Near-Patient Molecular Solutions Market Report 2026

Near-Patient Molecular Solutions Market Report 2026
Global Outlook – By Product Type (Infectious Diseases Testing Kits, Urinalysis Testing Kits, Tumor Or Cancer Markers, Pregnancy And Fertility, Hematology Testing Kits, Glucose Monitoring Kits, Drugs-Of-Abuse Testing Kits, Coagulation Monitoring Kits, Cardiometabolic Monitoring Kits, Other Product Types), By Technology (Polymerase Chain Reaction (PCR)-Based, Genetic Sequencing-Based, Hybridization-Based, Microarray-Based), By End-User (Hospitals And Clinics, Ambulatory Care Centers, Research Laboratories, Home Care Settings, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Near-Patient Molecular Solutions Market Overview
• Near-Patient Molecular Solutions market size has reached to $4.54 billion in 2025 • Expected to grow to $7.17 billion in 2030 at a compound annual growth rate (CAGR) of 9.5% • Growth Driver: Surge In Demand For Home‑based Point‑Of‑Care Devices Fueling The Growth Of The Market Due To Increasing Consumer Autonomy And Convenience • Market Trend: Integration Of Advanced Molecular Amplification Technologies To Enhance Near-Patient Diagnostic Performance • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Near-Patient Molecular Solutions Market?
Near-patient molecular solutions are diagnostic tests that use molecular biology techniques, such as deoxyribonucleic acid (DNA) or ribonucleic acid (RNA) detection, and are performed at or very close to where the patient receives care. These tests analyze small biological samples on-site instead of sending them to a central laboratory, allowing rapid and accurate detection of pathogens or biomarkers using portable or simplified instruments. By delivering quick and reliable results in outpatient, emergency, or remote settings, they support timely treatment decisions and better health outcomes. The main product types of near-patient molecular solutions include infectious disease testing kits, urinalysis testing kits, tumor or cancer markers, pregnancy and fertility, hematology testing kits, glucose monitoring kits, drugs-of-abuse testing kits, coagulation monitoring kits, cardiometabolic monitoring kits, and others. Infectious disease testing kits are diagnostic tools designed to rapidly detect and identify pathogens such as bacteria, viruses, or parasites in patient samples to aid in timely disease diagnosis and treatment. The different technologies involved are polymerase chain reaction (PCR)-based, genetic sequencing-based, hybridization-based, and are used for several end-users, including hospitals and clinics, ambulatory care centers, research laboratories, home care settings, and others.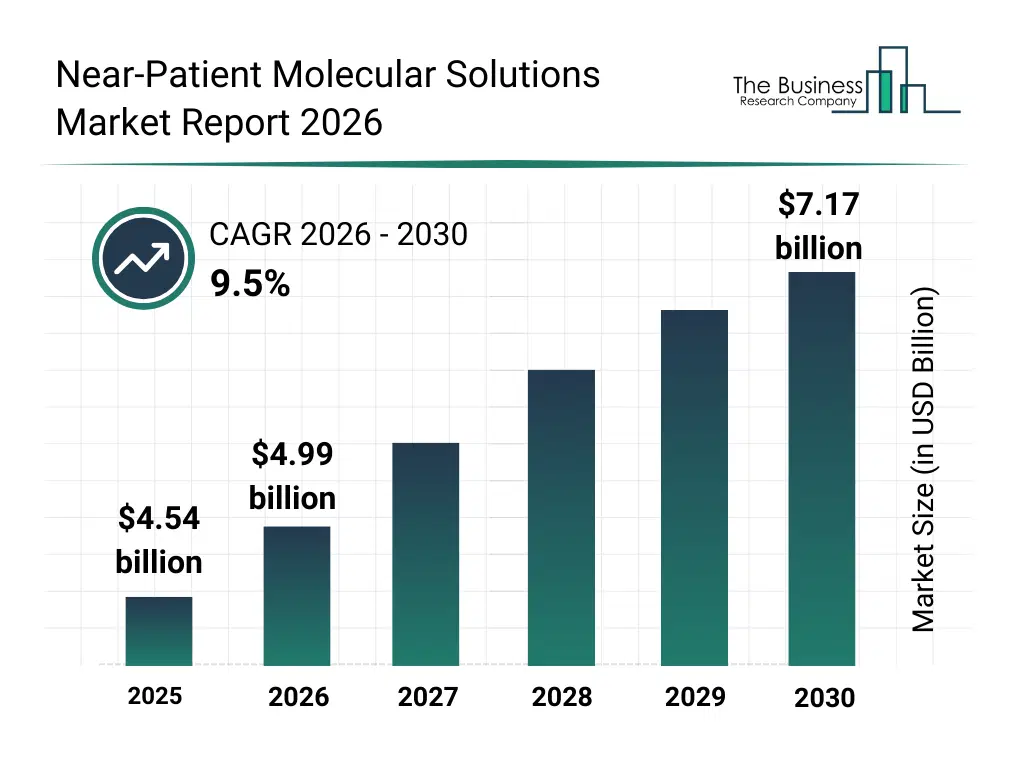
What Is The Near-Patient Molecular Solutions Market Size and Share 2026?
The near-patient molecular solutions market size has grown strongly in recent years. It will grow from $4.54 billion in 2025 to $4.99 billion in 2026 at a compound annual growth rate (CAGR) of 9.8%. The growth in the historic period can be attributed to expansion of molecular diagnostic techniques, demand for rapid test turnaround times, decentralization of laboratory services, growth in outpatient diagnostic settings, adoption of portable diagnostic instruments.What Is The Near-Patient Molecular Solutions Market Growth Forecast?
The near-patient molecular solutions market size is expected to see strong growth in the next few years. It will grow to $7.17 billion in 2030 at a compound annual growth rate (CAGR) of 9.5%. The growth in the forecast period can be attributed to increasing focus on personalized medicine, rising demand for home and remote testing solutions, technological advancements in miniaturized molecular platforms, expansion of infectious disease surveillance, integration with digital health ecosystems. Major trends in the forecast period include increasing adoption of point-of-care molecular testing devices, rising demand for rapid infectious disease diagnostics, growing integration of portable PCR platforms, expansion of decentralized diagnostic infrastructure, enhanced focus on near-patient testing accuracy.Global Near-Patient Molecular Solutions Market Segmentation
1) By Product Type: Infectious Diseases Testing Kits, Urinalysis Testing Kits, Tumor Or Cancer Markers, Pregnancy And Fertility, Hematology Testing Kits, Glucose Monitoring Kits, Drugs-Of-Abuse Testing Kits, Coagulation Monitoring Kits, Cardiometabolic Monitoring Kits, Other Product Types 2) By Technology: Polymerase Chain Reaction (PCR)-Based, Genetic Sequencing-Based, Hybridization-Based, Microarray-Based 3) By End-User: Hospitals And Clinics, Ambulatory Care Centers, Research Laboratories, Home Care Settings, Other End-Users Subsegments: 1) By Infectious Diseases Testing Kits: Hepatitis Testing Kits, Human Immunodeficiency Virus Testing Kits, Influenza Testing Kits, COVID-19 Testing Kits 2) By Urinalysis Testing Kits: Protein Testing Kits, Glucose Testing Kits, Ketone Testing Kits, Nitrite Testing Kits 3) By Tumor Or Cancer Markers: Prostate-Specific Antigen Testing Kits, Carcinoembryonic Antigen Testing Kits, Cancer Antigen 125 Testing Kits, Alpha-Fetoprotein Testing Kits 4) By Pregnancy And Fertility: Ovulation Testing Kits, Early Pregnancy Testing Kits, Digital Pregnancy Testing Kits, Home Fertility Monitoring Kits 5) By Hematology Testing Kits: Complete Blood Count Testing Kits, Hemoglobin Testing Kits, White Blood Cell Count Testing Kits, Platelet Count Testing Kits 6) By Glucose Monitoring Kits: Self-Monitoring Blood Glucose Kits, Continuous Glucose Monitoring Kits, Non-Invasive Glucose Testing Kits, Disposable Glucose Test Strips 7) By Drugs-Of-Abuse Testing Kits: Urine Drug Testing Kits, Saliva Drug Testing Kits, Hair Drug Testing Kits, Blood Drug Testing Kits 8) By Coagulation Monitoring Kits: Prothrombin Time Testing Kits, International Normalized Ratio Testing Kits, Activated Partial Thromboplastin Time Testing Kits, D-Dimer Testing Kits 9) By Cardiometabolic Monitoring Kits: Cholesterol Testing Kits, Triglyceride Testing Kits, Low-Density Lipoprotein Testing Kits, High-Density Lipoprotein Testing Kits 10) By Other Product Types: Allergy Testing Kits, Thyroid Function Testing Kits, Vitamin Deficiency Testing Kits, Liver Function Testing KitsWhat Is The Driver Of The Near-Patient Molecular Solutions Market?
Growing demand for home-based point-of-care devices is expected to propel the growth of the near patient molecular solutions market going forward. Home‑based point‑of‑care devices are diagnostic testing systems that patients or non‑laboratory users operate at or near their home, enabling sample collection, analysis, or result reading outside a conventional central laboratory setting and in or close to the place of care. The growing demand for home‑based point-of-care devices is increasing due to the rising consumer preference for convenient, rapid, and accessible health monitoring outside traditional healthcare settings. Near-patient molecular solutions support the home-based point-of-care devices by providing accurate, rapid, and portable molecular diagnostics that can be reliably used outside traditional laboratory settings. For instance, in February 2023, the US Food & Drug Administration (FDA), a US-based government agency, reported in its 2023 COVID-19 testing update that there was one Emergency Use Authorization (EUA) for a molecular prescription at-home test, two EUAs for antigen prescription at-home tests, 26 EUAs for antigen over-the-counter (OTC) at-home tests, and four EUAs for molecular OTC at-home tests. Therefore, growing demand for home-based point-of-care devices is driving the growth of the near-patient molecular solutions industry.Key Players In The Global Near-Patient Molecular Solutions Market
Major companies operating in the near-patient molecular solutions market are Bosch Healthcare Solutions GmbH, Roche Holding AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Hologic Inc., bioMérieux SA, Sysmex Corporation, QuidelOrtho Corporation, QIAGEN NV, DiaSorin SpA, OraSure Technologies Inc., Molbio Diagnostics Pvt Ltd, Cue Health Inc., Biocartis Group NV, Vela Diagnostics Pte Ltd, Co-Diagnostics Inc., Hyris Ltd, Genedrive plc.Global Near-Patient Molecular Solutions Market Trends and Insights
Major companies operating in the near-patient molecular solutions market are focusing on incorporating technological advancements such as isothermal nucleic acid amplification to enhance testing efficiency, portability, and diagnostic accuracy. Isothermal nucleic acid amplification is an advanced molecular diagnostic technology that amplifies target nucleic acid sequences at a constant temperature, eliminating the need for thermal cycling as in conventional polymerase chain reaction (PCR), thereby enabling rapid, sensitive, and compact molecular testing. For instance, in June 2023, Cue Health Inc., a US-based healthcare technology company specializing in molecular diagnostics, received FDA marketing authorization for the Cue COVID-19 Molecular Test, an isothermal amplification-based diagnostic device designed for at-home and point-of-care detection of SARS-CoV-2 RNA. The system features a single-use cartridge, a Bluetooth-enabled reader, and a mobile app interface that guides users through testing and delivers molecular results in about 20 minutes. The Cue COVID-19 molecular test improves access to accurate molecular-level diagnostics, reduces turnaround time, and supports real-time health decision-making outside traditional laboratory environments.What Are Latest Mergers And Acquisitions In The Near-Patient Molecular Solutions Market?
In July 2024, Roche Holding AG, a Switzerland-based provider of in-vitro diagnostics and pharmaceutical solutions, acquired LumiraDx’s point-of-care (POC) technology business for $353.5 million. With this acquisition, Roche gains access to LumiraDx’s portable, multi-assay diagnostic platform, enhancing its decentralized and near-patient testing capabilities, broadening its presence in primary care and community-based settings, and improving delivery of rapid and reliable diagnostic results outside traditional laboratory environments. LumiraDx Group Limited is a UK-based provider of next-generation point-of-care diagnostics known for its compact instrument that integrates immunoassay, clinical chemistry, and emerging molecular testing capabilities in near-patient applications.Regional Outlook
North America was the largest region in the near-patient molecular solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Near-Patient Molecular Solutions Market?
The near-patient molecular solutions market consists of sales of sample preparation devices, nucleic acid extraction kits, portable molecular analyzers, isothermal amplification kits, microfluidic cartridges, and multiplex detection panels. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Near-Patient Molecular Solutions Market Report 2026?
The near-patient molecular solutions market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the near-patient molecular solutions industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Near-Patient Molecular Solutions Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.99 billion |
| Revenue Forecast In 2035 | $7.17 billion |
| Growth Rate | CAGR of 9.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Bosch Healthcare Solutions GmbH, Roche Holding AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Hologic Inc., bioMérieux SA, Sysmex Corporation, QuidelOrtho Corporation, QIAGEN NV, DiaSorin SpA, OraSure Technologies Inc., Molbio Diagnostics Pvt Ltd, Cue Health Inc., Biocartis Group NV, Vela Diagnostics Pte Ltd, Co-Diagnostics Inc., Hyris Ltd, Genedrive plc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Near-Patient Molecular Solutions market was valued at $4.54 billion in 2025, increased to $4.99 billion in 2026, and is projected to reach $7.17 billion by 2030.
The global Near-Patient Molecular Solutions market is expected to grow at a CAGR of 9.5% from 2026 to 2035 to reach $7.17 billion by 2035.
Some Key Players in the Near-Patient Molecular Solutions market Include, Bosch Healthcare Solutions GmbH, Roche Holding AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Hologic Inc., bioMérieux SA, Sysmex Corporation, QuidelOrtho Corporation, QIAGEN NV, DiaSorin SpA, OraSure Technologies Inc., Molbio Diagnostics Pvt Ltd, Cue Health Inc., Biocartis Group NV, Vela Diagnostics Pte Ltd, Co-Diagnostics Inc., Hyris Ltd, Genedrive plc. .
Major trend in this market includes: Integration Of Advanced Molecular Amplification Technologies To Enhance Near-Patient Diagnostic Performance. For further insights on this market.
Request for SampleNorth America was the largest region in the near-patient molecular solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the near-patient molecular solutions market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
