
In Vitro Diagnostics (IVD) Quality Control Market Report 2026
Global Outlook – By Product type (Quality Control Products, Data Management Solutions, Quality Assurance Services), By Manufacturer Type (IVD Instrument Manufacturers, Third Party Quality Control Manufacturers), By Application (Clinical Chemistry, Hematology, Immunoassay, Molecular Diagnostics, Microbiology, Coagulation Or Hemostasis, Other Applications), By End-Users (Hospitals, Clinical Laboratories, Research And Academic Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
In Vitro Diagnostics (IVD) Quality Control Market Overview
• In Vitro Diagnostics (IVD) Quality Control market size has reached to $1.48 billion in 2025 • Expected to grow to $2.24 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Rise In Prevalence Of Infectious Diseases Driving Growth Of In Vitro Diagnostics Quality Control Market • Market Trend: Newland EMEA Launches Advanced Barcode Scanners to Enhance In Vitro Diagnostics • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In Vitro Diagnostics (IVD) Quality Control Market?
In-vitro diagnostics (IVD) quality control is a set of processes that ensure accuracy, performance, and environmental conditions impact in vitro diagnostic tests including in vitro nucleic acid testing processes for pathogen detection, healthcare-associated infections (HAIs) such as pneumonia, urinary tract infections, and other conditions. The main types of products in in-vitro diagnostics quality control are quality control products, data management solutions, and quality assurance services. The quality control products consist of serum/ plasma-based control products, whole blood-based control products, urine-based control products, and other control products manufactured by the IVD instrument manufacturers to cure mitigate or treat via in vitro diagnostics quality control. The different manufacturer types include IVD instrument manufacturers, and third-party quality control manufacturers and are used in various applications such as clinical chemistry, hematology, immunoassay, molecular diagnostics, microbiology, coagulation, or hemostasis, among others. It is implemented in various areas including hospitals, clinical laboratories, research and academic institutes, and others.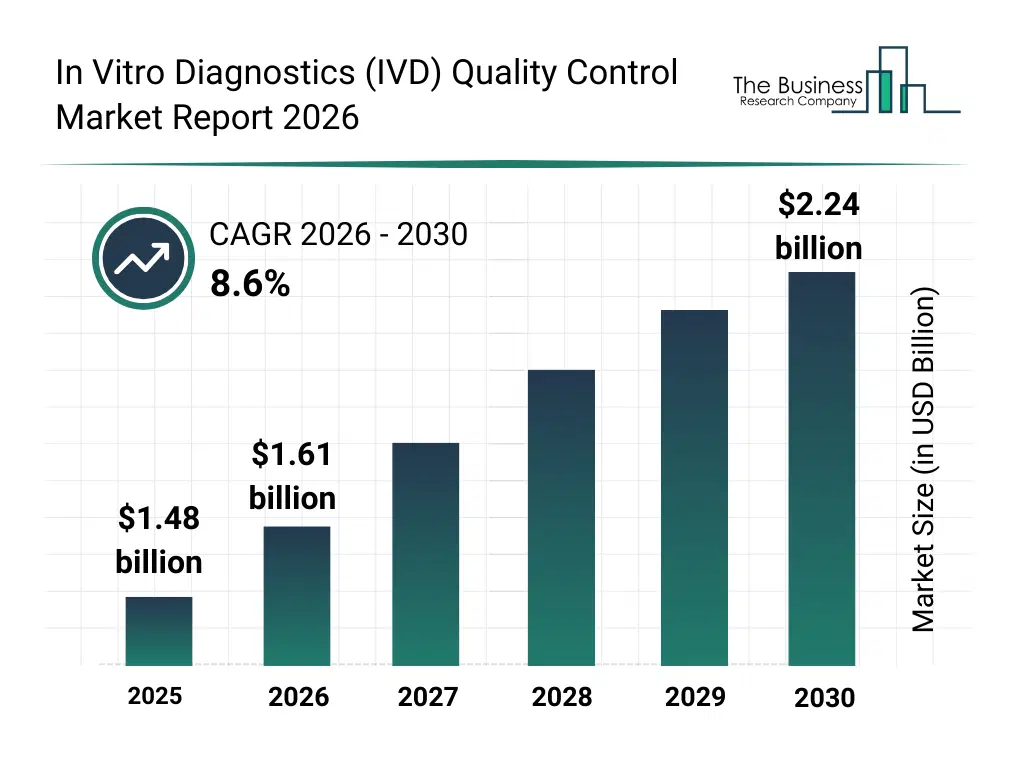
What Is The In Vitro Diagnostics (IVD) Quality Control Market Size and Share 2026?
The in vitro diagnostics (ivd) quality control market size has grown strongly in recent years. It will grow from $1.48 billion in 2025 to $1.61 billion in 2026 at a compound annual growth rate (CAGR) of 8.5%. The growth in the historic period can be attributed to growing burden of infectious diseases, rising adoption of in vitro diagnostic devices, regulatory focus on diagnostic accuracy, expansion of clinical laboratory networks, increasing awareness of testing quality standards.What Is The In Vitro Diagnostics (IVD) Quality Control Market Growth Forecast?
The in vitro diagnostics (ivd) quality control market size is expected to see strong growth in the next few years. It will grow to $2.24 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to rising demand for molecular diagnostics precision, increasing healthcare digitalization, growing adoption of automated lab systems, expanding healthcare infrastructure investments, stricter regulatory compliance requirements. Major trends in the forecast period include increasing emphasis on diagnostic accuracy and reliability, rising focus on quality compliance and standardization, growing adoption of external quality assessment programs, expansion of multiplex and molecular diagnostic testing validation, increasing use of automated quality monitoring systems.Global In Vitro Diagnostics (IVD) Quality Control Market Segmentation
1) By Product type: Quality Control Products, Data Management Solutions, Quality Assurance Services 2) By Manufacturer Type: IVD Instrument Manufacturers, Third Party Quality Control Manufacturers 3) By Application: Clinical Chemistry, Hematology, Immunoassay, Molecular Diagnostics, Microbiology, Coagulation Or Hemostasis, Other Applications 4) By End-Users: Hospitals, Clinical Laboratories, Research And Academic Institutes, Other End Users Subsegments: 1) By Quality Control Products: Control Materials, Quality Control Kits, Proficiency Testing Samples 2) By Data Management Solutions: Software For Quality Control Management, Data Analysis Tools, Cloud-Based Solutions 3) By Quality Assurance Services: Consulting Services, Training And Education Services, Audit And Compliance ServicesWhat Is The Driver Of The In Vitro Diagnostics (IVD) Quality Control Market?
The rising prevalence of infectious diseases is expected to propel the growth of the in vitro diagnostics (IVD) quality control market going forward. Infectious diseases refer to illnesses caused by pathogenic microorganisms such as bacteria, viruses, fungi, or parasites that can spread directly or indirectly between individuals. Infectious diseases are rising due to the growing antimicrobial resistance that makes common infections harder to treat and easier to spread. In vitro diagnostics (IVD) quality control helps ensure accurate and reliable detection of infectious pathogens, enabling timely diagnosis and effective patient management. For instance, in October 2024, according to the World Health Organization, a Switzerland-based international public health organization, approximately 8.2 million people were newly diagnosed with tuberculosis in 2023, up from about 7.5 million in 2022, placing tuberculosis as the leading infectious disease killer. Therefore, the rising prevalence of infectious diseases is driving the growth for the in vitro diagnostics (IVD) quality control industry.Key Players In The Global In Vitro Diagnostics (IVD) Quality Control Market
Major companies operating in the in vitro diagnostics (ivd) quality control market report are Bio-Rad Laboratories Inc., Thermo Fisher Scientific Inc., LGC Limited., Abbott Laboratories, Roche Diagnostics AG, Siemens AG, Ortho Clinical Diagnostics Inc., Helena Laboratories Corporation, Quidel Corporation, Microbiologics Inc., Sun Diagnostics LLC, Fortress Diagnostics Ltd., Seegene Inc., Bio-Techne Corp., Randox Laboratories, Danaher Corporation, Sysmex Corporation, Beckman Coulter Inc., Grifols India Healthcare Private Limited., Hologic Inc., Qiagen NV, Becton Dickinson and Company, Luminex Corporation, BioMerieux SA, Meridian Bioscience Inc., Sekisui Diagnostics LLC, ZeptoMetrix Corporation, Maine Molecular Quality Controls Inc.Global In Vitro Diagnostics (IVD) Quality Control Market Trends and Insights
Major companies operating in the in vitro diagnostics (IVD) quality control market are focusing on integrating advanced technologies such as embedded barcode-scanning systems to enhance efficiency, streamline diagnostic workflows, and reduce processing errors. Embedded barcode-scanning systems are compact fixed-mount devices and smart vision modules that automatically read barcodes and capture data in real time, enabling precise specimen identification, automated data entry, and improved traceability. For instance, in November 2023, according to the Newland EMEA, a Netherlands-based Digital Technology company, launched an innovative line of in vitro diagnostics (IVD) products at the MEDICA trade fair, featuring five advanced barcode scanners tailored for the healthcare sector. These scanners, including the high-speed FM510 and FM530, as well as the versatile FM550 and the smart FM600 camera, are designed to enhance automation in IVD processes, improving diagnostic speed and accuracy. This launch underscores Newland EMEA's commitment to advancing technology in automatic identification and data collection, ultimately aiming to improve patient care through more efficient healthcare solutions.What Are Latest Mergers And Acquisitions In The In Vitro Diagnostics (IVD) Quality Control Market?
In May 2024, Bruker, a US-based Analytical instrumentation company, acquired ELITechGroup for €870 million. This acquisition Bruker is to enhance its position in the molecular diagnostics market by integrating ELITech's innovative sample-to-answer platforms and a diverse portfolio of assays for infectious diseases. ELITech is a US-based in-vitro diagnostics company, develops, manufactures, and markets a comprehensive range of cutting-edge diagnostic products and solutions, including instruments, reagents, and software.Regional Outlook
North America was the largest region in the in vitro diagnostics (IVD) quality control market in 2025.Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the In Vitro Diagnostics (IVD) Quality Control Market?
The in-vitro diagnostics (IVD) quality control market consists of revenues earned by entities that evaluate and validate the reliability of in-vitro diagnostics testing systems. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the In Vitro Diagnostics (IVD) Quality Control Market Report 2026?
The in vitro diagnostics (ivd) quality control market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the in vitro diagnostics (ivd) quality control industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.In Vitro Diagnostics (IVD) Quality Control Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.61 billion |
| Revenue Forecast In 2035 | $2.24 billion |
| Growth Rate | CAGR of 8.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product type, Manufacturer Type, Application, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Bio-Rad Laboratories Inc., Thermo Fisher Scientific Inc., LGC Limited., Abbott Laboratories, Roche Diagnostics AG, Siemens AG, Ortho Clinical Diagnostics Inc., Helena Laboratories Corporation, Quidel Corporation, Microbiologics Inc., Sun Diagnostics LLC, Fortress Diagnostics Ltd., Seegene Inc., Bio-Techne Corp., Randox Laboratories, Danaher Corporation, Sysmex Corporation, Beckman Coulter Inc., Grifols India Healthcare Private Limited., Hologic Inc., Qiagen NV, Becton Dickinson and Company, Luminex Corporation, BioMerieux SA, Meridian Bioscience Inc., Sekisui Diagnostics LLC, ZeptoMetrix Corporation, Maine Molecular Quality Controls Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
