
Biologics CDMO Market Report 2026
Global Outlook – By Cell Type (Mammalian, Non-Mammalian), By Product Type (Biologics, Biosimilars), By Service Type (Drug Substance Manufacturing, Drug Product (Fill–Finish) Manufacturing, Process Development, Analytical & Quality Testing Services) – Market Size, Trends, Strategies, and Forecast to 2035
Biologics CDMO Market Overview
• Biologics CDMO market size has reached to $24.15 billion in 2025 • Expected to grow to $48.07 billion in 2030 at a compound annual growth rate (CAGR) of 14.9% • Growth Driver: Rise In Demand For Cell And Genetic Therapy Fuels Biologics CDMO Market Growth • Market Trend: Advancements In Biologics CDMO • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Biologics CDMO Market?
A biologics contract development and manufacturing organization (CDMO) is a service provider that supports pharmaceutical companies in the development and production of biologic drugs, which include vaccines, blood and blood components, allergens, somatic cells, gene therapies, tissues, and recombinant therapeutic proteins. The main cell types in biologics CDMO are mammalian and non-mammalian. Mammalian refers to cell lines derived from mammals, commonly used in the production of biologics and cell-based therapies due to their ability to produce complex proteins and maintain post-translational modifications. The various products types include biologics and biosimilars.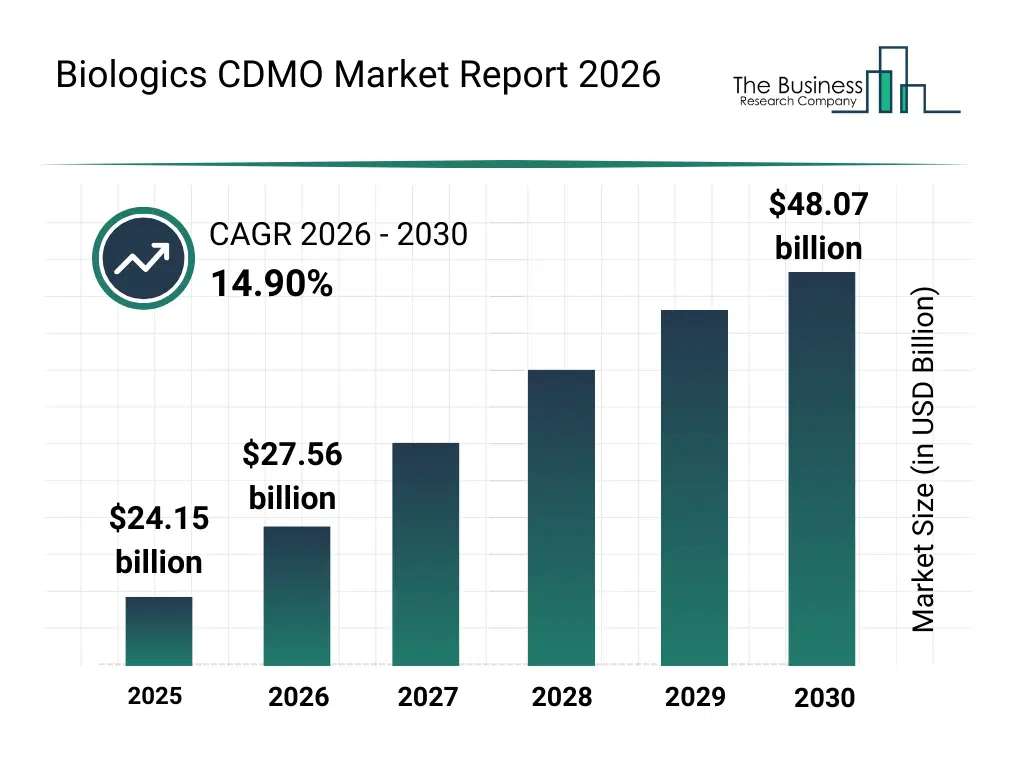
What Is The Biologics CDMO Market Size and Share 2026?
The biologics cdmo market size has grown rapidly in recent years. It will grow from $24.15 billion in 2025 to $27.56 billion in 2026 at a compound annual growth rate (CAGR) of 14.1%. The growth in the historic period can be attributed to rise in biologics research, advancements in cell culture technologies, increasing prevalence of chronic diseases, growth of biosimilar pipelines, government support for biopharma manufacturing.What Is The Biologics CDMO Market Growth Forecast?
The biologics cdmo market size is expected to see rapid growth in the next few years. It will grow to $48.07 billion in 2030 at a compound annual growth rate (CAGR) of 14.9%. The growth in the forecast period can be attributed to surge in gene and cell therapy development, adoption of automation and robotics in CDMO processes, expansion of global biologics manufacturing capacity, increasing investment in precision medicine, rising demand for cost-effective biologics. Major trends in the forecast period include expansion of biologics cdmo services, growth in mammalian cell-based production, increase in outsourcing of gene therapy manufacturing, adoption of advanced bioprocessing techniques, rising demand for biosimilars.Global Biologics CDMO Market Segmentation
1) By Cell Type: Mammalian, Non-Mammalian 2) By Product Type: Biologics, Biosimilars 3) By Service Type: Drug Substance Manufacturing, Drug Product (Fill–Finish) Manufacturing, Process Development, Analytical & Quality Testing Services Subsegments: 1) By Mammalian: Chinese Hamster Ovary (CHO) Cells, Human Embryonic Kidney (HEK) Cells, Other Mammalian Cells 2) By Non-Mammalian: Yeast Cells, Bacterial Cells, Insect Cells, Other Non-Mammalian CellsWhat Is The Driver Of The Biologics CDMO Market?
The surge in demand for cell and genetic therapy is expected to propel the growth of the biologic CDMO market going forward. Cell and gene therapy are innovative medical approaches that utilize living cells or genetic material to address underlying disease causes at the cellular or genetic level, providing potential treatments for conditions that are difficult to manage or incurable with traditional pharmaceuticals. The demand for cell and gene therapy is rising due to their potential to offer innovative treatments for diseases by addressing underlying genetic or cellular causes. Biologic CDMOs provide expertise in handling living cells, viral vectors, and genetic material, facilitating safe and efficient production of cell and gene therapies while assisting biopharmaceutical companies in navigating regulatory requirements and technical challenges, thereby facilitating translation from research to clinical application. For instance, in March 2024, according to the IQVIA Holdings Inc., a US-based provider of advanced analytics, technology solutions, and clinical research services for the life sciences and healthcare industries, global spending on cell and gene therapies surged to $5.9 billion in 2023, an increase of 38% from 2022. Therefore, the surge in demand for cell and genetic therapy is driving the growth of the biologic CDMO market.Key Players In The Global Biologics CDMO Market
Major companies operating in the biologics cdmo market are Lonza Group Ltd, Samsung Biologics Co Ltd, WuXi Biologics Cayman Inc, Catalent Inc, Thermo Fisher Scientific Inc, Boehringer Ingelheim International GmbH, Emergent BioSolutions Inc, Grifols SA, AbbVie Inc, Novartis AG, Sandoz International GmbH, Fujifilm Holdings Corporation, AGC Inc, JSR Corporation, Shenzhen Hepalink Pharmaceutical Co Ltd, Kemwell Biopharma Pvt Ltd, Bora Pharmaceuticals Co Ltd, Binex Co Ltd, 3P Biopharmaceuticals SLU, Cytovance BiologicsGlobal Biologics CDMO Market Trends and Insights
Major companies operating in the biologics CDMO market are developing technologically advanced solutions, such as new cell lines, to enhance biopharmaceutical development and manufacturing efficiency and capabilities. The technology produces therapeutic antibodies with enhanced potency, addressing immunotherapy research and development challenges. For instance, in November 2023, Lonza Group, a Switzerland-based manufacturing company that specializes in the pharmaceutical and biotechnology sectors, launched GS Effex cell lines, designed to produce antibodies that are 100% free of fucose, a sugar molecule and demonstrate increased potency without compromising on other performance attributes. It helps overcome immunotherapy research and development challenges related to the need for antibody therapeutics to increase potency. It is a stable, scalable, and productive cell line that provides a solution for therapeutic development from discovery-stage research through commercial manufacturing.What Are Latest Mergers And Acquisitions In The Biologics CDMO Market?
In December 2024, Novo Holdings A/S, a Denmark-based investment company, completed the acquisition of Catalent, Inc., for an undisclosed amount. With this acquisition, Novo Holdings aims to strengthen its portfolio in the contract development and manufacturing organization (CDMO) sector, enhance global manufacturing capabilities, and support the expansion of innovative drug development solutions. Catalent, Inc., is a US-based CDMO that provides advanced delivery technologies, development, and manufacturing services for drugs, biologics, and consumer health products.Regional Insights
North America was the largest region in the biologics CDMO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biologics CDMO Market?
The biologics CDMO market consist of revenues earned by entities by providing services such as process development, analytical development, cell line development, supply chain management, and fill-finish services. The biologics CDMO market also includes sales of monoclonal antibodies, recombinant proteins, antisense and molecular therapy, and vaccines. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biologics CDMO Market Report 2026?
The biologics cdmo market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biologics cdmo industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biologics CDMO Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $27.56 billion |
| Revenue Forecast In 2035 | $48.07 billion |
| Growth Rate | CAGR of 14.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Cell Type, Product Type, Service Type |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Lonza Group Ltd, Samsung Biologics Co Ltd, WuXi Biologics Cayman Inc, Catalent Inc, Thermo Fisher Scientific Inc, Boehringer Ingelheim International GmbH, Emergent BioSolutions Inc, Grifols SA, AbbVie Inc, Novartis AG, Sandoz International GmbH, Fujifilm Holdings Corporation, AGC Inc, JSR Corporation, Shenzhen Hepalink Pharmaceutical Co Ltd, Kemwell Biopharma Pvt Ltd, Bora Pharmaceuticals Co Ltd, Binex Co Ltd, 3P Biopharmaceuticals SLU, Cytovance Biologics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
