
Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Report 2026
Global Outlook – By Type (Instruments, Reagents And Consumables, Software And Services, Panels And Assays, Sample Preparation Kits), By Application (Microbiology, Infectious Diseases, Oncology, Genetic Tests, Other Applications), By End User (Research Centers And Academics, Hospitals And Clinical Laboratories, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Overview
• Next-Generation Sequencing (NGS)-Based Molecular Diagnostics market size has reached to $2.26 billion in 2025 • Expected to grow to $3.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.3% • Growth Driver: Increasing Incidence Of Infectious Diseases Fueling The Growth Of The Market Due To Rising Travel And Pathogen Spread • Market Trend: FDA-Approved NGS Panels Enable Comprehensive Detection Of Mutations And Genomic Variations • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market?
Next-generation sequencing (NGS)-based molecular diagnostics is a technology-driven approach that utilizes high-throughput sequencing to analyze genetic material with high accuracy and speed. It enables the detection of genetic variations, mutations, and disease markers at a molecular level, providing deeper insights into the genetic basis of health and disease. This approach supports precise, data-driven diagnostics by combining sequencing technologies with advanced bioinformatics. The main types of next-generation sequencing (NGS)-based molecular diagnostics are instruments, reagents and consumables, software and services, panels and assays, and sample preparation kits. Next-generation sequencing instruments are the hardware platforms, including sequencers and automation workstations, used to physically perform high-throughput DNA/RNA sequencing. The various applications include microbiology, infectious diseases, oncology, genetic tests, and others, and cater to several end-users, including research centres and academics, hospitals and clinical laboratories, and others.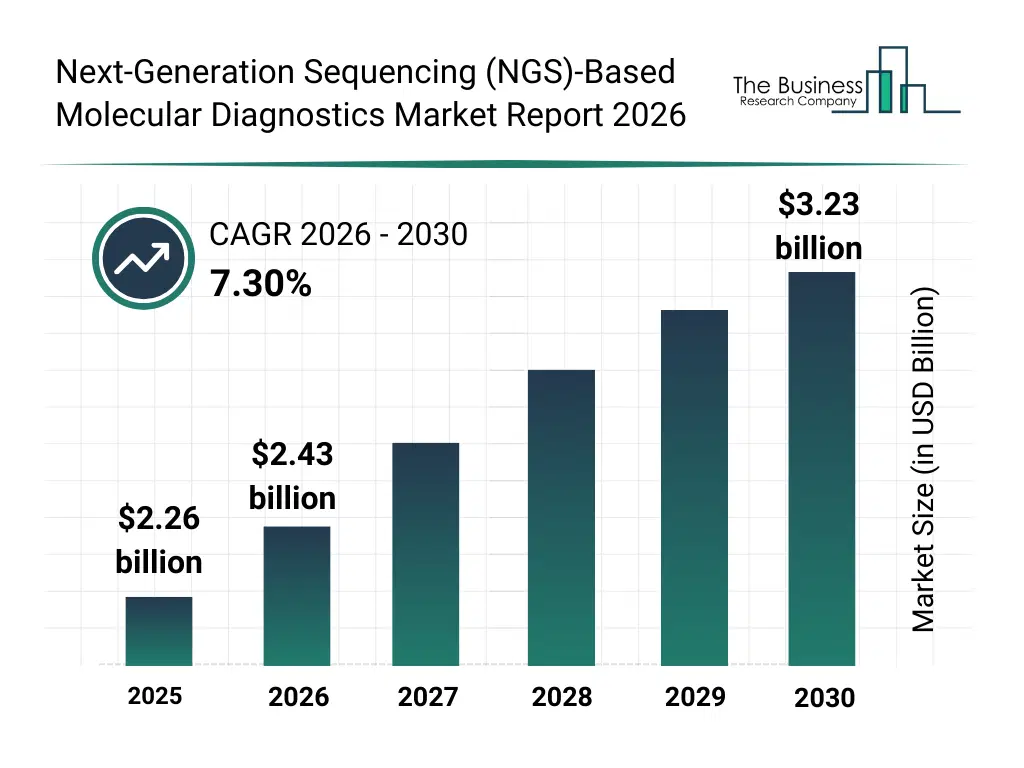
What Is The Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Size and Share 2026?
The next-generation sequencing (ngs)-based molecular diagnostics market size has grown strongly in recent years. It will grow from $2.26 billion in 2025 to $2.43 billion in 2026 at a compound annual growth rate (CAGR) of 7.6%. The growth in the historic period can be attributed to growth in genetic disorder testing, development of high-throughput sequencing, rising awareness of molecular diagnostics, adoption in clinical labs, technological improvements in sequencing reagents.What Is The Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Growth Forecast?
The next-generation sequencing (ngs)-based molecular diagnostics market size is expected to see strong growth in the next few years. It will grow to $3.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.3%. The growth in the forecast period can be attributed to integration of ai and machine learning in diagnostics, expansion of cloud-based genomic platforms, development of portable and point-of-care sequencing devices, rise in personalized medicine applications, collaborations between diagnostics and biotech firms. Major trends in the forecast period include AI-assisted molecular diagnostics, cloud genomic analysis platforms, portable sequencing devices, integration of bioinformatics pipelines, rapid disease marker detection.Global Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Segmentation
1) By Type: Instruments, Reagents And Consumables, Software And Services, Panels And Assays, Sample Preparation Kits 2) By Application: Microbiology, Infectious Diseases, Oncology, Genetic Tests, Other Applications 3) By End User: Research Centers And Academics, Hospitals And Clinical Laboratories, Other End Users Subsegments: 1) By Instruments: Benchtop Sequencers, High-Throughput Sequencers, Portable Or Handheld Sequencers, Clinical Or Diagnostic Sequencers, Automated Library Preparation Systems, Data Analysis And Bioinformatics Hardware 2) By Reagents And Consumables: Sequencing Reagents And Kits, Library Preparation Reagents, Target Enrichment Reagents, Amplification Reagents, Sequencing Flow Cells And Chips, Deoxyribonucleic Acid (DNA) Or Ribonucleic Acid (RNA) Extraction Reagents, Quality Control Reagents 3) By Software And Services: Data Analysis Software, Bioinformatics Pipelines, Cloud-Based Genomic Platforms, Clinical Interpretation Tools, Data Storage And Management Services, Custom Bioinformatics Services, Genomic Database Services 4) By Panels And Assays: Oncology Panels, Infectious Disease Panels, Carrier Screening Panels, Reproductive Health Panels, Rare Disease Or Genetic Disorder Panels, Pharmacogenomics Panels, Whole-Exome Or Whole-Genome Assays 5) By Sample Preparation Kits: Deoxyribonucleic Acid (DNA) Extraction Kits, Ribonucleic Acid (RNA) Extraction Kits, Library Preparation Kits, Target Enrichment Kits, Amplification And Cleanup Kits, Single-Cell Preparation Kits, Low-Input Or Degraded Sample KitsWhat Is The Driver Of The Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market?
The increasing incidence of infectious diseases is expected to propel the growth of the next-generation sequencing (NGS)-based molecular diagnostics market going forward. Infectious diseases are disorders caused by microorganisms such as bacteria, viruses, fungi, or parasites that invade the body, multiply, and disrupt normal body functions. The prevalence of infectious diseases is increasing as rising travel and migration accelerate the transmission of pathogens across regions and borders. Next-generation sequencing (NGS)-based molecular diagnostics helps detect infectious diseases by providing comprehensive and precise analysis of pathogens. It reduces diagnostic time by simultaneously identifying multiple microorganisms, improving treatment accuracy and patient care. For instance, in February 2024, according to the UK Health Security Agency, a UK-based government agency, tuberculosis (a contagious bacterial infection that primarily affects the lungs) cases in England rose to 4,850 in 2023, an increase of 10.7% from 4,380 cases in 2022. Therefore, the increasing incidence of infectious diseases is driving the growth of the next-generation sequencing (NGS)-based molecular diagnostics industry.Key Players In The Global Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market
Major companies operating in the next-generation sequencing (ngs)-based molecular diagnostics market are Thermo Fisher Scientific, Siemens Healthineers AG, Agilent Technologies Inc., Illumina Inc., PerkinElmer Inc., Bio-Rad Laboratories Inc., Qiagen N.V., Amoy Diagnostics Co. Ltd., Tempus Labs Inc., Myriad Genetics Inc., Epigenomics AG, Precision for Medicine Inc., Invitae Corporation, NeoGenomics Laboratories Inc., Guardant Health Inc., Caris Life Sciences Inc., Foundation Medicine Inc., Oxford Nanopore Technologies Ltd., Freenome Inc., Burning Rock Biotech, Element Biosciences Inc., Asuragen Inc., Bionano Genomics Inc., Genomic Health Inc., TATAA Biocenter ABGlobal Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Trends and Insights
Major companies operating in the next-generation sequencing (NGS)-based molecular diagnostics market are focusing on developing advanced next-generation sequencing (NGS)-based panels to enhance the accuracy, accessibility, and efficiency of genetic testing. NGS panels are targeted sets of genes or genomic regions analyzed simultaneously using NGS technology to detect clinically relevant mutations for faster and more precise molecular diagnostics. For instance, in April 2024, Pillar Biosciences, a US-based diagnostics company, received FDA approval for its oncoReveal CDx pan-cancer solid tumor IVD kit, which analyzes 22 clinically relevant genes in a single multiplex reaction. Designed for use with Illumina’s MiSeq Dx system, the panel supports a sample-to-report time of approximately 48 hours and can process up to 46 clinical samples per run. OncoReveal CDx includes companion diagnostic claims for KRAS (for EGFR-targeted therapy in colorectal cancer) and EGFR (TKI therapy in non–small-cell lung cancer), enabling more comprehensive and efficient detection of actionable genomic alterations to improve clinical decision-making.What Are Latest Mergers And Acquisitions In The Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market?
In January 2023, QIAGEN NV, a Germany-based molecular diagnostics and life sciences company, acquired Verogen Inc. for an undisclosed amount. With this acquisition, QIAGEN aims to strengthen its leadership in human identification and forensic genomics by integrating Verogen’s specialized NGS technologies into its broader molecular diagnostics portfolio, expanding its reach in both public sector forensics and private sector applications such as genetic genealogy. Verogen Inc. is a US-based company that specializes in next-generation sequencing (NGS) solutions for forensic genomics.Regional Outlook
North America was the largest region in the next-generation sequencing (NGS)-based molecular diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market?
The next-generation sequencing (NGS)-based molecular diagnostics market includes revenues earned by entities by providing services, such as clinical genomic testing, cancer genomic profiling, infectious disease detection, liquid biopsy testing, and pharmacogenomics testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The next-generation sequencing (NGS)-based molecular diagnostics market also includes sales of tumor mutation panels, carrier screening kits, drug response panels, companion diagnostic kits, and whole-genome metagenomics kits. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Next-Generation Sequencing (NGS)-Based Molecular Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.43 billion |
| Revenue Forecast In 2035 | $3.23 billion |
| Growth Rate | CAGR of 7.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific, Siemens Healthineers AG, Agilent Technologies Inc., Illumina Inc., PerkinElmer Inc., Bio-Rad Laboratories Inc., Qiagen N.V., Amoy Diagnostics Co. Ltd., Tempus Labs Inc., Myriad Genetics Inc., Epigenomics AG, Precision for Medicine Inc., Invitae Corporation, NeoGenomics Laboratories Inc., Guardant Health Inc., Caris Life Sciences Inc., Foundation Medicine Inc., Oxford Nanopore Technologies Ltd., Freenome Inc., Burning Rock Biotech, Element Biosciences Inc., Asuragen Inc., Bionano Genomics Inc., Genomic Health Inc., TATAA Biocenter AB |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
