
Viral Vector-Based Gene Therapy Market Report 2026
Global Outlook – By Vector Type (Adenoviral Vectors, Adeno-Associated Viral Vectors, Lentiviral Vectors, Retroviral Vectors), By Delivery Method (Intravenous Injection, Intrathecal Injection, Intramuscular Injection, Subcutaneous Injection, Topical), By Application (Oncology, Genetic Disorders, Cardiovascular Diseases, Infectious Diseases, Neurological Disorders, Other Applications), By End-User (Hospitals, Clinical Research Organizations (CROs), Academic And Research Institutes, Pharmaceutical And Biotechnology Companies, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Viral Vector-Based Gene Therapy Market Overview
• Viral Vector-Based Gene Therapy market size has reached to $6.77 billion in 2025 • Expected to grow to $14.81 billion in 2030 at a compound annual growth rate (CAGR) of 16.9% • Growth Driver: Increasing Prevalence Of Genetic Diseases Fueling The Growth Of The Market Due To Rising Diagnosis And Awareness • Market Trend: Integrated Systems Boost Efficiency And Reduce Costs • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Vector-Based Gene Therapy Market?
Viral vector-based gene therapy is a medical approach that uses engineered viruses to deliver therapeutic genes into a patient’s cells to treat or prevent diseases. The viral vectors are modified to be safe and replication-deficient while efficiently transferring genetic material to target cells. This therapy enables correction of genetic defects, modulation of gene expression, or stimulation of immune responses to achieve long-lasting therapeutic effects. The main vector types of viral vector-based gene therapy are adenoviral vectors, adeno-associated viral vectors, lentiviral vectors, retroviral vectors, and others. Adenoviral vectors are engineered viruses derived from adenoviruses that are modified to safely deliver therapeutic genes into target cells without replicating and are commonly used in gene therapy and vaccine applications. It is delivered through various routes, including intravenous injection, intrathecal injection, intramuscular injection, subcutaneous injection, and topical application. It finds applications in oncology, genetic disorders, cardiovascular diseases, infectious diseases, neurological disorders, and others, serving end users such as hospitals, clinical research organizations (CROs), academic and research institutes, pharmaceutical and biotechnology companies, and others.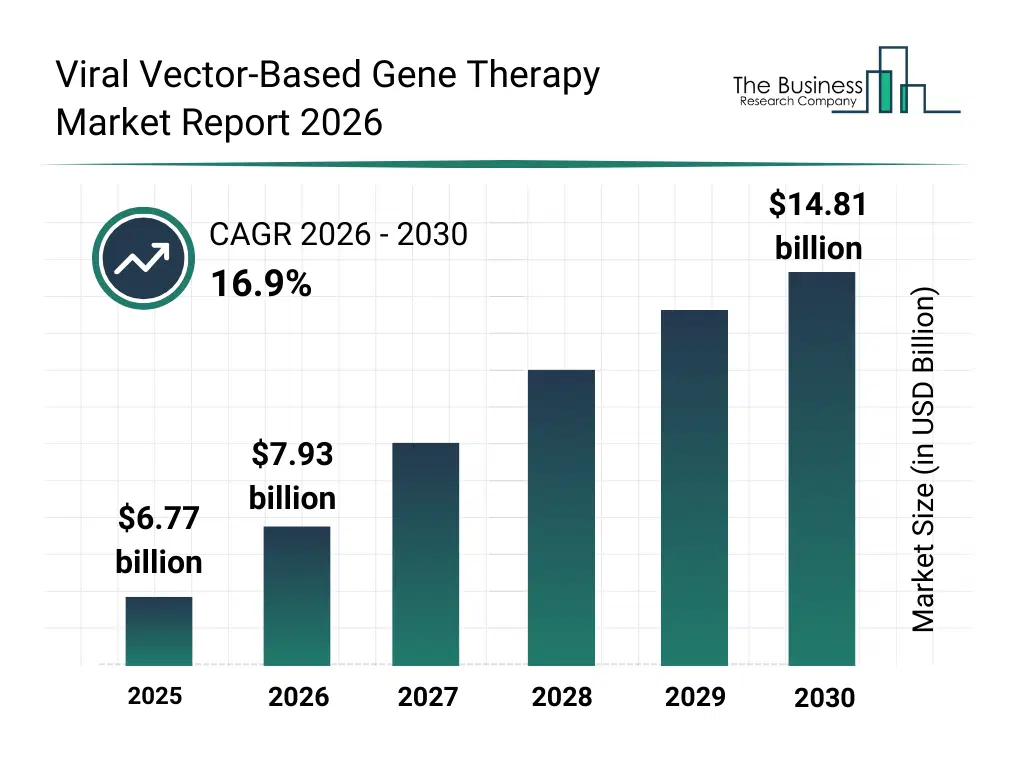
What Is The Viral Vector-Based Gene Therapy Market Size and Share 2026?
The viral vector-based gene therapy market size has grown rapidly in recent years. It will grow from $6.77 billion in 2025 to $7.93 billion in 2026 at a compound annual growth rate (CAGR) of 17.2%. The growth in the historic period can be attributed to advancements in molecular biology research, increasing understanding of genetic disorders, early clinical successes of viral vector therapies, expansion of orphan drug programs, availability of viral vector platforms.What Is The Viral Vector-Based Gene Therapy Market Growth Forecast?
The viral vector-based gene therapy market size is expected to see rapid growth in the next few years. It will grow to $14.81 billion in 2030 at a compound annual growth rate (CAGR) of 16.9%. The growth in the forecast period can be attributed to increasing approvals of gene therapy products, rising investment in rare disease treatments, expansion of precision medicine initiatives, growing adoption of personalized healthcare, continuous improvements in vector engineering technologies. Major trends in the forecast period include rising adoption of targeted gene therapies, growing development of personalized treatment approaches, expansion of clinical trials for rare diseases, increasing focus on long-term therapeutic efficacy, enhanced emphasis on vector safety and performance.Global Viral Vector-Based Gene Therapy Market Segmentation
1) By Vector Type: Adenoviral Vectors, Adeno-Associated Viral Vectors, Lentiviral Vectors, Retroviral Vectors 2) By Delivery Method: Intravenous Injection, Intrathecal Injection, Intramuscular Injection, Subcutaneous Injection, Topical 3) By Application: Oncology, Genetic Disorders, Cardiovascular Diseases, Infectious Diseases, Neurological Disorders, Other Applications 4) By End-User: Hospitals, Clinical Research Organizations (CROs), Academic And Research Institutes, Pharmaceutical And Biotechnology Companies, Other End-Users Subsegments: 1) By Adenoviral Vectors: Human Adenovirus Serotype Five, Human Adenovirus Serotype Two, Canine Adenovirus 2) By Adeno-Associated Viral Vectors: Adeno-Associated Virus Serotype One, Adeno-Associated Virus Serotype Two, Adeno-Associated Virus Serotype Five, Adeno-Associated Virus Serotype Eight, Other Adeno-Associated Viruses 3) By Lentiviral Vectors: Human Immunodeficiency Virus One, Human Immunodeficiency Virus Two 4) By Retroviral Vectors: Gammaretrovirus, Alpharetrovirus, Spumaretrovirus, Other RetrovirusesWhat Is The Driver Of The Viral Vector-Based Gene Therapy Market?
The increasing prevalence of genetic diseases is expected to propel the growth of the viral vector‑based gene therapy market going forward. Genetic diseases are disorders caused by changes or mutations in DNA, which can be inherited or occur spontaneously, affecting health, development, or body functions. The rising prevalence of genetic diseases is due to increased awareness and advancements in diagnostic technologies, as these enable earlier and more accurate detection of previously undiagnosed conditions. Viral vector-based gene therapy enhances genetic disease management by providing targeted delivery of therapeutic genes, making it ideal for treating inherited disorders. It reduces disease progression by enabling precise and long-lasting correction of genetic defects, improving patient outcomes and quality of life. For instance, in April 2025, according to the International Diabetes Federation (IDF), a Belgium-based non-profit organization, approximately 589 million people were living with diabetes in 2024, and this number is projected to rise to 853 million by 2050. Therefore, the increasing prevalence of genetic diseases is driving the growth of the viral vector‑based gene therapy market.Key Players In The Global Viral Vector-Based Gene Therapy Market
Major companies operating in the viral vector-based gene therapy market are F. Hoffmann-La Roche AG, Novartis AG, BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., Ultragenyx Inc., Rocket Pharmaceuticals Inc., PTC Therapeutics Inc., 4D Molecular Therapeutics Inc., Editas Medicine Inc., Regenxbio Inc., Oxford Biomedica plc, MeiraGTx Holdings plc, Voyager Therapeutics Inc., Solid Biosciences Inc., Sangamo Therapeutics Inc., Adverum Biotechnologies Inc., Passage Bio Inc., Dyno Therapeutics Inc., uniQure N.V., Bluebird Bio Inc.Global Viral Vector-Based Gene Therapy Market Trends and Insights
Major companies operating in the viral vector-based gene therapy market are focusing on developing advanced platforms, such as integrated manufacturing platforms, to boost efficiency, enhance reliability, and reduce the cost and time of therapy production. Integrated manufacturing platforms refer to comprehensive systems that streamline the entire process of producing gene therapies, from vector design to final product, using standardized, high-yield processes. For instance, in October 2024, Forge Biologics Holdings LLC, a US-based biotechnology company, launched the FUEL AAV Manufacturing Platform. It is a proprietary, suspension-based manufacturing system designed to provide high productivity and scalability. It includes a fully integrated plasmid system and optimized processes, enabling robust and consistent production of adeno-associated viral (AAV) vectors for gene therapies, thereby accelerating developer timelines from research to clinical trials.What Are Latest Mergers And Acquisitions In The Viral Vector-Based Gene Therapy Market?
In July 2023, Sartorius AG, a Germany-based company that provides laboratory and biopharmaceutical equipment and services, acquired Polyplus SAS for an undisclosed amount. Through this acquisition, Sartorius aims to strengthen its capabilities in supplying critical transfection reagents and related technologies that enable the efficient production of viral vectors for gene and gene-modified cell therapies. Polyplus SAS is a France-based company that provides upstream technologies, products, and services for the manufacturing of viral vectors used in gene therapies.Regional Outlook
North America was the largest region in the viral vector-based gene therapy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector-Based Gene Therapy Market?
The viral vector-based gene therapy market includes revenues earned by entities through process development, vector design, cell line development, assay development, process optimization, manufacturing, upstream production, and downstream purification. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector-Based Gene Therapy Market Report 2026?
The viral vector-based gene therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector-based gene therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector-Based Gene Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.93 billion |
| Revenue Forecast In 2035 | $14.81 billion |
| Growth Rate | CAGR of 17.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Vector Type, Delivery Method, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Novartis AG, BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., Ultragenyx Inc., Rocket Pharmaceuticals Inc., PTC Therapeutics Inc., 4D Molecular Therapeutics Inc., Editas Medicine Inc., Regenxbio Inc., Oxford Biomedica plc, MeiraGTx Holdings plc, Voyager Therapeutics Inc., Solid Biosciences Inc., Sangamo Therapeutics Inc., Adverum Biotechnologies Inc., Passage Bio Inc., Dyno Therapeutics Inc., uniQure N.V., Bluebird Bio Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
