
Pharma ADMET Testing Market Report 2026
Global Outlook – By Testing Type (In Vivo ADMET Testing, In Vitro ADMET Testing, In Silico ADMET Testing), By Technology (Cell Culture, High Throughput, Molecular Imaging, OMICS Technology), By Application (Systemic Toxicity, Renal Toxicity, Hepatotoxicity, Neurotoxicity, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Pharma ADMET Testing Market Overview
• Pharma ADMET Testing market size has reached to $10.7 billion in 2025 • Expected to grow to $19.27 billion in 2030 at a compound annual growth rate (CAGR) of 12.6% • Growth Driver: The Impact Of Escalating Clinical Trials On The Pharma ADMET Testing Market • Market Trend: Artificial Intelligence Driven Drug Design And ADMET Prediction Redefining Pharma ADMET Testing With In Silico Platforms • Asia-Pacific was the largest region in 2025.What Is Covered Under Pharma ADMET Testing Market?
Pharma ADMET testing refers to a set of procedures and assays used in the pharmaceutical industry to assess the Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) of drug candidates. It is used for determining the safety and efficacy of potential drugs before they can be approved for clinical use. The main types of pharma ADMET testing are in vivo ADMET testing, in vitro ADMET testing, and in silico ADMET testing. In vivo ADMET testing refers to the assessment of a drug's absorption, distribution, metabolism, excretion, and toxicity properties within a living organism. Pharma ADMET testing is used in drug discovery and development processes to identify potential drug candidates with desirable pharmacokinetic properties. The technologies include cell culture, high throughput, molecular imaging, and OMICS technology which are used in systemic toxicity, renal toxicity, hepatotoxicity, neurotoxicity, and others.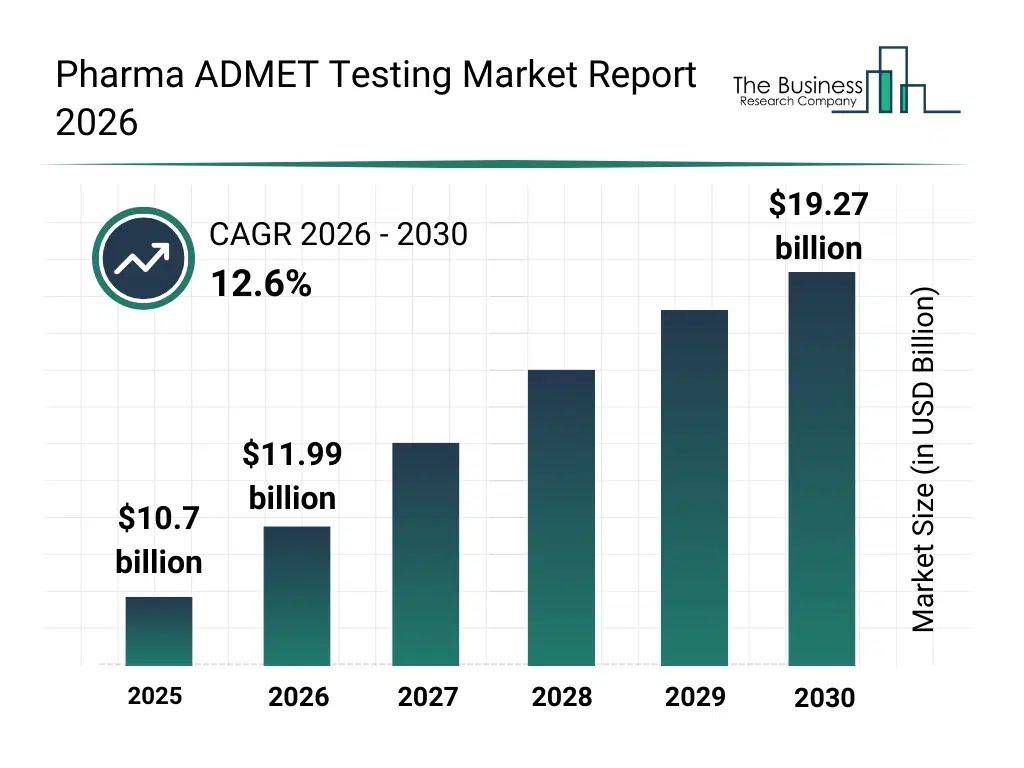
What Is The Pharma ADMET Testing Market Size and Share 2026?
The pharma admet testing market size has grown rapidly in recent years. It will grow from $10.7 billion in 2025 to $11.99 billion in 2026 at a compound annual growth rate (CAGR) of 12.1%. The growth in the historic period can be attributed to increasing pharmaceutical r&d investments, expansion of preclinical drug testing requirements, rising complexity of drug molecules, growing regulatory emphasis on safety profiling, availability of advanced laboratory technologies.What Is The Pharma ADMET Testing Market Growth Forecast?
The pharma admet testing market size is expected to see rapid growth in the next few years. It will grow to $19.27 billion in 2030 at a compound annual growth rate (CAGR) of 12.6%. The growth in the forecast period can be attributed to increasing demand for cost-efficient drug development, rising adoption of predictive admet analytics, expansion of personalized medicine pipelines, growing integration of machine learning in drug testing, increasing outsourcing of admet testing services. Major trends in the forecast period include increasing adoption of in silico admet modeling tools, growing use of high-throughput screening technologies, expansion of AI-based toxicity prediction platforms, rising integration of omics data in drug testing, enhanced focus on early-stage drug safety evaluation.Global Pharma ADMET Testing Market Segmentation
1) By Testing Type: In Vivo ADMET Testing, In Vitro ADMET Testing, In Silico ADMET Testing 2) By Technology: Cell Culture, High Throughput, Molecular Imaging, OMICS Technology 3) By Application: Systemic Toxicity, Renal Toxicity, Hepatotoxicity, Neurotoxicity, Other Applications Subsegments: 1) By In Vivo ADMET Testing: Animal Studies, Pharmacokinetics Studies, Toxicology Studies, Biodistribution Studies 2) By In Vitro ADMET Testing: Metabolism Studies, Drug-Drug Interaction Studies, Absorption Studies, Cytotoxicity And Safety Testing 3) By In Silico ADMET Testing: Predictive Modeling and Simulation, Quantitative Structure-Activity Relationship (QSAR) Analysis, Machine Learning Algorithms For ADMET Predictions, Software Tools For ADMET ProfilingWhat Is The Driver Of The Pharma ADMET Testing Market?
The increase in the number of clinical trials is expected to boost the growth of the pharma ADMET testing market going forward. A clinical trial is a research study conducted with human participants to evaluate the safety, efficacy, and potential side effects of a medical intervention, such as a new drug, medical device, diagnostic procedure, or treatment regimen. The upswing in clinical trials markedly elevates the demand for thorough assessments of drug safety and efficacy within the pharma ADMET testing market, emphasizing the critical need for tailored evaluations across diverse drug candidates and patient populations. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based nonprofit organization, the number of industry-initiated clinical trials in the UK grew from 411 trials in 2022 to 426 trials in 2023, marking a 3.7% increase. Therefore, the increase in the number of clinical trials is driving the growth of the pharma ADMET testing industry.Key Players In The Global Pharma ADMET Testing Market
Major companies operating in the pharma admet testing market are Thermo Fisher Scientific Inc., The Merck Group, Laboratory Corporation of America Holdings, IQVIA Inc., PPD ICON plc, Agilent Technologies Inc., Wuxi AppTec Co. Ltd., Catalent Inc., Covance Inc., Charles River Laboratories International Inc., Eurofins Scientific SE, Beckman Coulter Inc., Perkinelmer Inc., Albany Molecular Research Inc., Evotec A.G., CMIC Holdings Co. Ltd., Promega Corporation, Aptuit LLC, Symeres B.V., Biovia Corp., BioIVT LLC, Cyprotex Limited, InSphero AG, Admescope Ltd., Accelrys Inc.Global Pharma ADMET Testing Market Trends and Insights
Major companies in the pharma ADMET testing market are introducing innovative products such as artificial intelligence-driven drug design, which allows for automated de novo generation of novel small molecule structures. Artificial intelligence-driven drug design refers to the use of artificial intelligence (AI) to design and optimize drug molecules without relying solely on traditional lab-based screening or manual medicinal chemistry. For instance, in June 2025, Simulations Plus Inc., a US-based company specializing in cheminformatics software for the pharmaceutical sector, launched ADMET Predictor 13, an advanced artificial intelligence-driven drug-design and ADMET-prediction platform integrating enhanced high-throughput PBPK simulations and a generative AI-enabled drug-design engine (AIDD). Additionally, it offers streamlined workflows enabling faster and more informed decision-making in drug development.What Are Latest Mergers And Acquisitions In The Pharma ADMET Testing Market?
In November 2024, Concept Life Sciences Limited, a UK-based integrated drug discovery and development company specializing in medicinal chemistry, acquired Charnwood Discovery Ltd for an undisclosed amount. Through this acquisition, Concept Life Sciences aims to enhance its drug discovery and development capabilities by integrating Charnwood’s expertise in medicinal chemistry and ADME/ADMET testing services, thereby streamlining early-stage drug development and offering end-to-end solutions to a broader pharmaceutical and biotech customer base. Charnwood Discovery is a UK-based contract research organization specializing in medicinal ADMET testing services.Regional Outlook
Asia-Pacific was the largest region in the pharma ADMET testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharma ADMET Testing Market?
The pharma ADMET testing market consists of revenues earned by entities by providing services such as in vitro screening, in vitro metabolism, and metabolite testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharma ADMET testing market also includes the sales of membranes, buffer systems, and analytes that are used in the assays. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharma ADMET Testing Market Report 2026?
The pharma admet testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharma admet testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharma ADMET Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.99 billion |
| Revenue Forecast In 2035 | $19.27 billion |
| Growth Rate | CAGR of 12.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Testing Type, Technology, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., The Merck Group, Laboratory Corporation of America Holdings, IQVIA Inc., PPD ICON plc, Agilent Technologies Inc., Wuxi AppTec Co. Ltd., Catalent Inc., Covance Inc., Charles River Laboratories International Inc., Eurofins Scientific SE, Beckman Coulter Inc., Perkinelmer Inc., Albany Molecular Research Inc., Evotec A.G., CMIC Holdings Co. Ltd., Promega Corporation, Aptuit LLC, Symeres B.V., Biovia Corp., BioIVT LLC, Cyprotex Limited, InSphero AG, Admescope Ltd., Accelrys Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
