
Sterile Injectables CDMO Market Report 2026
Global Outlook – By Type (Small Molecule, Large Molecule), By Service (Formulation Development, Analytical And Testing Services, Manufacturing, Packaging, Storage, Other Services), By Route Of Administration (Subcutaneous (SC), Intravenous (IV), Intramuscular (IM), Other Routes Of Administration), By Therapeutic Area (Oncology, Cardiovascular Diseases, Central Nervous System Diseases, Infectious Disorders, Musculoskeletal Diseases, Hormonal Diseases, Other Therapeutic Areas), By End-User (Pharmaceutical Companies, Biopharmaceutical Companies, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Sterile Injectables CDMO Market Overview
• Sterile Injectables CDMO market size has reached to $6.65 billion in 2025 • Expected to grow to $10.91 billion in 2030 at a compound annual growth rate (CAGR) of 10.3% • Growth Driver: Surge In Biologics And Biosimilar Demand Fueling Market Growth Due To Rising Chronic Disease Burden And Therapeutic Advancements • Market Trend: advancements In Modular Aseptic Technologies Driving Growth In The Sterile Injectables CDMO • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Sterile Injectables CDMO Market?
Sterile injectables CDMO refers to a contract development and manufacturing organization that specializes in the production of injectable pharmaceutical products under sterile conditions. These organizations operate in compliance with stringent regulatory standards to ensure product safety, efficacy, and quality. They play a crucial role in the pharmaceutical supply chain by providing the infrastructure, expertise, and controlled environments required for aseptic processing. The main types of sterile injectable CDMO include small molecule and large molecule. Small molecules are low molecular weight compounds that can easily enter cells due to their simple chemical structure. It offers various services such as formulation development, analytical and testing services, manufacturing, packaging, storage, and others and is administered through various routes, including subcutaneous (SC), intravenous (IV), intramuscular (IM), and others. It is used for several therapeutic areas such as oncology, cardiovascular diseases, central nervous system diseases, infectious disorders, musculoskeletal diseases, hormonal diseases, and others, by different end-users, including pharmaceutical companies, biopharmaceutical companies, research institutes, and others.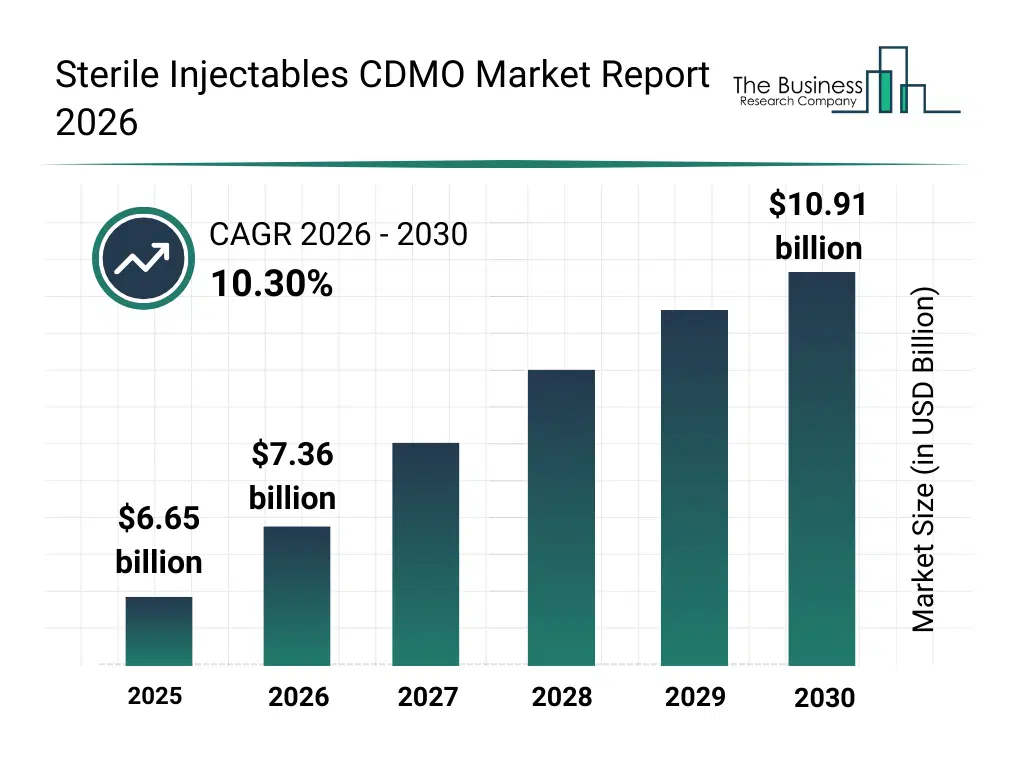
What Is The Sterile Injectables CDMO Market Size and Share 2026?
The sterile injectables cdmo market size has grown rapidly in recent years. It will grow from $6.65 billion in 2025 to $7.36 billion in 2026 at a compound annual growth rate (CAGR) of 10.7%. The growth in the historic period can be attributed to limited cdmo infrastructure for sterile injectables, growing demand for small molecule injectables, reliance on in-house formulation development, increasing regulatory scrutiny for sterile manufacturing, rising global pharmaceutical production.What Is The Sterile Injectables CDMO Market Growth Forecast?
The sterile injectables cdmo market size is expected to see rapid growth in the next few years. It will grow to $10.91 billion in 2030 at a compound annual growth rate (CAGR) of 10.3%. The growth in the forecast period can be attributed to expansion of large molecule biologics manufacturing, increasing demand for cell and gene therapies, adoption of advanced aseptic processing technologies, rising outsourcing to cdmos, growth in specialized packaging and cold chain storage requirements. Major trends in the forecast period include rising demand for contract manufacturing of sterile injectables, expansion of analytical and testing services for quality assurance, growth in monoclonal antibodies, peptides, and cell/gene therapy production, increasing adoption of lyophilized, emulsion, and reconstitutable injectable formats, integration of formulation development with packaging and storage solutions.Global Sterile Injectables CDMO Market Segmentation
1) By Type: Small Molecule, Large Molecule 2) By Service: Formulation Development, Analytical And Testing Services, Manufacturing, Packaging, Storage, Other Services 3) By Route Of Administration: Subcutaneous (SC), Intravenous (IV), Intramuscular (IM), Other Routes Of Administration 4) By Therapeutic Area: Oncology, Cardiovascular Diseases, Central Nervous System Diseases, Infectious Disorders, Musculoskeletal Diseases, Hormonal Diseases, Other Therapeutic Areas 5) By End-User: Pharmaceutical Companies, Biopharmaceutical Companies, Research Institutes, Other End-Users Subsegments: 1) By Small Molecule: Solutions, Suspensions, Lyophilized Injectables, Emulsions, Reconstitutable Powders 2) By Large Molecule: Monoclonal Antibodies (mAbs), Peptides And Proteins, Vaccines, Cell And Gene Therapies, Insulins And BiosimilarsWhat Is The Driver Of The Sterile Injectables CDMO Market?
The increasing demand for biologics and biosimilars is expected to propel the growth of the sterile injectables CDMO market going forward. Biologics are medicines made from living cells or organisms that target specific components of diseases, while biosimilars are nearly identical copies of approved biologics that match them in quality, safety, and effectiveness. The demand for biologics and biosimilars is increasing due to the rising incidence of chronic and complex health conditions, as these innovative therapies offer more targeted and effective treatment solutions compared to traditional medications. The demand for biologics and biosimilars results in a higher reliance on sterile injectable CDMO services, as these treatments are commonly administered via injection and require carefully maintained sterile conditions to ensure safety, quality, and regulatory adherence. For instance, in February 2025, according to the Center for Biosimilars, a US-based nonprofit research organization, the US spent an estimated US $324 billion on biologic medicines in 2023, representing about 45% of the nation’s total prescription drug expenditure. Therefore, the increasing demand for biologics and biosimilars is driving the growth of the sterile injectables CDMO industry.Key Players In The Global Sterile Injectables CDMO Market
Major companies operating in the sterile injectables cdmo market are Pfizer Inc., Thermo Fisher Scientific Inc., Boehringer Ingelheim, Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotechnology Inc.Global Sterile Injectables CDMO Market Trends and Insights
Major companies operating in the Sterile Injectables CDMO market are focusing on developing innovative solutions, such as modular aseptic filling platforms, to enhance flexibility and efficiency in sterile drug production. Modular aseptic filling platforms are flexible isolator-based sterile filling systems built from interchangeable modules that allow CDMOs to quickly switch between vial, syringe, or cartridge formats, helping reduce changeover time, minimize product loss, and efficiently support small- to mid-scale batches for clinical and commercial sterile injectables. For instance, in February 2025, Recipharm AB, a Sweden-based contract development and manufacturing organization (CDMO), launched a fully operational modular sterile filling system at its Wasserburg, Germany site, designed for aseptic filling of small to mid-scale batches (500–50,000 units). The line minimizes product loss (less than 500 milliliters), supports interchangeable filling modules, and enables efficient processing of high-value biologics within a fully enclosed isolator.What Are Latest Mergers And Acquisitions In The Sterile Injectables CDMO Market?
In October 2023, Advent International and Warburg Pincus, US-based private equity firms, acquired Baxter’s BioPharma Solutions business for $4.25 billion. Following the acquisition, the business has transitioned into an independent contract development and manufacturing organization under the name Simtra BioPharma Solutions, maintaining its offerings in sterile manufacturing, parenteral delivery, and specialized support for pharmaceutical and biotech clients. With support from Advent and Warburg Pincus, Simtra now operates as a fully standalone CDMO, delivering end-to-end services ranging from clinical-stage development to commercial fill-finish production. Baxter International is a US-based provider of sterile injectable CDMO services.Regional Insights
North America was the largest region in the sterile injectables CDMO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Sterile Injectables CDMO Market?
The sterile injectables CDMO market consists of revenues earned by entities by providing services such as microbiological testing, sterilization validation, labeling, and regulatory support. The market value includes the value of related goods sold by the service provider or included within the service offering. The sterile injectables CDMO market also includes sales of cartridges, emulsions, infusion bags, and implants. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Sterile Injectables CDMO Market Report 2026?
The sterile injectables cdmo market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the sterile injectables cdmo industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Sterile Injectables CDMO Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.36 billion |
| Revenue Forecast In 2035 | $10.91 billion |
| Growth Rate | CAGR of 10.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Service, Route Of Administration, Therapeutic Area, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Thermo Fisher Scientific Inc., Boehringer Ingelheim, Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotechnology Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
