
Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Report 2026
Global Outlook – By Dosage Form (Oral Solids, Oral Liquids, Injectables, Topicals, Inhalation Products, Transdermal And Patches, Other Dosage Forms), By Therapeutic Area (Oncology, Cardiology, Central Nervous System, Gastroenterology, Infectious Diseases, Endocrinology), By End-User (Pharmaceutical Companies, Biopharmaceutical Companies, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Overview
• Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations market size has reached to $54.73 billion in 2025 • Expected to grow to $90.76 billion in 2030 at a compound annual growth rate (CAGR) of 10.6% • Growth Driver: The Growing Drug Development And Manufacturing Activities Is Fueling The Growth Of The Market Due To Rising Demand For Novel Therapeutics And Accelerated Production Timelines • Market Trend: Advancement Of High-Concentration Biologic Formulation Platforms To Enhance Drug Stability And Delivery Efficiency • Asia-Pacific was the largest region in 2025 and North America is the fastest growing region.What Is Covered Under Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market?
A pharmaceutical contract development and manufacturing organization (CDMO) for formulations refers to a company that provides outsourced services to develop and produce drug formulations for pharmaceutical firms. These services include activities such as formulation development, analytical testing, clinical trial material production, and large-scale commercial manufacturing. The main types of dosage forms for pharmaceutical contract development and manufacturing organizations (CDMO) for formulations are oral solids, oral liquids, injectables, topicals, inhalation products, transdermals and patches, and others. Oral solid dosage forms are medications taken by mouth, formulated to deliver the drug within the digestive system. It is used in various therapeutic areas such as oncology, cardiology, central nervous system, gastroenterology, infectious diseases, and endocrinology, and is used by several end-users, including pharmaceutical companies, biopharmaceutical companies, and others.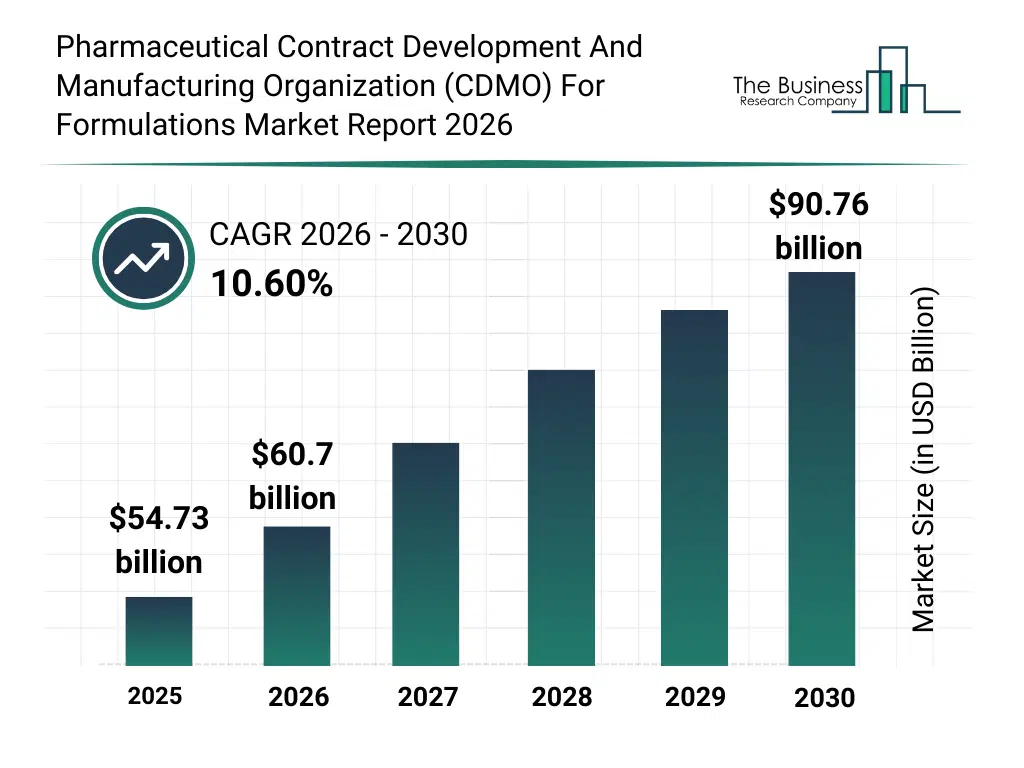
What Is The Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Size and Share 2026?
The pharmaceutical contract development and manufacturing organization (cdmo) for formulations market size has grown rapidly in recent years. It will grow from $54.73 billion in 2025 to $60.7 billion in 2026 at a compound annual growth rate (CAGR) of 10.9%. The growth in the historic period can be attributed to limited availability of specialized peptide api manufacturers, reliance on in-house peptide production, growing peptide therapeutics pipelines, increasing regulatory compliance requirements, adoption of basic chemical synthesis methods.What Is The Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Growth Forecast?
The pharmaceutical contract development and manufacturing organization (cdmo) for formulations market size is expected to see rapid growth in the next few years. It will grow to $90.76 billion in 2030 at a compound annual growth rate (CAGR) of 10.6%. The growth in the forecast period can be attributed to advancements in custom peptide synthesis technologies, expansion of commercial-scale gmp manufacturing, increasing outsourcing to cros and smes, development of lifecycle management and process optimization strategies, rising demand for clinical and preclinical peptide api manufacturing. Major trends in the forecast period include rising outsourcing of peptide api manufacturing to contract manufacturers, growth in gmp-grade production for clinical and commercial supply, increasing demand for custom peptide synthesis and purification services, expansion of peptide therapeutics pipelines across pharmaceutical companies, focus on process optimization and lifecycle management for peptide apis.Global Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Segmentation
1) By Dosage Form: Oral Solids, Oral Liquids, Injectables, Topicals, Inhalation Products, Transdermal And Patches, Other Dosage Forms 2) By Therapeutic Area: Oncology, Cardiology, Central Nervous System, Gastroenterology, Infectious Diseases, Endocrinology 3) By End-User: Pharmaceutical Companies, Biopharmaceutical Companies, Other End-Users Subsegments: 1) By Oral Solids: Tablets, Capsules, Granules, Powders, Modified-Release Formulations 2) By Oral Liquids: Syrups, Suspensions, Solutions, Emulsions, Elixirs 3) By Injectables: Vials, Ampoules, Prefilled Syringes, Lyophilized Injections, Injectable Emulsions 4) By Topicals: Creams, Ointments, Gels, Lotions, Foams, 5) By Inhalation Products: Metered Dose Inhalers (MDIs), Dry Powder Inhalers (DPIs), Nebulizer Solutions, Nasal Sprays, Inhalation Capsules 6) By Transdermal And Patches: Matrix Patches, Reservoir Patches, Drug-In-Adhesive Patches, Microneedle Patches, Iontophoretic Patches 7) By Other Dosage Forms: Suppositories, Ophthalmic Preparations, Buccal Tablets, Sublingual Tablets, ImplantsWhat Is The Driver Of The Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market?
The growing drug development and manufacturing activities are expected to propel the growth of the pharmaceutical contract development and manufacturing organization (CDMO) for formulations market going forward. Drug development and manufacturing activities refer to the increasing efforts by pharmaceutical and biopharmaceutical companies to research, develop, and produce new drugs. Drug development and manufacturing activities are rising primarily due to the growing global demand for novel and effective therapeutics, which pushes companies to expand their development pipelines and speed up manufacturing timelines. A pharmaceutical CDMO aids drug development by formulating and optimizing dosage forms, performing stability testing, and ensuring regulatory compliance. It also manages scale-up and production, enabling faster, cost-efficient product launches. For instance, in June 2023, according to the European Federation of Pharmaceutical Industries and Associations (EFPIA), a Belgium-based trade association that represents the pharmaceutical industry in Europe, the total pharmaceutical production in Europe accounted for $422,803 million (€390,000 million) in 2023, up from $393,857 million (€363,300 million) in 2022. Therefore, growing drug development and manufacturing activities are driving the growth of the pharmaceutical contract development and manufacturing organization (CDMO) for formulations industry.Key Players In The Global Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market
Major companies operating in the pharmaceutical contract development and manufacturing organization (cdmo) for formulations market are Thermo Fisher Scientific Inc., Lonza Group, Catalent Inc., WuXi Biologics, Samsung Biologics, Recipharm AB, Siegfried Holding AG, Evonik Industries AG, Almac Group, Aenova Group, Piramal Pharma Limited, Cambrex, Jubilant Pharmova Limited, CordenPharma, Altasciences, Hovione, Aurigene Pharmaceutical Services Ltd., DPT Laboratories Ltd., CARBOGEN AMCIS, Biopharma Group, LGM Pharma, Aarti Pharmalabs Limited.Global Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Trends and Insights
Major companies operating in the pharmaceutical contract development and manufacturing organization (CDMO) for formulations market are focusing on developing advanced solutions, such as biologic formulation platforms, to enhance drug stability and accelerate time-to-market for complex biologic therapies. Biologic formulation platforms are specialized systems designed to create stable and efficient formulations of biologic drugs. They fine-tune elements such as pH, excipients, and delivery methods to enhance stability, bioavailability, and safety across the product lifecycle. For instance, in June 2025, WuXi Biologics, a China-based contract research, development, and manufacturing organization, launched WuXiHigh 2.0, a next-generation high-concentration formulation platform. This advanced platform enables biologic formulations with protein concentrations up to 230 mg/mL, surpassing the current Food and Drug Administration-approved maximum of 200 mg/mL, while reducing viscosity by up to 90% through proprietary excipient blends and high-throughput instrumentation. It addresses key challenges in developing high-concentration biologics, such as viscosity and aggregation, and is designed to improve injection efficiency, patient compliance, and manufacturing outcomes across clinical and commercial stages.What Are Latest Mergers And Acquisitions In The Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market?
In February 2025, Jabil Inc., a US-based provider of engineering, manufacturing, and supply chain solutions, acquired Pharmaceutics International Inc. (Pii) for an undisclosed amount. The acquisition aims to enhance Jabil’s pharmaceutical manufacturing and development capabilities by leveraging Pii’s expertise in aseptic filling, lyophilization, and oral solid dose production. This strengthens Jabil’s position in parenteral drug delivery and expands its end-to-end support across clinical and commercial manufacturing. Pharmaceutics International Inc. (Pii) is a US-based pharmaceutical contract development and manufacturing organization that provides formulation development services for injectable to oral drug forms.Regional Insights
Asia-Pacific was the largest region in the pharmaceutical contract development and manufacturing organization (CDMO) for formulations market in 2025. North America is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market?
The pharmaceutical contract development and manufacturing organization (CDMO) for formulations market consists of revenues earned by entities by providing services such as analytical and stability testing, formulation development, process development and scale-up, regulatory support and documentation, and commercial-scale manufacturing and packaging. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharmaceutical contract development and manufacturing organization (CDMO) for formulations market also includes sales of suppositories, ophthalmic preparations, buccal and sublingual tablets, liposomal formulations, and microneedle patches. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Report 2026?
The pharmaceutical contract development and manufacturing organization (cdmo) for formulations market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical contract development and manufacturing organization (cdmo) for formulations industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Contract Development And Manufacturing Organization (CDMO) For Formulations Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $60.7 billion |
| Revenue Forecast In 2035 | $90.76 billion |
| Growth Rate | CAGR of 10.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Dosage Form, Therapeutic Area, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Lonza Group, Catalent Inc., WuXi Biologics, Samsung Biologics, Recipharm AB, Siegfried Holding AG, Evonik Industries AG, Almac Group, Aenova Group, Piramal Pharma Limited, Cambrex, Jubilant Pharmova Limited, CordenPharma, Altasciences, Hovione, Aurigene Pharmaceutical Services Ltd., DPT Laboratories Ltd., CARBOGEN AMCIS, Biopharma Group, LGM Pharma, Aarti Pharmalabs Limited. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
