Antibody Validation Market Report 2026

Antibody Validation Market Report 2026
Global Outlook – By Product Type (Monoclonal Antibodies, Polyclonal Antibodies, Anti-Body Drug Complexes), By Disease Indication (Cardiovascular Diseases, Central Nervous System (CNS) Disorders, Cancer, Autoimmune Disorders), By End User (Pharmaceutical And Biotechnological Firms, Contract Research Organizations, Research Institutes) - Market Size, Trends, And Global Forecast 2026-2035
Antibody Validation Market Overview
• Antibody Validation market size has reached to $487.5 billion in 2025 • Expected to grow to $1060.68 billion in 2030 at a compound annual growth rate (CAGR) of 16.8% • Growth Driver: Rising Prevalence Of Chronic Diseases Fueling The Growth Of The Market Due To Increasing Need For Accurate And Reliable Diagnostic And Therapeutic Tools • Market Trend: Advancements In Validated Antibody Technologies For Rare Cell And Tumor Analysis • North America was the largest region in 2025.What Is Covered Under Antibody Validation Market?
Antibody validation is the process of confirming the specificity, sensitivity, and reproducibility of antibodies for their intended biological applications. It ensures that antibodies accurately bind to their target molecules without cross-reactivity or non-specific interactions. This process is critical for generating reliable and consistent experimental results in research and diagnostics. The main product types of the antibody validation are monoclonal antibodies, polyclonal antibodies, and antibody-drug complexes. Monoclonal antibodies refer to laboratory-engineered antibodies that bind to a single specific epitope, providing high specificity and reproducibility, and are widely used for research, diagnostic, and therapeutic purposes. It is are widely used for disease indications such as cardiovascular diseascentral nervous system (CNS) CNS disorders, cancer, and autoimmune disorders, serving end-users such asT pharmaceutical and biotechnological firms, contract research organizations, and research institutes.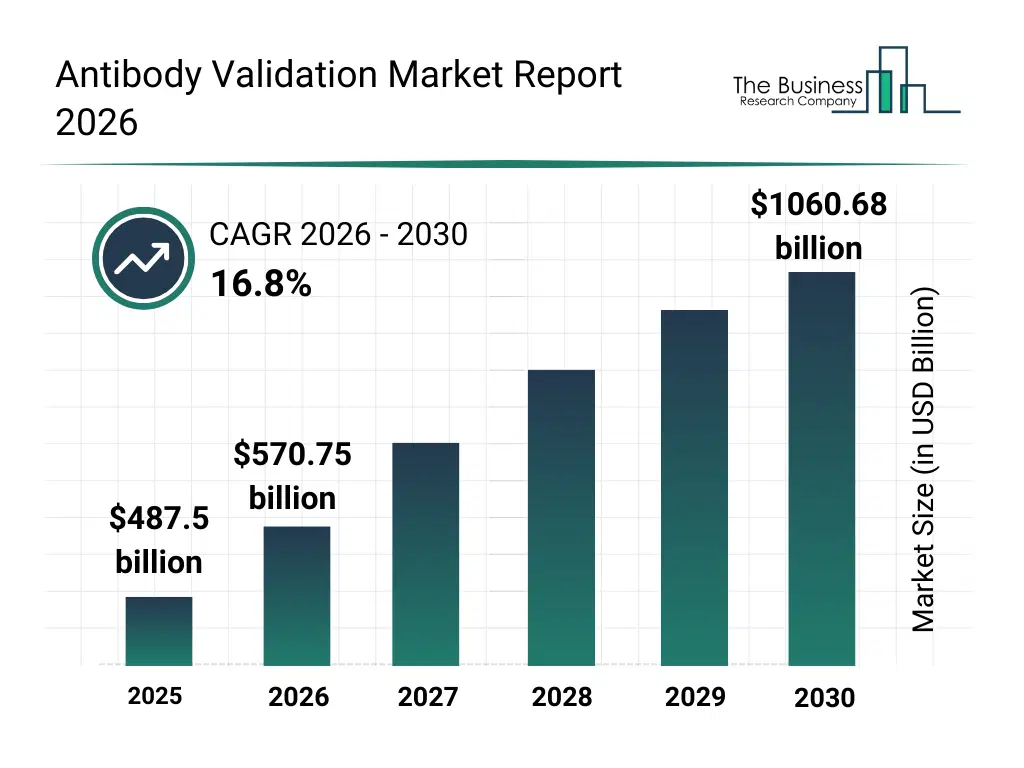
What Is The Antibody Validation Market Size and Share 2026?
The antibody validation market size has grown rapidly in recent years. It will grow from $487.5 billion in 2025 to $570.75 billion in 2026 at a compound annual growth rate (CAGR) of 17.1%. The growth in the historic period can be attributed to increasing variability concerns in research antibodies, expansion of proteomics and genomics research, rising regulatory scrutiny on research reproducibility, growth in antibody-based assays, increasing funding for life sciences research.What Is The Antibody Validation Market Growth Forecast?
The antibody validation market size is expected to see rapid growth in the next few years. It will grow to $1060.68 billion in 2030 at a compound annual growth rate (CAGR) of 16.8%. The growth in the forecast period can be attributed to increasing demand for rigor and reproducibility, rising use of antibodies in clinical research, expansion of precision diagnostics, growing adoption of validated antibodies in drug discovery, increasing integration of digital validation data platforms. Major trends in the forecast period include increasing adoption of standardized antibody validation protocols, rising demand for application-specific validation services, growing use of multiplex validation techniques, expansion of reproducibility-focused testing frameworks, enhanced emphasis on data transparency.Global Antibody Validation Market Segmentation
1) By Product Type: Monoclonal Antibodies, Polyclonal Antibodies, Anti-Body Drug Complexes 2) By Disease Indication: Cardiovascular Diseases, Central Nervous System (CNS) Disorders, Cancer, Autoimmune Disorders 3) By End User: Pharmaceutical And Biotechnological Firms, Contract Research Organizations, Research Institutes Subsegments: 1) By Monoclonal Antibodies: Recombinant Monoclonal Antibodies, Humanized Monoclonal Antibodies, Chimeric Monoclonal Antibodies, Murine Monoclonal Antibodies 2) By Polyclonal Antibodies: Rabbit Polyclonal Antibodies, Goat Polyclonal Antibodies, Sheep Polyclonal Antibodies, Donkey Polyclonal Antibodies 3) By Antibody Drug Complexes: Immunoconjugates, Radioimmunoconjugates, Immunotoxins, Bispecific Antibody Drug ComplexesWhat Is The Driver Of The Antibody Validation Market?
The rising prevalence of chronic diseases is expected to propel the growth of the antibody validation market going forward. Chronic diseases are persistent health conditions that develop slowly and last for an extended period, often requiring continuous medical care. The growing prevalence of chronic diseases is partly due to inactive lifestyles, as prolonged sitting and minimal physical activity raise the risk of diseases such as heart conditions and diabetes. Antibody validation aids in chronic disease management by ensuring the accuracy, specificity, and reliability of antibodies used in diagnostics and therapeutics. It enhances the detection of disease biomarkers, supports precise monitoring, and improves the development of targeted treatments, ultimately advancing effective and personalized chronic disease care. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes (a condition with elevated blood sugar levels, not high enough to be classified as diabetes) in 2023, marking an 18% increase from 3,065,825 cases in 2022. Therefore, the rising prevalence of chronic diseases is driving the growth of the antibody validation industry.Key Players In The Global Antibody Validation Market
Major companies operating in the antibody validation market are F. Hoffmann-La Roche Ltd., Merck & Co. Inc., Thermo Fisher Scientific Inc., Danaher Corporation, Becton Dickinson and Company, Agilent Technologies Inc., PerkinElmer Inc., Bio-Rad Laboratories Inc., Azenta Inc., GenScript Biotech Corporation, Promega Corporation, Sino Biological Inc., Cell Signaling Technology Inc., Fortis Life Sciences, Proteintech Group Inc., Santa Cruz Biotechnology Inc., Novus Biologicals LLC, Creative Diagnostics, Rockland Immunochemicals Inc., Biorbyt Ltd.Global Antibody Validation Market Trends and Insights
Major companies operating in the antibody validation market are focusing on advanced innovations, such as Celselect slides enumeration stain kits, to enhance the accuracy and sensitivity of rare cell and circulating tumor cell identification, improve validation efficiency, and support high-precision cancer research and diagnostic applications. Celselect slides enumeration stain kits refer to specialized reagent kits designed to enable the detection, staining, and enumeration of rare cells and circulating tumor cells (CTCs). For instance, in March 2024, Bio-Rad Laboratories Inc., a US-based life science research company, launched the celselect slides validated antibodies for rare cell and circulating tumor cell (CTC) enumeration to enable sensitive and specific identification of CTC surface markers, enhance tumor heterogeneity studies, and support high-precision cancer research through improved rare cell capture, enrichment, and analysis. This launch expanded the company’s single-cell oncology portfolio by providing researchers with validated reagents for accurate immunostaining and enumeration of CTCs.What Are Latest Mergers And Acquisitions In The Antibody Validation Market?
In September 2024, Leinco Technologies Inc., a US-based biotechnology company, acquired QED Biosciences Inc. for an undisclosed amount. With this acquisition, Leinco Technologies aims to expand its antibody development capabilities, strengthen its presence in the research and in vitro diagnostics sectors, and enhance its global portfolio of high-quality antibodies, reagents, and recombinant proteins to better support advanced discovery and diagnostic applications. QED Biosciences Inc. is a US-based company specializing in antibody validation, development, and production of high-quality antibodies.Regional Outlook
North America was the largest region in the antibody validation market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Antibody Validation Market?
The antibody validation market includes revenues earned by entities through flow cytometry validation, enzyme-linked immunosorbent assay validation, immunoprecipitation validation, and protein array validation. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Antibody Validation Market Report 2026?
The antibody validation market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the antibody validation industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Antibody Validation Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $570.75 billion |
| Revenue Forecast In 2035 | $1060.68 billion |
| Growth Rate | CAGR of 17.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Disease Indication, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Merck & Co. Inc., Thermo Fisher Scientific Inc., Danaher Corporation, Becton Dickinson and Company, Agilent Technologies Inc., PerkinElmer Inc., Bio-Rad Laboratories Inc., Azenta Inc., GenScript Biotech Corporation, Promega Corporation, Sino Biological Inc., Cell Signaling Technology Inc., Fortis Life Sciences, Proteintech Group Inc., Santa Cruz Biotechnology Inc., Novus Biologicals LLC, Creative Diagnostics, Rockland Immunochemicals Inc., Biorbyt Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Antibody Validation market was valued at $487.5 billion in 2025, increased to $570.75 billion in 2026, and is projected to reach $1060.68 billion by 2030.
The global Antibody Validation market is expected to grow at a CAGR of 16.8% from 2026 to 2035 to reach $1060.68 billion by 2035.
Some Key Players in the Antibody Validation market Include, F. Hoffmann-La Roche Ltd., Merck & Co. Inc., Thermo Fisher Scientific Inc., Danaher Corporation, Becton Dickinson and Company, Agilent Technologies Inc., PerkinElmer Inc., Bio-Rad Laboratories Inc., Azenta Inc., GenScript Biotech Corporation, Promega Corporation, Sino Biological Inc., Cell Signaling Technology Inc., Fortis Life Sciences, Proteintech Group Inc., Santa Cruz Biotechnology Inc., Novus Biologicals LLC, Creative Diagnostics, Rockland Immunochemicals Inc., Biorbyt Ltd. .
Major trend in this market includes: Advancements In Validated Antibody Technologies For Rare Cell And Tumor Analysis. For further insights on this market.
Request for SampleNorth America was the largest region in the antibody validation market in 2025. The regions covered in the antibody validation market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
