
In Vitro Diagnostics Market Report 2026
Global Outlook – By Diagnostic (Point-of-Care Diagnostics Devices And Equipment, Immunochemistry Diagnostic Devices And Equipment, Clinical Chemistry Diagnostics Devices And Equipment, Molecular Diagnostics Devices And Equipment, Microbiology Diagnostic Devices And Equipment, Hemostasis Diagnostic Devices And Equipment, Hematology Diagnostic Devices And Equipment, Immunohematology Diagnostic Devices And Equipment), By Type of Expenditure (Public, Private), By End User (Hospitals And Clinics, Diagnostic Laboratories, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
In Vitro Diagnostics Market Overview
• In Vitro Diagnostics market size has reached to $136.55 billion in 2025 • Expected to grow to $241.14 billion in 2030 at a compound annual growth rate (CAGR) of 12.2% • Growth Driver: Rising Cancer Prevalence Fuels Robust Growth In The In Vitro Diagnostics Market • Market Trend: New Technologies Enhance Accuracy And Efficienc • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In Vitro Diagnostics Market?
In vitro diagnostics (IVD) refer to tests and procedures performed on biological samples, such as blood, tissue, or urine, taken from the human body to detect diseases, conditions, or infections. These tests are conducted outside of the body in a controlled environment, such as a laboratory, using specialized equipment and reagents. IVDs are crucial for diagnosing a wide range of health conditions, guiding treatment decisions, and monitoring patient health over time. The main types of in-vitro diagnostics equipment are point-of-care diagnostics devices and equipment, immunochemistry diagnostic devices and equipment, clinical chemistry diagnostics devices and equipment, molecular diagnostics devices and equipment, microbiology diagnostic devices and equipment, hemostasis diagnostic devices and equipment, hematology diagnostic devices, and equipment, and immunohematology diagnostic devices, and equipment. Hematology instruments are machines that analyze blood. The hematology instruments perform blood counts, detect proteins or enzymes, and help to diagnose illnesses or genetic defects. The type of expenditure is public and private. The products used are instruments/equipment and disposables. The end-users are hospitals and clinics, diagnostic laboratories, and others.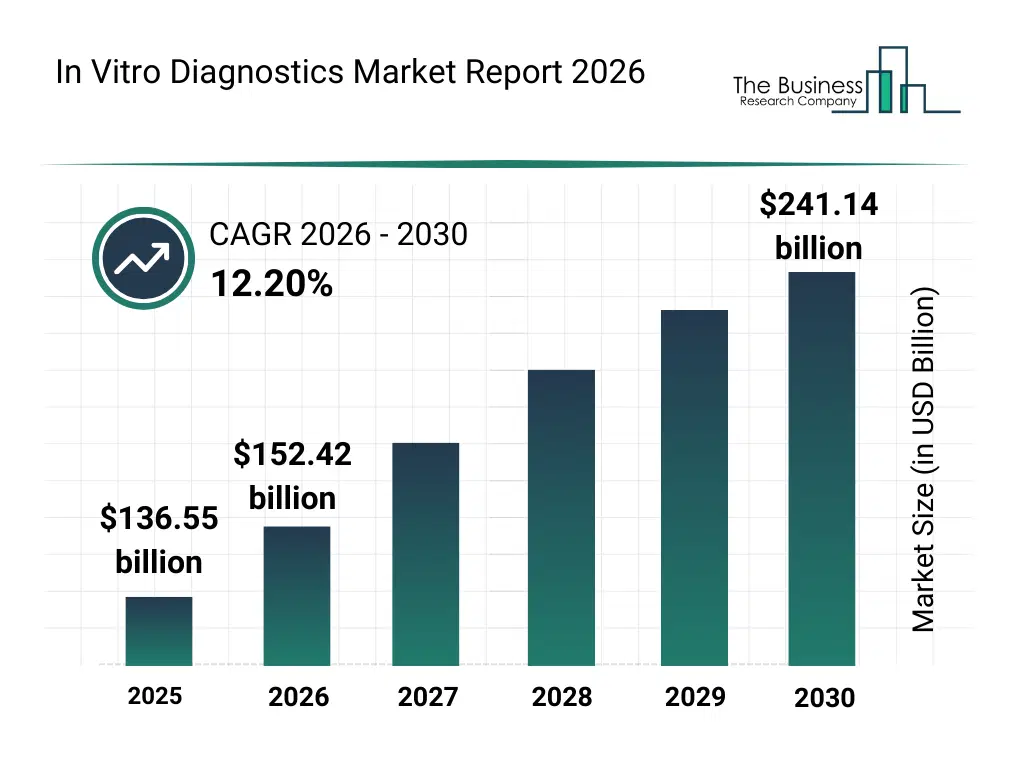
What Is The In Vitro Diagnostics Market Size and Share 2026?
The in vitro diagnostics market size has grown rapidly in recent years. It will grow from $136.55 billion in 2025 to $152.42 billion in 2026 at a compound annual growth rate (CAGR) of 11.6%. The growth in the historic period can be attributed to expansion of diagnostic testing volumes, growth in infectious disease screening, rising burden of chronic diseases, improvements in laboratory infrastructure, increasing healthcare access.What Is The In Vitro Diagnostics Market Growth Forecast?
The in vitro diagnostics market size is expected to see rapid growth in the next few years. It will grow to $241.14 billion in 2030 at a compound annual growth rate (CAGR) of 12.2%. The growth in the forecast period can be attributed to increasing adoption of decentralized diagnostics, rising investments in molecular testing technologies, expansion of personalized medicine diagnostics, growing demand for rapid and accurate testing, increasing integration of ai-driven diagnostics. Major trends in the forecast period include increasing adoption of point-of-care diagnostic devices, rising demand for molecular and immunoassay testing, growing use of automated laboratory systems, expansion of multiplex diagnostic platforms, enhanced focus on rapid testing.Global In Vitro Diagnostics Market Segmentation
1) By Diagnostic: Point-of-Care Diagnostics Devices And Equipment, Immunochemistry Diagnostic Devices And Equipment, Clinical Chemistry Diagnostics Devices And Equipment, Molecular Diagnostics Devices And Equipment, Microbiology Diagnostic Devices And Equipment, Hemostasis Diagnostic Devices And Equipment, Hematology Diagnostic Devices And Equipment, Immunohematology Diagnostic Devices And Equipment 2) By Type of Expenditure: Public, Private 3) By End User: Hospitals And Clinics, Diagnostic Laboratories, Other End Users Subsegments: 1) By Point-Of-Care Diagnostics Devices And Equipment: Blood Glucose Monitoring, Pregnancy And Fertility Testing, Infectious Disease Testing, Cardiac Markers Testing, Coagulation Testing 2) By Immunochemistry Diagnostic Devices And Equipment: Enzyme-Linked Immunosorbent Assay (ELISA), Radioimmunoassay (RIA), Western Blot, Immunofluorescence Assay, Chemiluminescence Immunoassay (CLIA) 3) By Clinical Chemistry Diagnostic Devices And Equipment: Blood Glucose Testing, Cholesterol Testing, Liver Function Testing, Kidney Function Testing, Electrolyte Testing 4) By Molecular Diagnostics Devices And Equipment: PCR (Polymerase Chain Reaction) Instruments, Next-Generation Sequencing (NGS) Instruments, Microarray Instruments, FISH (Fluorescence In Situ Hybridization) Devices 5) By Microbiology Diagnostic Devices And Equipment: Microbial Culture Systems, Automated Identification Systems, Antimicrobial Susceptibility Testing Systems, Blood Culture Systems 6) By Hemostasis Diagnostic Devices and Equipment: Coagulation Analyzers, Thromboelastography Systems, Platelet Function Analyzers, Clotting Factor Analyzers 7) By Hematology Diagnostic Devices and Equipment: Hematology Analyzers, Hemoglobin Testing Systems, Complete Blood Count (CBC) Devices, Blood Cell Counters 8) By Immunohematology Diagnostic Devices and Equipment: Blood Typing Systems, Crossmatching Systems, Antibody Screening Systems, Blood Bank AnalyzersWhat Is The Driver Of The In Vitro Diagnostics Market?
An increase in the prevalence of cancer is anticipated to propel the growth of the in vitro diagnostics market. Cancer encompasses a diverse range of over 100 diseases that have the potential to emerge in various locations throughout the body. The rising prevalence of cancer is due to lifestyle changes, including increased tobacco use, poor diet, physical inactivity, and environmental pollution, which collectively heighten cancer risk across populations. In vitro diagnostics (IVDs) represent a category of tests or medical devices that analyze samples extracted from the human body, furnishing crucial information for the screening, diagnosis, and treatment of cancer. For instance, in January 2023, according to the National Center for Biotechnology Information, a US-based intergovernmental organization, in the United States there were 1,958,310 new cancer cases, and 609,820 cancer deaths are projected to occur in 2023. Therefore, the increasing prevalence of cancer is expected to propel the growth of the in vitro diagnostics industry.Key Players In The Global In Vitro Diagnostics Market
Major companies operating in the in vitro diagnostics market are Siemens Healthineers, F. Hoffmann-La Roche AG, Danaher Corporation, Abbott Laboratories Inc., Thermo Fisher Scientific Inc., Agilent Technologies Inc., Alere Inc., Arkray Inc., Becton Dickinson and Company (BD), Bio-Rad Laboratories Inc., bioMérieux SA, Grifols SA, Hologic Inc., Werfen SA, Laboratory Corporation, Luminex Corporation, Ortho Clinical Diagnostics, PerkinElmer Inc., Qiagen NV, Quest Diagnostics Inc., QuidelOrtho Corporation, Sysmex Corporation, Illumina Inc., Menarini Silicon Biosystems SPA, SpeeDx Pty. Ltd., GENSPEED Biotech GmbH, Accelerate Diagnostics Inc., Merck KGaA, Caris Life Sciences Inc., Bio-Techne Corporation, Myriad Genetics Inc., Genomic Health Inc., Exact Sciences Crop., Natera Inc., Invitae Crop., Veracyte Inc.Global In Vitro Diagnostics Market Trends and Insights
Major companies operating in the in vitro diagnostics market are adopting advanced technologies such as UIMG image technology and a 2D decoder chip to enhance the accuracy and efficiency of diagnostic tests, improve data analysis, streamline workflow processes, and ultimately provide better patient outcomes. UIMG image technology is an advanced imaging method used in diagnostics to capture and analyze high-resolution images for improved visualization and interpretation of biological samples. For instance, in November 2023, a Newland, EMEA, Netherlands-based technology company launched the IVD (in vitro diagnostics) product line. The new IVD product line consists of five barcode scanners that are compact, rugged, and optimized for automated diagnostic processes. These scanners aim to improve the speed and accuracy of diagnostics, which is crucial given the increasing demand for efficient healthcare solutions.What Are Latest Mergers And Acquisitions In The In Vitro Diagnostics Market?
In June 2025, bioMérieux, a France-based provider of in vitro diagnostics solutions, acquired Day Zero Diagnostics for approximately $25 million. With this acquisition, bioMérieux aims to strengthen its next-generation sequencing (NGS) capabilities and enhance its rapid diagnostics portfolio to better address antibiotic-resistant infections and improve the management of life-threatening infectious diseases. Day Zero Diagnostics is a US-based infectious disease diagnostics company specializing in genome sequencing and machine learning technologies that enable rapid identification of bacterial pathogens and their antibiotic resistance profiles directly from whole blood samples.Regional Outlook
North America was the largest region in the global in-vitro diagnostics market in 2025. Asia-Pacific was the second largest region in the global in-vitro diagnostics market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are China, India, Japan, Australia, Indonesia, South Korea, Bangladesh, Thailand, Vietnam, Malaysia, Singapore, Philippines, Hong Kong, Taiwan, New Zealand, UK, Germany, France, Italy, Spain, Austria, Belgium, Denmark, Finland, Ireland, Netherlands, Norway, Portugal, Sweden, Switzerland, Russia, Czech Republic, Poland, Romania, Ukraine, USA, Canada, Mexico, Brazil, Chile, Argentina, Colombia, Peru, Saudi Arabia, Israel, Iran, Turkey, UAE, Egypt, Nigeria, South Africa.What Defines the In Vitro Diagnostics Market?
The in-vitro diagnostics devices and equipment market consist of sales of kits, reagents, piece of software, system, and instrument. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.In Vitro Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $152.42 billion |
| Revenue Forecast In 2035 | $241.14 billion |
| Growth Rate | CAGR of 11.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Diagnostic, Type of Expenditure, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Siemens Healthineers, F. Hoffmann-La Roche AG, Danaher Corporation, Abbott Laboratories Inc., Thermo Fisher Scientific Inc., Agilent Technologies Inc., Alere Inc., Arkray Inc., Becton Dickinson and Company (BD), Bio-Rad Laboratories Inc., bioMérieux SA, Grifols SA, Hologic Inc., Werfen SA, Laboratory Corporation, Luminex Corporation, Ortho Clinical Diagnostics, PerkinElmer Inc., Qiagen NV, Quest Diagnostics Inc., QuidelOrtho Corporation, Sysmex Corporation, Illumina Inc., Menarini Silicon Biosystems SPA, SpeeDx Pty. Ltd., GENSPEED Biotech GmbH, Accelerate Diagnostics Inc., Merck KGaA, Caris Life Sciences Inc., Bio-Techne Corporation, Myriad Genetics Inc., Genomic Health Inc., Exact Sciences Crop., Natera Inc., Invitae Crop., Veracyte Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
