
Viral Vector Development Service Market Report 2026
Global Outlook – By Vector Type (Adenoviral Vectors, Lentiviral Vectors, Retroviral Vectors, Adeno-Associated Viral Vectors, Other Vector types), By Distribution Channel (Online Sales, Direct Sales, Distributors, Retailers), By Application (Gene Therapy, Vaccinology, Cancer Therapy, Other Applications), By End-User (Pharmaceutical Companies, Academic And Research Institutions) - Market Size, Trends, And Global Forecast 2026-2035
Viral Vector Development Service Market Overview
• Viral Vector Development Service market size has reached to $2.1 billion in 2025 • Expected to grow to $4.82 billion in 2030 at a compound annual growth rate (CAGR) of 18% • Growth Driver: Surge In Demand For Gene Therapies Fueling The Growth Of The Market Due To Increasing Need For Efficient And Targeted Gene Delivery • Market Trend: Innovative Viral Vector Solutions Drive Faster And Cost-Efficient Therapeutic Development • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Viral Vector Development Service Market?
Viral vector development service involves the specialized processes of designing, optimizing, and producing viral vectors used for gene therapy, vaccine, and cell therapy applications. It focuses on ensuring high-quality vector development through process optimization, scalability, and regulatory compliance. This service plays a critical role in accelerating research and clinical advancements by supporting biopharmaceutical companies in bringing gene-based treatments to market efficiently. The main vector types of viral vector development service are adenoviral vectors, lentiviral vectors, retroviral vectors, adeno-associated viral vectors, and others. Adenoviral vectors are engineered viral particles derived from adenoviruses, designed to deliver therapeutic genes into target cells efficiently. It is distributed through online sales, direct sales, distributors, and retailers, and finds applications in gene therapy, vaccinology, cancer therapy, and others. The key end-users include pharmaceutical companies, academic and research institutions, food manufacturers, and cosmetic manufacturers.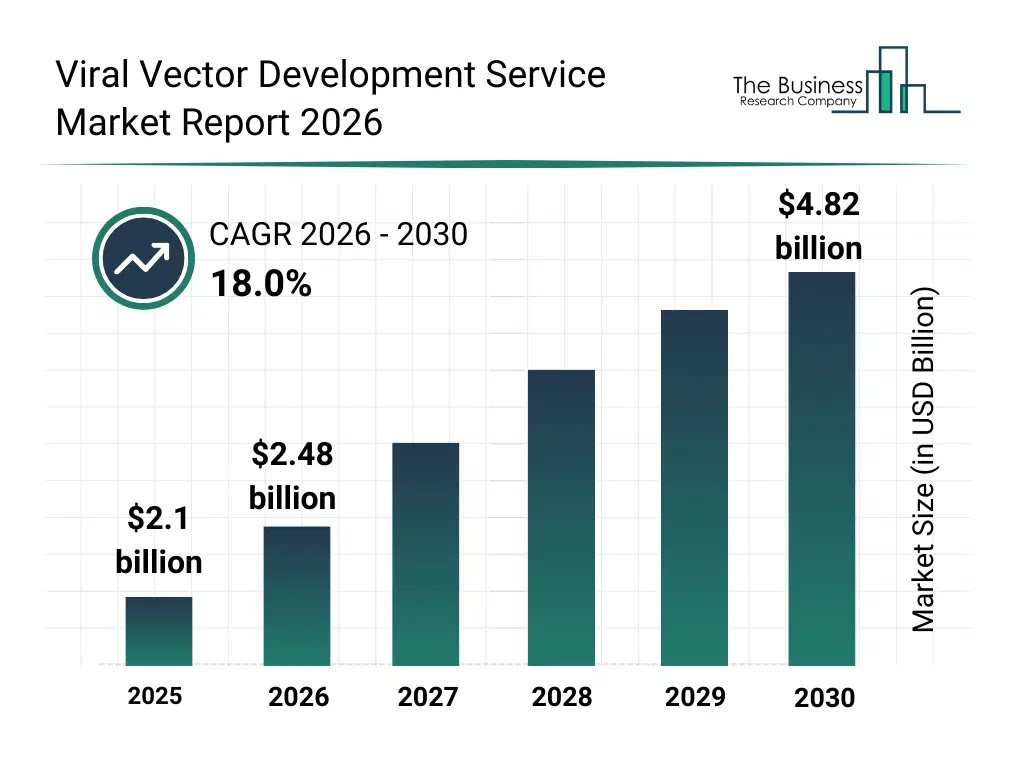
What Is The Viral Vector Development Service Market Size and Share 2026?
The viral vector development service market size has grown rapidly in recent years. It will grow from $2.1 billion in 2025 to $2.48 billion in 2026 at a compound annual growth rate (CAGR) of 18.3%. The growth in the historic period can be attributed to increasing complexity of gene therapy development, rising reliance on specialized service providers, expansion of clinical research programs, growth in biopharmaceutical r&d spending, increasing regulatory scrutiny on vector quality.What Is The Viral Vector Development Service Market Growth Forecast?
The viral vector development service market size is expected to see rapid growth in the next few years. It will grow to $4.82 billion in 2030 at a compound annual growth rate (CAGR) of 18.0%. The growth in the forecast period can be attributed to increasing commercialization of gene and cell therapies, rising demand for rapid development timelines, expansion of cdmo partnerships, growing focus on scalable manufacturing readiness, increasing integration of digital bioprocess tools. Major trends in the forecast period include increasing outsourcing of viral vector development activities, rising demand for scalable process optimization services, growing focus on regulatory-compliant vector design, expansion of end-to-end development platforms, enhanced use of data-driven process optimization.Global Viral Vector Development Service Market Segmentation
1) By Vector Type: Adenoviral Vectors, Lentiviral Vectors, Retroviral Vectors, Adeno-Associated Viral Vectors, Other Vector types 2) By Distribution Channel: Online Sales, Direct Sales, Distributors, Retailers 3) By Application: Gene Therapy, Vaccinology, Cancer Therapy, Other Applications 4) By End-User: Pharmaceutical Companies, Academic And Research Institutions Subsegments: 1) By Adenoviral Vectors: First Generation, Second Generation, Third Generation, Helper Dependent, Chimeric 2) By Lentiviral Vectors: Human Immunodeficiency Virus Based, Simian Immunodeficiency Virus Based, Non Integrating, Self Inactivating, Chimeric 3) By Retroviral Vectors: Gammaretroviral, Alpharetroviral, Spumaretroviral, Moloney Murine Leukemia Virus Based, Avian Leukosis Virus Based 4) By Adeno Associated Viral Vectors: Serotype One, Serotype Two, Serotype Five, Serotype Eight, Other Serotypes 5) By Other Vector Types: Herpes Simplex Virus Based, Baculovirus Based, Foamy Virus Based, Vaccinia Virus Based, Hybrid VectorsWhat Is The Driver Of The Viral Vector Development Service Market?
The surge in demand for gene therapies is expected to propel the growth of the viral vector development service market going forward. Gene therapies are medical treatments that involve introducing, removing, or altering genetic material within a patient’s cells to treat or prevent diseases. The demand for gene therapies is increasing due to advancements in delivery systems, as they enable safe, efficient, and targeted transport of genetic material into cells. Viral vector development services support gene therapies by providing end-to-end solutions for designing, producing, and optimizing viral vectors, making them essential for advanced therapeutic development. They improve treatment efficiency by enabling targeted and safe gene delivery, accelerating research and clinical applications. For instance, in December 2024, according to the American Society of Gene & Cell Therapy (ASGCT), a US-based non-profit medical and scientific organization, the Food and Drug Administration (FDA) approved six gene therapy products in 2023, up from five approvals in 2022. Therefore, the surge in demand for gene therapies is driving the growth of the viral vector development service industry.Key Players In The Global Viral Vector Development Service Market
Major companies operating in the viral vector development service market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec Co. Ltd., Catalent Inc., Charles River Laboratories International Inc., FUJIFILM Diosynth Biotechnologies Ltd., AGC Biologics Inc., GenScript Biotech Corporation, Takara Bio Inc., Oxford Biomedica plc, Cell and Gene Therapy Catapult Ltd., BioNTech IMFS GmbH, FinVector Oy, Aldevron LLC, ViroCell Biologics Ltd., Cobra Biologics Ltd., SIRION Biotech GmbH, Vigene Biosciences Inc., Creative Biogene Co. Ltd.Global Viral Vector Development Service Market Trends and Insights
Major companies operating in the viral vector development service market are focusing on developing advancements in biotechnological research, such as next-generation viral vector technology platforms, to enhance gene delivery efficiency. Next-generation viral vector technology platforms are advanced systems for safer, more efficient, and targeted delivery of genes using viral vectors, with improved production scalability and reduced immune response. For instance, in November 2023, SK Pharmteco Co. Ltd., a US-based contract development and manufacturing organization (CDMO), launched new viral vector technology platforms LentiSure and AAVelocity to strengthen its position in the rapidly expanding cell and gene therapy field. LentiSure is designed to optimize lentiviral vector production by improving transfection efficiency, scalability, and batch consistency, while AAVelocity enhances adeno-associated viral (AAV) vector yields through advanced process control and purification methods. These platforms enable faster, more reliable, and cost-efficient viral vector manufacturing for next-generation therapeutic development.What Are Latest Mergers And Acquisitions In The Viral Vector Development Service Market?
In November 2023, Ajinomoto Co. Inc., a Japan-based food and biotechnology company, acquired Forge Biologics Holdings LLC for $620 million. Through this acquisition, Ajinomoto Co. Inc. aims to expand its capabilities in viral vector development and manufacturing, strengthening its Bio-Pharma Services business to support the production of life-changing gene therapies. Forge Biologics Holdings LLC is a US-based gene therapy contract development and manufacturing organization (CDMO) that focuses on the end-to-end production of viral vectors for advanced therapeutic applications.Regional Outlook
North America was the largest region in the viral vector development service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Viral Vector Development Service Market?
The viral vector development service market includes revenues earned by entities through viral vector process development, viral vector analytical testing, viral vector scale-up and optimization, viral vector quality control and validation, and viral vector fill-finish and packaging services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Viral Vector Development Service Market Report 2026?
The viral vector development service market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the viral vector development service industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Viral Vector Development Service Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.48 billion |
| Revenue Forecast In 2035 | $4.82 billion |
| Growth Rate | CAGR of 18.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Vector Type, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec Co. Ltd., Catalent Inc., Charles River Laboratories International Inc., FUJIFILM Diosynth Biotechnologies Ltd., AGC Biologics Inc., GenScript Biotech Corporation, Takara Bio Inc., Oxford Biomedica plc, Cell and Gene Therapy Catapult Ltd., BioNTech IMFS GmbH, FinVector Oy, Aldevron LLC, ViroCell Biologics Ltd., Cobra Biologics Ltd., SIRION Biotech GmbH, Vigene Biosciences Inc., Creative Biogene Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
