Direct Oral Anticoagulants (DOACs) Device Market Report 2026

Direct Oral Anticoagulants (DOACs) Device Market Report 2026
Global Outlook – By Product (Factor Xa Inhibitors, Direct Thrombin Inhibitors), By Disease (Atrial Fibrillation, Deep Vein Thrombosis, Pulmonary Embolism, Heart Attacks, Post-Surgical Thromboprophylaxis, Other Diseases), By Application (Stroke Prevention, Venous Thromboembolism Prevention, Treatment Of Acute Thrombotic Events, Long-Term Prophylaxis, Other Applications), By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Homecare Settings, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Direct Oral Anticoagulants (DOACs) Device Market Overview
• Direct Oral Anticoagulants (DOACs) Device market size has reached to $32.8 billion in 2025 • Expected to grow to $49.48 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Rising Cardiovascular Disease Prevalence Fueling The Growth Of The Market Due To Increasing Need For Effective Stroke And Clot Prevention • Market Trend: Innovative Automated Assays Enhance Accuracy And Efficiency In Anticoagulant Monitoring • North America was the largest region in 2025.What Is Covered Under Direct Oral Anticoagulants (DOACs) Device Market?
Direct oral anticoagulants device refers to medical instruments or diagnostic tools used to monitor and manage the blood-thinning effects of direct oral anticoagulant medications. These devices help clinicians assess drug levels and anticoagulation status in patients taking medications such as rivaroxaban, apixaban, edoxaban, or dabigatran, ensuring safe and effective treatment. The main products of direct oral anticoagulants (DOACs) device are factor Xa inhibitors and direct thrombin inhibitors. Factor Xa inhibitors refer to a class of anticoagulant medications that work by selectively blocking the activity of factor Xa, an essential enzyme in the blood coagulation cascade, thereby preventing the formation of harmful blood clots. These drugs are commonly used to manage conditions such as atrial fibrillation, deep vein thrombosis, pulmonary embolism, heart attacks, post-surgical thromboprophylaxis, and other diseases. Their applications span across stroke prevention, venous thromboembolism prevention, treatment of acute thrombotic events, long-term prophylaxis, and others, and are used by several end users, including hospitals, ambulatory surgical centers, specialty clinics, homecare settings, and others.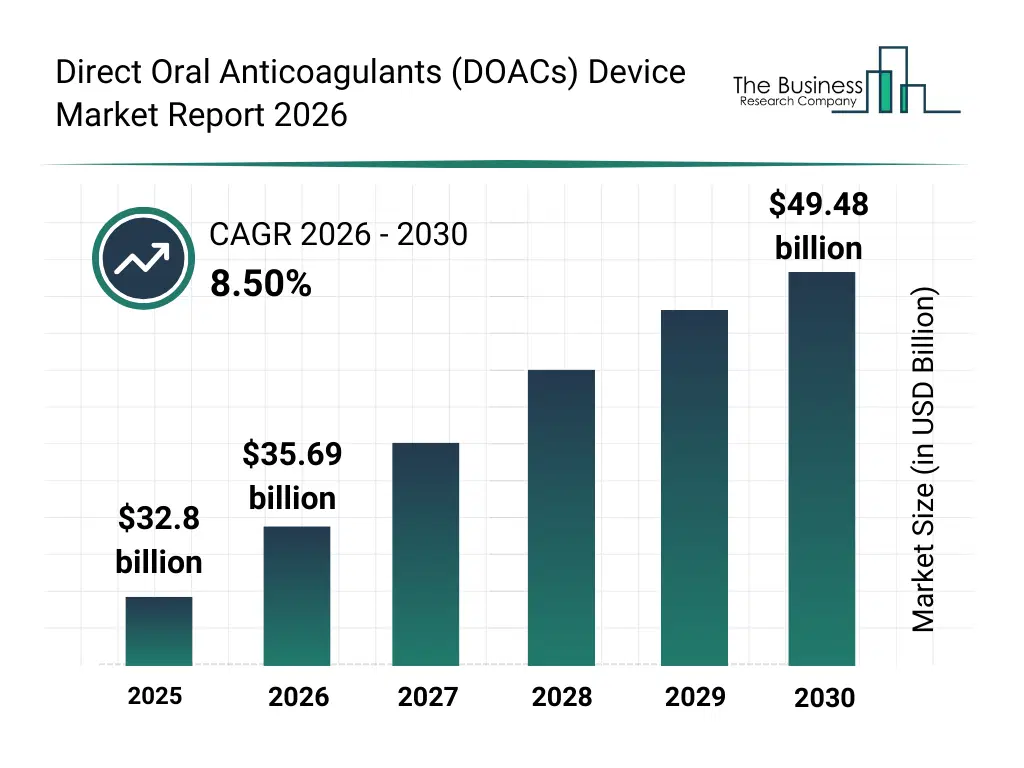
What Is The Direct Oral Anticoagulants (DOACs) Device Market Size and Share 2026?
The direct oral anticoagulants (doacs) device market size has grown strongly in recent years. It will grow from $32.8 billion in 2025 to $35.69 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to limited availability of doac monitoring devices, reliance on hospital-based coagulation tests, growing prevalence of atrial fibrillation and thrombotic disorders, adoption of oral anticoagulant therapies, increasing demand for patient safety and anticoagulation monitoring.What Is The Direct Oral Anticoagulants (DOACs) Device Market Growth Forecast?
The direct oral anticoagulants (doacs) device market size is expected to see strong growth in the next few years. It will grow to $49.48 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to technological advancements in portable and point-of-care devices, integration with digital health platforms and cloud-based monitoring, rising adoption of personalized anticoagulation management, expansion of homecare and specialty clinic services, increasing regulatory approvals for novel monitoring systems. Major trends in the forecast period include growing adoption of point-of-care doac monitoring devices, increasing use of factor xa and direct thrombin inhibitor monitoring, rising awareness of stroke and thromboembolism prevention, expansion of homecare and ambulatory monitoring solutions, enhanced focus on accuracy, sensitivity, and rapid testing capabilities.Global Direct Oral Anticoagulants (DOACs) Device Market Segmentation
1) By Product: Factor Xa Inhibitors, Direct Thrombin Inhibitors 2) By Disease: Atrial Fibrillation, Deep Vein Thrombosis, Pulmonary Embolism, Heart Attacks, Post-Surgical Thromboprophylaxis, Other Diseases 3) By Application: Stroke Prevention, Venous Thromboembolism Prevention, Treatment Of Acute Thrombotic Events, Long-Term Prophylaxis, Other Applications 4) By End User: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Homecare Settings, Other End Users Subsegments: 1) By Factor Xa Inhibitors: Rivaroxaban Monitoring Devices, Apixaban Monitoring Devices, Edoxaban Monitoring Devices, Betrixaban Monitoring Devices 2) By Direct Thrombin Inhibitors: Dabigatran Etexilate, Dabigatran MesylateWhat Is The Driver Of The Direct Oral Anticoagulants (DOACs) Device Market?
The increasing prevalence of cardiovascular disorders is expected to propel the growth of the direct oral anticoagulants (DOACs) device market going forward. Cardiovascular disorders refer to a range of conditions that impact the heart and blood vessels, often leading to issues such as heart attacks, strokes, or other circulatory system problems. Cardiovascular disorders are rising due to increasingly sedentary lifestyles, which lead to weight gain, elevated blood pressure, and weakened cardiovascular function over time. Direct oral anticoagulants (DOACs) help manage cardiovascular disorders by effectively lowering the risk of stroke and blood clots in patients with atrial fibrillation, offering reliable anticoagulation without the need for frequent blood tests. For instance, in October 2024, according to the Centers for Disease Control and Prevention, a US-based government agency, in 2023, cardiovascular disease claimed 919,032 lives, accounting for one in every three deaths. Coronary heart disease remained the most prevalent form of heart disease, and notably, about one in six deaths from cardiovascular diseases occurred among adults younger than 65 years. Therefore, the increasing prevalence of cardiovascular disorders is driving the growth of the direct oral anticoagulants (DOACs) device industry.Key Players In The Global Direct Oral Anticoagulants (DOACs) Device Market
Major companies operating in the direct oral anticoagulants (doacs) device market are Pfizer Inc., Roche Diagnostics Corporation, Bristol-Myers Squibb Company, Siemens Healthcare GmbH, Becton Dickinson And Company, Boehringer Ingelheim International GmbH, Teva Pharmaceutical Industries Limited, Daiichi Sankyo Co. Ltd, Sysmex Corporation, Dr. Reddy’s Laboratories Limited, Werfen S.A., Alembic Pharmaceuticals Limited, Diagnostica Stago SAS, Helena Laboratories Corporation, iLine Microsystems S.L., Technoclone GmbH, Anthos Therapeutics Inc., FLoBio LLC, Perosphere Technologies Inc., DOASENSE GmbH.Global Direct Oral Anticoagulants (DOACs) Device Market Trends and Insights
Major companies operating in the direct oral anticoagulants (DOACs) device market are focusing on developing advanced solutions, such as automated coagulation assays, to improve the accuracy, efficiency, and consistency of direct oral anticoagulant level monitoring in clinical laboratories. Automated coagulation assays are laboratory tests performed by machines that measure how quickly blood clots to help diagnose bleeding or clotting disorders. For instance, in February 2024, Roche Holding AG, a Switzerland-based pharmaceutical company, launched three new coagulation tests specifically designed for Factor Xa inhibitors. These tests enhance clinicians' ability to accurately evaluate the anticoagulation status of patients taking direct Factor Xa inhibitors such as rivaroxaban and apixaban. They aid clinical decision-making by precisely measuring the anti-Xa activity of apixaban, edoxaban, rivaroxaban, and heparin in critical scenarios like bleeding, overdose, or urgent surgery. Utilizing Roche’s reagent cassette technology on cobas t analyzers, these automated tests deliver high-quality results, minimize manual handling, and streamline laboratory workflow efficiency.What Are Latest Mergers And Acquisitions In The Direct Oral Anticoagulants (DOACs) Device Market?
In July 2024, Perosphere Technologies Inc., a US-based medical diagnostics company, partnered with CoRRect Medical to exclusively distribute its reagent-free point-of-care coagulometer in Germany. With this partnership, Perosphere aims to address unmet needs in coagulation testing by enabling rapid, bedside diagnosis of anticoagulation status, particularly for patients on direct oral anticoagulants, thus improving treatment decisions, enhancing patient outcomes, and reducing healthcare costs. CoRRect Medical is a Germany-based medical device company.Regional Outlook
North America was the largest region in the direct oral anticoagulants (DOACs) device market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Direct Oral Anticoagulants (DOACs) Device Market?
The direct oral anticoagulants (DOACs) device market consists of sales of drug delivery devices, monitoring devices, adherence management tools and digital health platforms. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Direct Oral Anticoagulants (DOACs) Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $35.69 billion |
| Revenue Forecast In 2035 | $49.48 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Disease, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Roche Diagnostics Corporation, Bristol-Myers Squibb Company, Siemens Healthcare GmbH, Becton Dickinson And Company, Boehringer Ingelheim International GmbH, Teva Pharmaceutical Industries Limited, Daiichi Sankyo Co. Ltd, Sysmex Corporation, Dr. Reddy’s Laboratories Limited, Werfen S.A., Alembic Pharmaceuticals Limited, Diagnostica Stago SAS, Helena Laboratories Corporation, iLine Microsystems S.L., Technoclone GmbH, Anthos Therapeutics Inc., FLoBio LLC, Perosphere Technologies Inc., DOASENSE GmbH. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Direct Oral Anticoagulants (DOACs) Device Market Report 2026 market was valued at $32.8 billion in 2025, increased to $35.69 billion in 2026, and is projected to reach $49.48 billion by 2030.
The expected CAGR for the Direct Oral Anticoagulants (DOACs) Device Market Report 2026 market during the forecast period 2025–2030 is 8.5%.
Major growth driver of the market includes: Rising Cardiovascular Disease Prevalence Fueling The Growth Of The Market Due To Increasing Need For Effective Stroke And Clot Prevention in the Direct Oral Anticoagulants (DOACs) Device Market Report 2026 market. For further insights on this market,
The direct oral anticoagulants (doacs) device market covered in this report is segmented –
1) By Product: Factor Xa Inhibitors, Direct Thrombin Inhibitors
2) By Disease: Atrial Fibrillation, Deep Vein Thrombosis, Pulmonary Embolism, Heart Attacks, Post-Surgical Thromboprophylaxis, Other Diseases
3) By Application: Stroke Prevention, Venous Thromboembolism Prevention, Treatment Of Acute Thrombotic Events, Long-Term Prophylaxis, Other Applications
4) By End User: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Homecare Settings, Other End Users Subsegments:
1) By Factor Xa Inhibitors: Rivaroxaban Monitoring Devices, Apixaban Monitoring Devices, Edoxaban Monitoring Devices, Betrixaban Monitoring Devices
2) By Direct Thrombin Inhibitors: Dabigatran Etexilate, Dabigatran Mesylate
1) By Product: Factor Xa Inhibitors, Direct Thrombin Inhibitors
2) By Disease: Atrial Fibrillation, Deep Vein Thrombosis, Pulmonary Embolism, Heart Attacks, Post-Surgical Thromboprophylaxis, Other Diseases
3) By Application: Stroke Prevention, Venous Thromboembolism Prevention, Treatment Of Acute Thrombotic Events, Long-Term Prophylaxis, Other Applications
4) By End User: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Homecare Settings, Other End Users Subsegments:
1) By Factor Xa Inhibitors: Rivaroxaban Monitoring Devices, Apixaban Monitoring Devices, Edoxaban Monitoring Devices, Betrixaban Monitoring Devices
2) By Direct Thrombin Inhibitors: Dabigatran Etexilate, Dabigatran Mesylate
Major trend in this market includes: Innovative Automated Assays Enhance Accuracy And Efficiency In Anticoagulant Monitoring For further insights on this market,
Request for SampleMajor companies operating in the Direct Oral Anticoagulants (DOACs) Device Market Report 2026 market are Major companies operating in the direct oral anticoagulants (doacs) device market are Pfizer Inc., Roche Diagnostics Corporation, Bristol-Myers Squibb Company, Siemens Healthcare GmbH, Becton Dickinson And Company, Boehringer Ingelheim International GmbH, Teva Pharmaceutical Industries Limited, Daiichi Sankyo Co. Ltd, Sysmex Corporation, Dr. Reddy’s Laboratories Limited, Werfen S.A., Alembic Pharmaceuticals Limited, Diagnostica Stago SAS, Helena Laboratories Corporation, iLine Microsystems S.L., Technoclone GmbH, Anthos Therapeutics Inc., FLoBio LLC, Perosphere Technologies Inc., DOASENSE GmbH.
North America was the largest region in the direct oral anticoagulants (DOACs) device market in 2025. The regions covered in the direct oral anticoagulants (doacs) device market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
