
Laboratory Proficiency Testing Market Report 2026
Global Outlook – By Technology (Cell Culture, Polymerase Chain Reaction (PCR), Immunoassays, Chromatography, Spectrophotometry, Other Technologies), By Test (Routine, Specialty), By Department (Parasitology, Virology, Hematology, Toxicology, Immunology/Serology, Histopathology, Urinalysis, Other Departments ), By End-Use (Hospitals, Contract Research Organizations, Pharmaceutical & Biotechnology Companies, Academic Research, Diagnostic Laboratories, Independent Laboratories, Specialty Laboratories) - Market Size, Trends, And Global Forecast 2026-2035
Laboratory Proficiency Testing Market Overview
• Laboratory Proficiency Testing market size has reached to $1.55 billion in 2025 • Expected to grow to $2.45 billion in 2030 at a compound annual growth rate (CAGR) of 9.6% • Growth Driver: Biosimilars Market Fuels Expansion Of Laboratory Proficiency Testing Services • Market Trend: Innovation In PT Management: Rise Of Web-Based PT-LIMS Solutions • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Laboratory Proficiency Testing Market?
Laboratory proficiency testing refers to a process by which the effectiveness of other labs for certain tests or metrics is evaluated and used to track lab performance over time. One or more artifacts are exchanged between participating labs during the laboratory proficiency test. Each laboratory makes measurements on the objects (such as a ring gauge or a set of gauge blocks) based on a preset set of criteria and communicates the results to the administrator. Each laboratory's results for a certain purpose are compared to the reference value for that purpose. The main technology in laboratory proficiency testing includes cell culture, PCR, immunoassays, chromatography, spectrophotometry, others that is used for specialty and routine tests. Cell Culture refers to the process of growing animal or plant cells in a sterile, well controlled setting. Cells can be taken directly from the organism and separated before cultivation, or they can come from a cell line or cell strain that has already been developed. The laboratory proficiency tests help to measure the efficacy of laboratories and check for contaminants and impurities in cell cultures. The laboratory proficiency testing is used in departments such as parasitology, virology, hematology, toxicology, immunology/serology, histopathology, urinalysis, and others. The end-users of laboratory proficiency testing include hospitals, contract research organizations, pharmaceutical & biotechnology companies, academic research, diagnostic laboratories, independent laboratories, and specialty laboratories.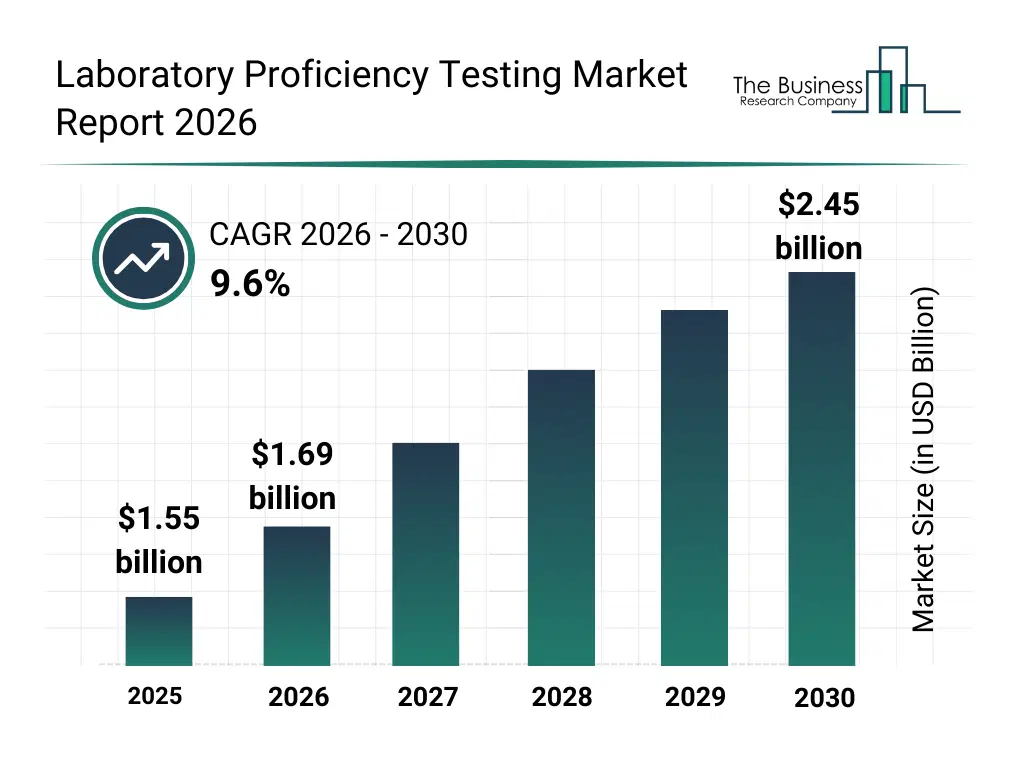
What Is The Laboratory Proficiency Testing Market Size and Share 2026?
The laboratory proficiency testing market size has grown strongly in recent years. It will grow from $1.55 billion in 2025 to $1.69 billion in 2026 at a compound annual growth rate (CAGR) of 9.1%. The growth in the historic period can be attributed to growing regulatory requirements for laboratory accreditation, established need for inter-laboratory comparison, expansion of diagnostic and testing laboratories, rising focus on quality assurance practices, increasing adoption of standardized testing protocols.What Is The Laboratory Proficiency Testing Market Growth Forecast?
The laboratory proficiency testing market size is expected to see strong growth in the next few years. It will grow to $2.45 billion in 2030 at a compound annual growth rate (CAGR) of 9.6%. The growth in the forecast period can be attributed to rising complexity of laboratory testing methods, increasing adoption of advanced diagnostics, growing demand for global laboratory standardization, expansion of clinical and research laboratories, increased scrutiny on laboratory performance and accuracy. Major trends in the forecast period include increasing emphasis on laboratory quality and accreditation, rising adoption of digital and remote proficiency testing programs, growing demand for specialty and advanced proficiency tests, expansion of proficiency testing across multi-disciplinary labs, higher frequency of performance monitoring and benchmarking.Global Laboratory Proficiency Testing Market Segmentation
1) By Technology: Cell Culture, Polymerase Chain Reaction (PCR), Immunoassays, Chromatography, Spectrophotometry, Other Technologies 2) By Test: Routine, Specialty 3) By Department: Parasitology, Virology, Hematology, Toxicology, Immunology/Serology, Histopathology, Urinalysis, Other Departments 4) By End-Use: Hospitals, Contract Research Organizations, Pharmaceutical & Biotechnology Companies, Academic Research, Diagnostic Laboratories, Independent Laboratories, Specialty Laboratories Subsegments: 1) By Cell Culture: Adherent Cell Culture, Suspension Cell Culture 2) By Polymerase Chain Reaction (PCR): Quantitative PCR (qPCR), Reverse Transcription PCR (RT-PCR), Digital PCR 3) By Immunoassays: Enzyme-Linked Immunosorbent Assay (ELISA), Radioimmunoassay (RIA), Chemiluminescent Immunoassay (CLIA) 4) By Chromatography: Liquid Chromatography, Gas Chromatography, Ion Chromatography 5) By Spectrophotometry: Ultraviolet-Visible Spectrophotometry, Infrared Spectrophotometry, Atomic Absorption Spectrophotometry 6) By Other Technologies: Mass Spectrometry, Flow Cytometry, Next-Generation Sequencing (NGS)What Is The Driver Of The Laboratory Proficiency Testing Market?
The growth in the biosimilars is expected to propel the growth of the laboratory proficiency testing market going forward. Biosimilars refer to biologic medicinal products that are essentially a replica of an original product that is produced by a separate firm. As the biosimilar market expands, there is a greater need for analytical and bioanalytical testing of these products. This can lead to an increased demand for laboratory proficiency testing services to ensure the accuracy and reliability of the test results. For instance, in January 2023, according to the Cardinal Health Inc., a US-based healthcare company, as of January 2022, the U.S. had 33 FDA-approved biosimilar products, with 21 available on the market. By 2023, this number increased to 40 FDA-approved biosimilars, with 25 of them commercially available. Therefore, the growth in the biosimilars market is driving the growth of the laboratory proficiency testing industry.Key Players In The Global Laboratory Proficiency Testing Market
Major companies operating in the laboratory proficiency testing market are LGC Limited, American Proficiency Institute, College of American Pathologists, Bio-Rad Laboratories, Randox Laboratories, Merck KGaA, FAPAS, QACS International Pvt. Ltd., Wales External Quality Assessment Scheme, AOAC INTERNATIONAL, Bureau Interprofessionnel d'Études Analytiques, SPEX CertiPrep Group LLC, Absolute Standards Inc, Trilogy Analytical Laboratories, NSI Lab Solutions, INSTAND e.V., Anresco Laboratories, BioScreen Testing Services Inc., EMSL Analytical Inc., Eurofins Scientific SE, Food Safety Net Services LLC, Analytical Laboratory Services Inc., Singapore Accreditation Council, Proctor & Allan East Africa LimitedGlobal Laboratory Proficiency Testing Market Trends and Insights
Major companies operating in the aboratory proficiency testing market are increasingly focusing on developing innovative solutions, such as web-based proficiency testing laboratory information management system, to meet the rising demand for scalable, user-friendly, and secure proficiency testing workflows driven by the need for digital transformation, regulatory compliance, and more efficient inter-laboratory comparisons. A web-based proficiency testing laboratory information management system is a cloud-accessible system that allows participating labs to submit test results, view reports, and track their performance through a browser in contrast to traditional paper-based or desktop-only PT systems that rely on manual entry, offline reporting, and fragmented workflows. For instance, in June 2025, APHA Scientific, a UK-based provider of veterinary proficiency testing (VETQAS), launched PT-LIMS, their modernised proficiency testing management platform. The system features a revamped, intuitive user interface, clearer reporting functionalities, enhanced accessibility, and improved data security with GDPR-compliant safeguards. It supports participants in submitting results online, processes those results to generate tabulated reports, and notifies users automatically when the reports are available. By digitising and centralising the PT workflow, PT-LIMS reduces administrative burden, improves turnaround time for feedback, and enhances traceability and user experience.What Are Latest Mergers And Acquisitions In The Laboratory Proficiency Testing Market?
In February 2025, Cormay Diagnostics S.A., a Poland-based healthcare technology company, partnered with Randox Laboratories Ltd. to make its IVD solutions available within the RIQAS external quality-assurance program. With this partnership, Cormay Diagnostics S.A. and Randox Laboratories Ltd. aim to improve diagnostic quality worldwide by integrating Cormay’s assay portfolio into the RIQAS proficiency-testing framework to enhance result accuracy, regulatory compliance, and laboratory performance monitoring across hematology, clinical chemistry, and immunochemistry. Randox Laboratories Ltd. is a UK-based diagnostics company that specializes in external quality-assessment services and laboratory quality-control solutions, including the RIQAS program.Regional Outlook
North America was the largest region in the laboratory proficiency testing market in 2025Asia-Pacific is expected to be the fastest-growing region in the global laboratory proficiency testing market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Laboratory Proficiency Testing Market?
The laboratory proficiency testing market includes revenues earned by entities providing services such as sample distribution, testing and analysis, result reporting, data evaluation and analysis, performance assessment, feedback, improvement, accreditation, and compliance. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Laboratory Proficiency Testing Market Report 2026?
The laboratory proficiency testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the laboratory proficiency testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Laboratory Proficiency Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.69 billion |
| Revenue Forecast In 2035 | $2.45 billion |
| Growth Rate | CAGR of 9.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Test, Department, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | LGC Limited, American Proficiency Institute, College of American Pathologists, Bio-Rad Laboratories, Randox Laboratories, Merck KGaA, FAPAS, QACS International Pvt. Ltd., Wales External Quality Assessment Scheme, AOAC INTERNATIONAL, Bureau Interprofessionnel d'Études Analytiques, SPEX CertiPrep Group LLC, Absolute Standards Inc, Trilogy Analytical Laboratories, NSI Lab Solutions, INSTAND e.V., Anresco Laboratories, BioScreen Testing Services Inc., EMSL Analytical Inc., Eurofins Scientific SE, Food Safety Net Services LLC, Analytical Laboratory Services Inc., Singapore Accreditation Council, Proctor & Allan East Africa Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
