiac biomarkers testing industry.Key Players In The Global Cardiac Biomarkers Testing Market
Major companies operating in the cardiac biomarkers testing market are Abbott Laboratories, Bio-Rad Laboratories Inc., Danaher Corporation, F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Thermo Fisher Scientific Inc., Biomérieux SA, Becton Dickinson and Company, Randox Laboratories Ltd., Enzo Biochem Inc., Creative Diagnostics Inc., Merck Co. & KGaA, Beckman Coulter Inc., QIAGEN N.V., PerkinElmer Inc., Tosoh Corporation, Eurolyser Diagnostica GmbH, Cortez Diagnostics Inc., DIALAB GmbH, CardioGenics Holdings Inc., Lepu Medical Technology (Beijing) Co. Ltd., Biosynex SA, Charles River Laboratories, Quidel Corporation, Life Diagnostics, Trinity Biotech plc, Guangzhou Wondfo Biotech Co. Ltd., Nova Biomedical Corp., Ortho Clinical Diagnostics Inc., Sekisui Diagnostics Inc., Abcam plc, Bio-Techne CorporationGlobal Cardiac Biomarkers Testing Market Trends and Insights
Major companies operating in the cardiac biomarker testing market are adopting strategic partnerships to provide technology for detecting early warning signs of heart conditions and improving patient outcomes. Strategic partnerships in cardiac biomarker testing foster collaboration between companies, enabling the development of innovative diagnostics, expanding market reach, and accelerating the availability of advanced testing solutions for improved cardiovascular care. For instance, in September 2024, MediBuddy, an Indian-based healthcare technology company providing a comprehensive platform for telehealth services, health management, and insurance assistance, partnered with Philips, a Netherlands-based conglomerate corporation. With this partnership, they aim to enhance cardiac care in India by incorporating Philips' HeartPrint technology, enabling regular monitoring and risk assessment of heart health. This forward-thinking approach empowers users with advanced tools on the MediBuddy platform, fostering proactive management of heart health and better outcomes.What Are Latest Mergers And Acquisitions In The Cardiac Biomarkers Testing Market?
In July 2024, Roche Holding AG, a Switzerland-based pharmaceutical company, acquired LumiraDx’s Point of Care technology for an undisclosed amount. This acquisition aims to enhance Roche's diagnostic portfolio, particularly in the area of decentralized diagnostics. By integrating LumiraDx’s innovative point-of-care platform, Roche seeks to strengthen its ability to provide rapid, accurate testing at or near patient locations. LumiraDx Ltd. is a UK-based hospital and healthcare company that specializes in offering cardiac biomarker testing, including the NT-proBNP test.Regional Insights
North America was the largest region in the cardiac biomarkers testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cardiac Biomarkers Testing Market?
The cardiac biomarkers testing market includes revenues earned by entities by providing ischemia-modified albumin (IMA), peptides, and C-reactive protein (CRP) biomarkers testing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cardiac Biomarkers Testing Market Report 2026?
The cardiac biomarkers testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cardiac biomarkers testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cardiac Biomarkers Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $16.7 billion |
| Revenue Forecast In 2035 | $25.92 billion |
| Growth Rate | CAGR of 11.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Biomarkers Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Bio-Rad Laboratories Inc., Danaher Corporation, F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Thermo Fisher Scientific Inc., Biomérieux SA, Becton Dickinson and Company, Randox Laboratories Ltd., Enzo Biochem Inc., Creative Diagnostics Inc., Merck Co. & KGaA, Beckman Coulter Inc., QIAGEN N.V., PerkinElmer Inc., Tosoh Corporation, Eurolyser Diagnostica GmbH, Cortez Diagnostics Inc., DIALAB GmbH, CardioGenics Holdings Inc., Lepu Medical Technology (Beijing) Co. Ltd., Biosynex SA, Charles River Laboratories, Quidel Corporation, Life Diagnostics, Trinity Biotech plc, Guangzhou Wondfo Biotech Co. Ltd., Nova Biomedical Corp., Ortho Clinical Diagnostics Inc., Sekisui Diagnostics Inc., Abcam plc, Bio-Techne Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
