
In-vitro Colorectal Cancer Screening Tests Market Report 2026
Global Outlook – By Test Type (Fecal Occult Blood Test, Stool Deoxyribonucleic Acid Test, Blood Biomarker Test, Other Test Types), By Imaging Type (In-vitro Confirmatory Diagnostics, Laboratory-Based Molecular Testing, Companion Diagnostic Testing, Genetic And Epigenetic Testing, Biomarker Validation Testing, Risk Stratification Assays), By Application (Screening, Diagnosis, Monitoring, Risk Assessment, Post-Treatment Surveillance), By End User (Hospitals, Diagnostic Laboratories, Outpatient Clinics, Home Care Settings, Research Institutions) - Market Size, Trends, And Global Forecast 2026-2035
In-vitro Colorectal Cancer Screening Tests Market Overview
• In-vitro Colorectal Cancer Screening Tests market size has reached to $1.3 billion in 2025 • Expected to grow to $1.82 billion in 2030 at a compound annual growth rate (CAGR) of 6.8% • Growth Driver: Rising Prevalence Of Colorectal Cancer Fueling The Growth Of The Market Due To Increasing Risk From Unhealthy Lifestyles • Market Trend: Innovative Non-Invasive Test Enhances Patient Screening Experience • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In-vitro Colorectal Cancer Screening Tests Market?
In-vitro colorectal cancer screening tests are non-invasive laboratory-based tests that analyze stool or blood samples to detect signs of colorectal cancer or precancerous conditions. These tests help detect early signs of colorectal cancer in asymptomatic individuals, enabling early intervention and reducing cancer-related mortality. The main test types of in-vitro colorectal cancer screening tests are fecal occult blood test, stool deoxyribonucleic acid test, blood biomarker test, and others. The fecal occult blood test refers to a noninvasive diagnostic method used to detect hidden (occult) blood in the stool, which may be an early sign of colorectal cancer or other gastrointestinal disorders. The various imaging techniques include colonoscopy, proctoscopy, computed tomography scan, ultrasound, magnetic resonance imaging, and positron emission tomography scan, and support a wide range of applications, including screening, diagnosis, monitoring, risk assessment, and post-treatment surveillance, serving end users such as hospitals, diagnostic laboratories, outpatient clinics, home care settings, and research institutions.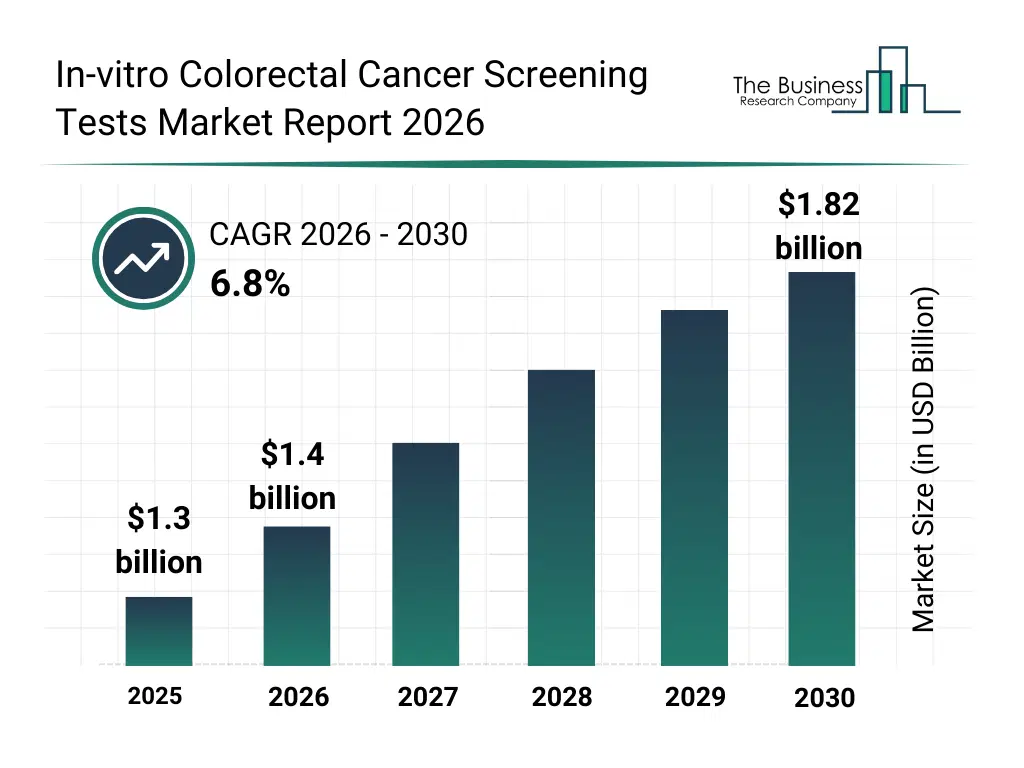
What Is The In-vitro Colorectal Cancer Screening Tests Market Size and Share 2026?
The in-vitro colorectal cancer screening tests market size has grown strongly in recent years. It will grow from $1.3 billion in 2025 to $1.4 billion in 2026 at a compound annual growth rate (CAGR) of 7.7%. The growth in the historic period can be attributed to increasing colorectal cancer incidence rates, expansion of population screening programs, growing awareness of early cancer detection, availability of laboratory-based diagnostic infrastructure, improvements in biomarker identification.What Is The In-vitro Colorectal Cancer Screening Tests Market Growth Forecast?
The in-vitro colorectal cancer screening tests market size is expected to see strong growth in the next few years. It will grow to $1.82 billion in 2030 at a compound annual growth rate (CAGR) of 6.8%. The growth in the forecast period can be attributed to increasing adoption of personalized screening approaches, rising demand for at-home diagnostic solutions, expansion of preventive healthcare initiatives, growing use of AI-assisted result interpretation, increasing focus on cost-effective cancer screening. Major trends in the forecast period include increasing adoption of non-invasive screening tests, rising use of multi-target stool DNA testing, growing integration of blood-based biomarker assays, expansion of home-based sample collection kits, enhanced focus on early detection in asymptomatic populations.Global In-vitro Colorectal Cancer Screening Tests Market Segmentation
1) By Test Type: Fecal Occult Blood Test, Stool Deoxyribonucleic Acid Test, Blood Biomarker Test, Other Test Types 2) By Imaging Type: In-vitro Confirmatory Diagnostics, Laboratory-Based Molecular Testing, Companion Diagnostic Testing, Genetic And Epigenetic Testing, Biomarker Validation Testing, Risk Stratification Assays 3) By Application: Screening, Diagnosis, Monitoring, Risk Assessment, Post-Treatment Surveillance 4) By End User: Hospitals, Diagnostic Laboratories, Outpatient Clinics, Home Care Settings, Research Institutions Subsegments: 1) By Fecal Occult Blood Test: Guaiac Based Fecal Occult Blood Test, Immunochemical Fecal Occult Blood Test 2) By Stool Deoxyribonucleic Acid Test: Single Target Stool Deoxyribonucleic Acid Test, Multi Target Stool Deoxyribonucleic Acid Test 3) By Blood Biomarker Test: Methylated Septin 9 Deoxyribonucleic Acid Test, Circulating Tumor Deoxyribonucleic Acid Test, Protein Biomarker Based Blood Test 4) By Other Test Types: Urine Based Biomarker Test, Serum Based Biomarker Test, Breath Based Volatile Organic Compound TestWhat Is The Driver Of The In-vitro Colorectal Cancer Screening Tests Market?
The rising prevalence of colorectal cancer is expected to propel the growth of the in vitro colorectal cancer screening tests market going forward. Colorectal cancer refers to a type of cancer that starts in the colon or rectum, often developing from precancerous polyps. The rising prevalence of colorectal cancer is due to unhealthy lifestyle habits, as poor diet, lack of exercise, and obesity increase the risk of polyp formation and cancer development. In-vitro colorectal cancer screening tests aid in colorectal cancer diagnosis by offering non-invasive methods to detect early signs of cancer. They improve screening accuracy through advanced biomarker detection, enhancing early intervention and patient outcomes. For instance, in February 2025, according to the American Cancer Society, a US-based nonprofit organization, the estimated number of new colorectal cancer cases was 154,270, an increase from 152,810 cases in 2024. Therefore, the rising prevalence of colorectal cancer is driving the growth of the in vitro colorectal cancer screening tests market.Key Players In The Global In-vitro Colorectal Cancer Screening Tests Market
Major companies operating in the in-vitro colorectal cancer screening tests market are Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers, Quest Diagnostics Incorporated, Roche Diagnostics International AG, Illumina Inc., Sysmex Corporation, Bio-Rad Laboratories Inc., Qiagen N.V., Exact Sciences Corporation, Natera Inc., BGI Genomics Co. Ltd., NeoGenomics Inc., Guardant Health Inc., CellMax Life Inc., Epigenomics AG, Acuamark Diagnostics Inc., Beacon Biomedical Inc., Mainz Biomed NV, Hologic Inc., Danaher Corporation, Agilent Technologies Inc.Global In-vitro Colorectal Cancer Screening Tests Market Trends and Insights
Major companies operating in the in vitro colorectal cancer screening tests market are focusing on developing advanced diagnostic solutions, such as multi-target stool DNA tests, to improve early detection accuracy, enhance patient convenience, and reduce colorectal cancer mortality rates. Multi-target stool DNA tests refer to innovative screening tools that analyze stool samples for genetic and biochemical markers associated with colorectal cancer and precancerous lesions. For instance, in March 2025, Exact Sciences Corp., a US-based molecular diagnostics company, launched Cologuard Plus, an enhanced at-home colorectal cancer screening test for average-risk adults aged 45 and older. This test incorporates advanced biomarker technology, offering higher sensitivity and specificity compared to its predecessor. It utilizes a refined algorithm to detect DNA mutations and hemoglobin biomarkers, improving the detection of both colorectal cancer and advanced adenomas. The Cologuard Plus test provides a non-invasive and convenient screening option that eliminates the need for bowel preparation or dietary restrictions, thereby increasing patient adherence to recommended screening guidelines.What Are Latest Mergers And Acquisitions In The In-vitro Colorectal Cancer Screening Tests Market?
In February 2025, Mainz Biomed N.V., a Germany-based biotech company, partnered with Labor Team W AG to jointly launch ColoAlert. This partnership aims to expand ColoAlert's availability across Switzerland, using Labor Team W AG’s diagnostic network to boost early colorectal cancer detection and improve patient outcomes. Labor Team W AG is a Switzerland-based diagnostic services company providing in-vitro colorectal cancer screening tests.Regional Outlook
North America was the largest region in the in-vitro colorectal cancer screening tests market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the In-vitro Colorectal Cancer Screening Tests Market?
The in-vitro colorectal cancer screening tests market consists of revenues earned by entities by providing services such as molecular diagnostic testing, lab sample collection and processing services, risk assessment and genetic counseling services, telehealth-based screening consultations, screening program management for institutions, and electronic health record (EHR) integration services. The market value includes the value of related goods sold by the service provider or included within the service offering. The in vitro colorectal cancer screening tests market also includes sales of sample collection devices, automated screening instruments, sample preservation and transport systems, and digital diagnostic platforms. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the In-vitro Colorectal Cancer Screening Tests Market Report 2026?
The in-vitro colorectal cancer screening tests market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the in-vitro colorectal cancer screening tests industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.In-vitro Colorectal Cancer Screening Tests Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.4 billion |
| Revenue Forecast In 2035 | $1.82 billion |
| Growth Rate | CAGR of 7.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Imaging Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers, Quest Diagnostics Incorporated, Roche Diagnostics International AG, Illumina Inc., Sysmex Corporation, Bio-Rad Laboratories Inc., Qiagen N.V., Exact Sciences Corporation, Natera Inc., BGI Genomics Co. Ltd., NeoGenomics Inc., Guardant Health Inc., CellMax Life Inc., Epigenomics AG, Acuamark Diagnostics Inc., Beacon Biomedical Inc., Mainz Biomed NV, Hologic Inc., Danaher Corporation, Agilent Technologies Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
