
Hepatitis C Testing Market Report 2026
Global Outlook – By Test Type (Antibody Tests, Nucleic Acid Tests, Genotyping Tests), By Technology (Enzyme-Linked Immunosorbent Assay (ELISA), Rapid Diagnostic Tests, Polymerase Chain Reaction, Next-Generation Sequencing), By Application (Blood Screening, Viral Load Monitoring, Disease Diagnosis), By End-User (Hospital-Based Laboratories, Stand-Alone Laboratories, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Hepatitis C Testing Market Overview
• Hepatitis C Testing market size has reached to $1.32 billion in 2025 • Expected to grow to $1.78 billion in 2030 at a compound annual growth rate (CAGR) of 6.1% • Growth Driver: Rise In Hepatitis Cases Prevalence Fueling the Growth Of The Market Due To Increasing Global Disease Burden And Demand For Early Diagnosis • Market Trend: Innovative Dual Antigen-Antibody Diagnostic Tests Enhance Early And Comprehensive Hepatitis C Screening • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hepatitis C Testing Market?
Hepatitis C testing involves diagnostic methods to identify HCV infection or the body’s immune response to it. It helps detect both current and past infections, allowing timely treatment. These tests are crucial for high-risk individuals and support monitoring treatment outcomes. Early diagnosis reduces the risk of severe liver complications such as cirrhosis and liver cancer. The main types of hepatitis C testing are antibody tests, nucleic acid tests, and genotyping tests. Antibody tests are diagnostic tests that detect the presence of antibodies in the blood, indicating a past or ongoing immune response to an infection such as hepatitis C. The products include test kits, instruments, reagents, and consumables with various test technologies such as enzyme-linked immunosorbent assay (ELISA), rapid diagnostic tests, polymerase chain reaction (PCR), and next-generation sequencing (NGS). It is applicable in several applications, including blood screening, viral load monitoring, and disease diagnosis, and they are used by various end-users such as hospital-based laboratories, stand-alone laboratories, and others.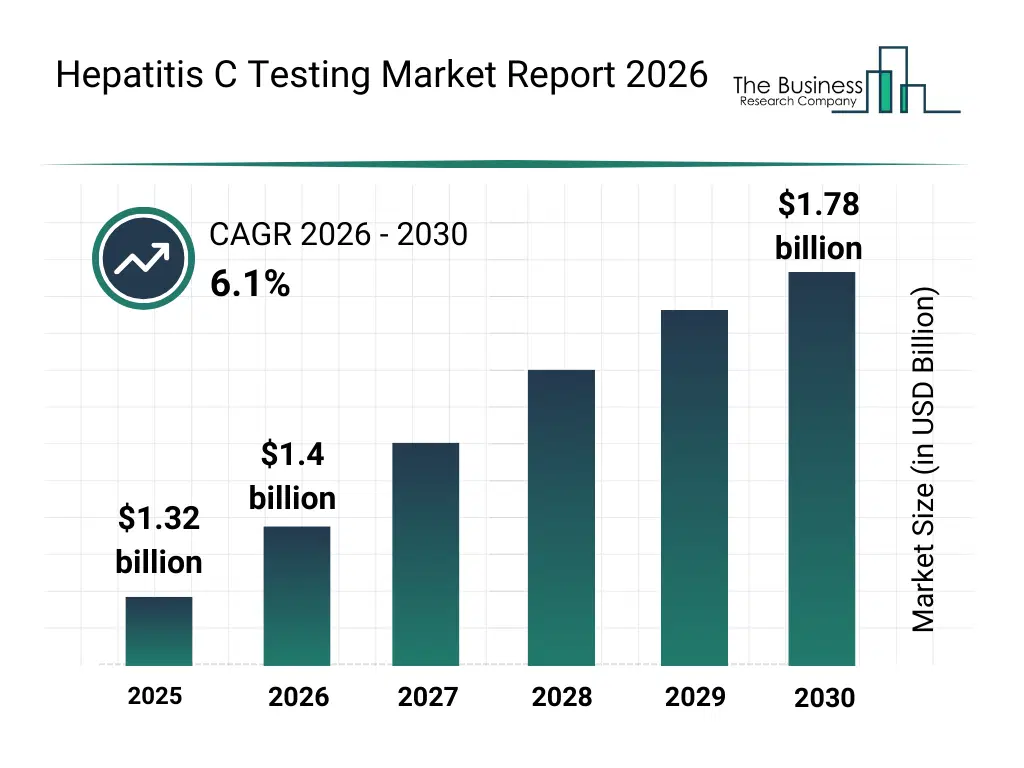
What Is The Hepatitis C Testing Market Size and Share 2026?
The hepatitis c testing market size has grown strongly in recent years. It will grow from $1.32 billion in 2025 to $1.4 billion in 2026 at a compound annual growth rate (CAGR) of 6.4%. The growth in the historic period can be attributed to expansion of blood screening programs, rising prevalence of hepatitis c infections, improved diagnostic awareness, availability of advanced testing kits, growth in public health surveillance initiatives.What Is The Hepatitis C Testing Market Growth Forecast?
The hepatitis c testing market size is expected to see strong growth in the next few years. It will grow to $1.78 billion in 2030 at a compound annual growth rate (CAGR) of 6.1%. The growth in the forecast period can be attributed to increasing global elimination initiatives, rising demand for point-of-care diagnostics, expansion of molecular testing access, growing investments in infectious disease diagnostics, increasing focus on early intervention. Major trends in the forecast period include increasing adoption of rapid diagnostic tests, rising use of molecular testing technologies, growing focus on early disease screening, expansion of high-throughput laboratory testing, enhanced monitoring of treatment outcomes.Global Hepatitis C Testing Market Segmentation
1) By Test Type: Antibody Tests, Nucleic Acid Tests, Genotyping Tests 2) By Technology: Enzyme-Linked Immunosorbent Assay (ELISA), Rapid Diagnostic Tests, Polymerase Chain Reaction, Next-Generation Sequencing 3) By Application: Blood Screening, Viral Load Monitoring, Disease Diagnosis 4) By End-User: Hospital-Based Laboratories, Stand-Alone Laboratories, Other End-Users Subsegments: 1) By Antibody Tests: Enzyme Immunoassay (EIA), Recombinant Immunoblot Assay (RIBA), Rapid Diagnostic Tests (RDTs) 2) By Nucleic Acid Tests (NATs): Quantitative Ribonucleic Acid (RNA) Tests, Qualitative Ribonucleic Acid (RNA) Tests, Transcription-Mediated Amplification (TMA), Reverse Transcription Polymerase Chain Reaction (RT-PCR) 3) By Genotyping Tests: Line Probe Assay (LiPA), Sequencing-Based Assay, Real-Time Polymerase Chain Reaction (PCR)-Based GenotypingWhat Is The Driver Of The Hepatitis C Testing Market?
The rise in the prevalence of hepatitis cases is expected to propel the growth of the hepatitis C testing market going forward. Hepatitis C is a viral infection that causes liver inflammation and can lead to serious liver damage if left untreated. Hepatitis cases are increasing mainly because low vaccination rates leave many individuals unprotected, facilitating the virus’s spread. Hepatitis C testing allows for early virus detection, facilitating prompt treatment to avoid liver damage and serious complications. It also identifies infected individuals to help prevent the spread and enhance disease control. For instance, in April 2024, according to the World Health Organization (WHO), a Switzerland-based intergovernmental organization responsible for global public health, over 50 million people are living with chronic hepatitis C virus infection globally, with approximately 1.0 million new infections occurring each year. Therefore, the rising prevalence of hepatitis cases is driving the growth of the hepatitis C testing industry.Key Players In The Global Hepatitis C Testing Market
Major companies operating in the hepatitis c testing market are F. Hoffmann-La Roche Ltd., Bayer AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Quest Diagnostics Incorporated, Hologic Inc., QuidelOrtho Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., SD Biosensor INC., QIAGEN N.V., DiaSorin S.p.A., Cepheid, OraSure Technologies Inc., Meridian Bioscience Inc., Trivitron Healthcare Private Limited, General Biologicals Corporation, Biogate Laboratories Ltd., Cosara Diagnostics Private Limited, BioGenex Life Sciences Private LimitedGlobal Hepatitis C Testing Market Trends and Insights
Major companies operating in the hepatitis C testing market are focused on developing advanced solutions such as dual antigen-antibody diagnostic tests to enable more effective screening and monitoring of the infection. An antigen-antibody diagnostic test detects viral proteins (antigens) and immune response proteins (antibodies) to identify both active and past infections. This dual approach offers a thorough evaluation of the infection status. For instance, in July 2023, Roche Diagnostics India Pvt. Ltd., an India-based provider of diagnostic tests and systems, launched Elecsys HCV Duo, a fully automated immunoassay capable of simultaneously and independently detecting both HCV antigens and antibodies from one plasma or serum sample. This innovation enables early diagnosis, supports patient management, and aligns with India’s national strategy to eliminate hepatitis C through timely and comprehensive screening.What Are Latest Mergers And Acquisitions In The Hepatitis C Testing Market?
In October 2024, QIAGEN N.V., a Netherlands-based molecular diagnostics and sample technologies company, partnered with Bio-Manguinhos/Fiocruz, a Brazil-based public health institution, to enhance malaria, dengue, and hepatitis C detection in Brazil’s national screening programs. With this partnership, QIAGEN N.V. and Bio-Manguinhos/Fiocruz aim to improve the sensitivity, efficiency, and scalability of diagnostic testing for these diseases, supporting timely and accurate disease monitoring nationwide. Bio-Manguinhos/Fiocruz is a Brazil-based vaccine and diagnostics manufacturing institute that provides vaccine, diagnostic, and biopharmaceutical development.Regional Outlook
North America was the largest region in the hepatitis C testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Hepatitis C Testing Market?
The hepatitis C testing market consists of revenues earned by entities by providing services such as laboratory-based HCV RNA testing, rapid diagnostic tests, and home-based HCV test kits. The market value includes the value of related goods sold by the service provider or included within the service offering. The hepatitis C testing market also includes sales of quality control materials, sample collection devices, and point-of-care (POC) diagnostic devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Hepatitis C Testing Market Report 2026?
The hepatitis c testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the hepatitis c testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Hepatitis C Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.4 billion |
| Revenue Forecast In 2035 | $1.78 billion |
| Growth Rate | CAGR of 6.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Technology, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Bayer AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Quest Diagnostics Incorporated, Hologic Inc., QuidelOrtho Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., SD Biosensor INC., QIAGEN N.V., DiaSorin S.p.A., Cepheid, OraSure Technologies Inc., Meridian Bioscience Inc., Trivitron Healthcare Private Limited, General Biologicals Corporation, Biogate Laboratories Ltd., Cosara Diagnostics Private Limited, BioGenex Life Sciences Private Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
