
Endoscopy Sterilization Market Report 2026
Global Outlook – By Product Type (Device, Consumables And Accessories), By Sterilization Method (Hydrogen Peroxide Sterilization, Ethylene Oxide (EtO) Sterilization, Liquid Chemical Sterilization, Other Sterilization Methods), By End Use (Hospitals, Ambulatory Surgical Centers, Other End Uses) – Market Size, Trends, Strategies, and Forecast to 2035
Endoscopy Sterilization Market Overview
• Endoscopy Sterilization market size has reached to $1.25 billion in 2025 • Expected to grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 8.3% • Growth Driver: Rising Hospital-Acquired Infections Fueling The Growth Of The Market Due To Increasing Demand For Infection Control Solutions • Market Trend: Technological Advancement Of Integrated Endoscope Washer-Disinfectors To Enhance Reprocessing Efficiency And Safety • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Endoscopy Sterilization Market?
Endoscopy sterilization is the process of eliminating all forms of microbial life from endoscopic instruments to ensure patient safety. It involves thorough cleaning followed by chemical or thermal disinfection to prevent infection transmission. Endoscopy sterilization is crucial due to the complex design and reusability of endoscopic devices. The main product types of endoscopy sterilization are devices, consumables, and accessories. An endoscopy sterilization device is specialized equipment designed to clean, disinfect, and sterilize endoscopic instruments to ensure they are safe and free from harmful microorganisms before reuse. The sterilization methods include hydrogen peroxide sterilization, ethylene oxide (EtO) sterilization, liquid chemical sterilization, and others. The key end users are hospitals, ambulatory surgical centers, and others.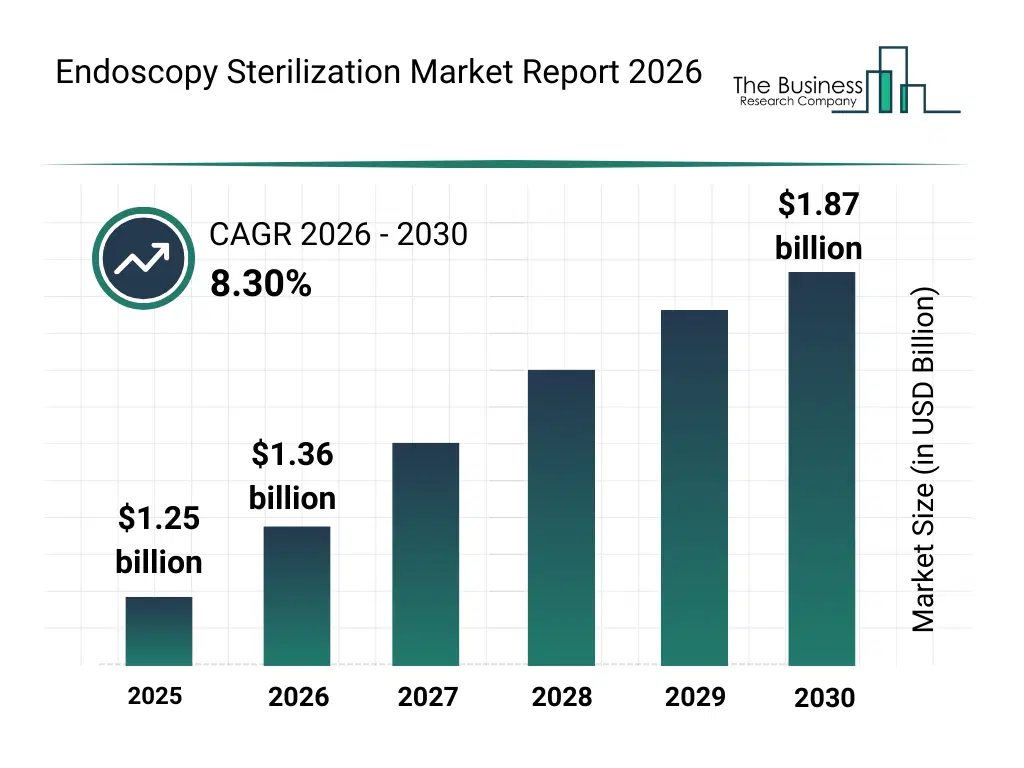
What Is The Endoscopy Sterilization Market Size and Share 2026?
The endoscopy sterilization market size has grown strongly in recent years. It will grow from $1.25 billion in 2025 to $1.36 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to limited automation in endoscope sterilization, reliance on manual cleaning procedures, growing hospital and surgical centers, rising awareness of nosocomial infections, conventional sterilization methods dominance.What Is The Endoscopy Sterilization Market Growth Forecast?
The endoscopy sterilization market size is expected to see strong growth in the next few years. It will grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 8.3%. The growth in the forecast period can be attributed to development of automated sterilization systems, expansion of minimally invasive procedures, increasing use of hydrogen peroxide and etoh sterilization, rising demand for consumables and accessories, integration of smart monitoring and compliance tracking. Major trends in the forecast period include rising adoption of automated endoscope reprocessors (aer), increased focus on low-temperature sterilization methods, growing use of enzymatic detergents and disinfectant solutions, expansion of sterilization accessories and consumables, enhanced regulatory compliance and patient safety measures.Global Endoscopy Sterilization Market Segmentation
1) By Product Type: Device, Consumables And Accessories 2) By Sterilization Method: Hydrogen Peroxide Sterilization, Ethylene Oxide (EtO) Sterilization, Liquid Chemical Sterilization, Other Sterilization Methods 3) By End Use: Hospitals, Ambulatory Surgical Centers, Other End Uses Subsegments: 1) By Device: Low-Temperature Sterilizers, High-Temperature Sterilizers, Ultrasonic Cleaners, Automated Endoscope Reprocessors (AER), Washer Disinfectors, Drying And Storage Cabinets 2) By Consumables And Accessories: Sterilization Indicators, Sterilization Wraps And Pouches, Cleaning Brushes, Enzymatic Detergents, Disinfectant Solutions, Lubricants And Oiling AgentsWhat Is The Driver Of The Endoscopy Sterilization Market?
The rising incidence of hospital-acquired infections (HAIs) is expected to propel the growth of the endoscopy sterilization market going forward. Hospital-acquired infections (HAIs) are infections that patients develop during their stay in a healthcare facility, typically after 48 hours of admission, that were not present or incubating at the time of arrival. The growth in the incidence of hospital-acquired infections (HAIs) is largely attributed to the overuse of broad-spectrum antibiotics, which contributes to antimicrobial resistance and enables drug-resistant pathogens to thrive in healthcare settings. The hospital-acquired infections (HAIs) have intensified the need for reliable endoscopy sterilization solutions, as improperly reprocessed endoscopic devices can serve as critical transmission sources for multidrug-resistant pathogens during diagnostic and surgical procedures. For instance, in March 2025, according to the UK Health Security Agency (UKHSA), a UK-based national government public health agency, healthcare-associated infections were present in 7.6% of hospital patients in 2023. Therefore, the rising incidence of hospital-acquired infections is driving the growth of the endoscopy sterilization industry.Key Players In The Global Endoscopy Sterilization Market
Major companies operating in the endoscopy sterilization market are 3M Company, Ecolab Inc., Olympus Corporation, STERIS plc, Getinge AB, Shinva Medical Instrument Co. Ltd., Belimed AG, ARC Group of Companies Inc., AURORA Cold Plasma Sterilisation (Aurora Sterilisation), Matachana S.A., Bionics Scientific, Tuttnauer Europe B.V., Life Science Outsourcing Inc., H.W. Andersen Products Ltd., MMM Group, Cistron Systems, MDD Company Gmbh, UV Smart Technologies B.V., HUMAN MEDITEK GmbH, Sharkclave SystemsGlobal Endoscopy Sterilization Market Trends and Insights
Major companies operating in the endoscopy sterilization market are focusing on developing technological advancements such as fully integrated endoscope washer-disinfectors with automated wash, disinfection, and documentation capabilities to enhance reprocessing efficiency and safety. Endoscope washer-disinfector systems combine cleaning, disinfecting, and data tracking in one closed-loop unit to minimize human error and ensure consistent hygiene standards. For instance, in June 2023, Olympus Corporation, a Japan-based manufacturer of optics and reprography products, launched the new EndoThermo Disinfector (ETD) Endoscope Washer Disinfector, offering both basic and premium versions. It features faster cycle times, real-time digital monitoring, and sustainable energy-efficient operations to improve workflow and patient safety. The ETD system is designed to meet modern infection control standards while supporting staff with intuitive automation and simplified maintenance.What Are Latest Mergers And Acquisitions In The Endoscopy Sterilization Market?
In November 2023, HOYA Corporation, a Japan-based medical technology company, acquired WASSENBURG Medical B.V. for an undisclosed amount. Through this acquisition, HOYA strengthens its capabilities in infection prevention by integrating WASSENBURG’s expertise in washer-disinfectors, drying cabinets, consumables, and traceability systems into its PENTAX Medical division. WASSENBURG Medical B.V. is a Netherlands-based provider of comprehensive endoscope reprocessing solutions, including endoscopic sterilization.Regional Outlook
North America was the largest region in the endoscopy sterilization market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Endoscopy Sterilization Market?
The endoscopy sterilization market consists of revenues earned by entities by providing services such as endoscope cleaning, high-level disinfection, sterilization, drying, storage, and quality assurance monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. The endoscopy sterilization market includes sales of automated preprocessors, sterilization equipment, chemical disinfectants, drying cabinets, and tracking systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Endoscopy Sterilization Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.36 billion |
| Revenue Forecast In 2035 | $1.87 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Sterilization Method, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | 3M Company, Ecolab Inc., Olympus Corporation, STERIS plc, Getinge AB, Shinva Medical Instrument Co. Ltd., Belimed AG, ARC Group of Companies Inc., AURORA Cold Plasma Sterilisation (Aurora Sterilisation), Matachana S.A., Bionics Scientific, Tuttnauer Europe B.V., Life Science Outsourcing Inc., H.W. Andersen Products Ltd., MMM Group, Cistron Systems, MDD Company Gmbh, UV Smart Technologies B.V., HUMAN MEDITEK GmbH, Sharkclave Systems |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
